JOURNAL OF VIROLOGY, May1974, P. 1030-1036
Copyright©1974 American Societyfor Microbiology Printed inVol. 13, No. 5U.S.A.
Effect of Ionic Strength
onthe
Binding of
Sindbis
Virus
to
Chick
Cells
JOHN S. PIERCE,' ELLEN G. STRAUSS, AND JAMES H. STRAUSS DivisionofBiology, California Institute of Technology, Pasadena, California91109
Received forpublication14January1974
Sindbis virus can adsorb tochicken embryo fibroblasts intwodifferent ways.
"Loosely" boundviruscanbe washedoffthe cell with buffersofionicstrength 0.2 or greater, whereas "tightly" bound virus remains attached under these conditions. When Sindbis virus is adsorbedto chick cellsat4C froma bufferof ionic strength 0.17, 40 to 50% of the adsorbed virus is loosely bound, the remainder tightly bound. Infection of chick cells by Sindbis virus has only small effects onthe total amountofvirus thatcanbe boundtothe cells. However, the
amountof Sindbis virus thatcanbe tightlybound declines rapidly beginningat2
to 3 h after infection. By 7 h after infection, the amount of virus that can be
tightly bound is only 10 to 20% ofthe amount bound to uninfected cells. The adsorption (and penetration) of virusat37 C ismostefficientatanionic strength of 0.15to0.17; atthis ionicstrengthmostofthe adsorbed virus istightly bound. At higher ionic strengths the virus adsorbspoorly. At lower ionic strengthsmost
of the virus isloosely bound. A second enveloped virus, vesicular stomatitis virus, has been studied for the purposesofcomparison; its adsorption behavior differs
from that ofSindbis virus.
The first step in the infection ofa cell by a virus is the adsorption of the virus to a specific receptor onthecell surface. This is followed by a configurational rearrangement of the virus particle that leads to the loss of particle infec-tivity ("eclipse") and, in some way, to the introduction of the virus nucleic acid into the cell. Many ofthe details ofthese early events have been established for bacteriophage (1, 18, 20), but relatively littleis known in the case of animal viruses. Studies on the adsorption of poliovirus to HeLa cells in suspension culture demonstrated that the adsorption of virus fol-lowed classical kinetics(2,8);adsorptionoccurs efficiently at 0C. Eclipse and penetration, however, donot occurinthe
cold;
temperatures of 20C or higher are required, and theeclipse
event is separate from the adsorption event.
After eclipse of the virus particle, a noninfec-tious particle can be eluted from the cell that still contains all ofthenucleic acid andprotein
ofthe virion (8, 11, 14). Furthermore, the virus particle mayeclipse upon reaction with subcel-lular fractions, which presumably contain the virus receptor.
Enveloped animal viruses also appear to
'Presentaddress:DepartmentofMicrobiology, University
ofIllinois at theMedicalCenter,Chicago,Ill.60680.
adsorbtospecific receptors onthesurface ofthe susceptible cell. The receptors for the myx-oviruses and paramyxoviruses involve sialic acid, because removal of sialic acid from other-wise sensitivecellsrenders thesecellsunableto adsorb the virus (12, 16). In the case of en-veloped viruses, however, theevent correspond-ing toeclipseofthe picornavirusesappears tobe a fusion ofthe virus envelope withthe surface membrane ofthe cell (17).
We have been studying the adsorption of Sindbis virus, a group A
arbovirus,
to chicken embryo fibroblasts and the influence of ionic strength upon thisearly eventin infection.MATERIALS AND METHODS
Virus strains. Sindbisvirus was the HR strainof
Burge and Pfefferkorn (6). Vesicularstomatitis virus
(VSV) (Indiana serotype) was obtained from Phil
Marcus.
Chickcells. Chickenembryofibroblastswere
pre-paredfrom10-day-oldembryos.Cellswereseededat a
concentration of 1.5 x 106 cells per ml in Eagle
minimal essential medium(10)containingEarle salts
and 2% fetal calf serum. Medium and serum were supplied by Grand Island Biological Company. The
volumes seededwere 4.5ml per 60-mmpetri plateand 150ml per 800-cm2 rollerbottle. Cellmonolayerswere used3to5daysafterseeding.
10:30
on November 10, 2019 by guest
http://jvi.asm.org/
EFFECT OFIONIC STRENGTHON SINDBIS
ADSORPTFION
Preparation of "C-labeled Sindbis virus.Virus
was grownin mediumof low ionicstrengthwhereno
virus releaseoccurred andsubsequently harvestedin
mediumor bufferofhighionicstrength,atechnique suggestedbythe results of Waite and Pfefferkorn(22). Medium bathing a roller-bottle monolayer was
changed 3 h before infection with 50 ml of Eagle medium containing 2% fetal calf serum. The cells
were infected with virusat a multiplicityof 20PFU
per cell from 10 ml of phosphate-buffered saline
(PBS) (9), containing 1% dialyzed fetal calfserum
and1,ugofactinomycinDperml. After1h,thevirus
inoculum wasremovedand 40 ml of modified Eagle
medium was added. This medium was oflow ionic
strengthandcontained(i)0.06 M NaCl insteadofthe
normal0.116M, (ii) one-tenth of the normal
concen-tration of all amino acids except glutamine, which
waspresent atthe normalconcentration,and
methio-nine, whichwaspresentatone-twentiethoftheusual concentration, (iii) 2%dialyzedfetal calfserum,and
(iv) 1 mg of actinomycin D per ml. At 3 h after
infection the medium waschangedwith 40 ml of the
same medium, but lackingactinomycin D and
con-taining 0.25 MCi (50 MCi/mM) of ['4CImethionine (Schwarz/Mann) perml.
At12to 14h after infection theculture fluid was
removed and discarded, and the monolayer was
washed twice with 10-ml volumes of low-ionic-strength medium. The virus was then harvested by
washing the monolayer for15minat37Cwith 5or10 ml ofa solution ofhigh ionic strength (either Eagle
medium containing 0.216 M NaCl, or Tris-sodium
chloride bufferofionicstrength0.25). The monolayers
were usually washed a second timewith 5 ml ofthe same high-ionic-strength solution, and the virus
elu-ateswerepooled. Occasionally, aneluatewasusedas a virus stock without further purification. Usually,
however, the virus was purified bysucrose gradient sedimentation. Up to 75% oftheradioactivity in an
eluatesedimented withthe sedimentation coefficient of intact virus(Fig. 1).Peakfractionswerepooled and
dialyzedfor 2 hat4Cagainstalargevolume of PBS.
Insome casestheviruswasconcentratedby
precip-itation with polyethylene glycol (13, 15) and then layered onto a sucrose gradient. The "C-virus was
mixed withanequalvolumeofnonradioactive carrier
virus (prepared byinfectingcellsat lowmultiplicity
inEaglemediumcontaining3%fetalcalfserum), and
one-fourthvolumeof 40%polyethylene glycolin 2M
NaClwasadded. After1hat0C,theprecipitatewas
collectedbycentrifugationat10,000rpmfor45minor
at 15,000 rpm for 15 min, resuspended in a small volume of 0.2MNaClcontaining 50mM Tris,pH 7.4, and EDTA, and clarified by centrifugation at 2,000
rpmfor 10min. Therecoveryof viruswasgreaterthan
90% by this procedure. (It is important, however, that the ionic strength ofthe resuspending buffer be
greater than 0.2; otherwise, virusadsorbs to cellular debris and is lost.) For some experiments the virus wasprecipitated without addition of carrier virus; in these casestherecoveryofviruswasvariable,
averag-ing about 50%. The carrier virus preparation is most probably effective because of membrane
frag-ments and other cellular debris present that
co-precipitatewith the virus.
Preparation oflabeled VSV. The conditions for labeling VSV were similarto those used forSindbis virus, but the radioactive medium had an ionic strength of0.16. Approximately 12h afterinfection the entire culture fluidwasharvested; and the virus
was then concentrated andpurified bypolyethylene
glycol precipitation (no carrier virus was necessary)
and sucrosegradient centrifugation as described for
Sindbis virus. (The time and speed ofcentrifugation
wereadjusted for the higher sedimentation coefficient
ofVSV.)
Infection of cells for adsorption experiments. Chick cells in 60-mm petri plates wereinfectedata multiplicityof 30to50 PFUpercellfrom 1 ml ofPBS containing1%fetal calfserum.Aftera1-hadsorption
3000C
z 0
2000-/LL
l
a-10001PELLET
-10 20 30 40 50
FRACTION NUMBER
FIG. 1. Sucrose gradient sedimentation of
"4C-labeledvirus released fromchick cellsinto high-ionic-strength medium. Sindbis virus wasgrown in chick
cells in low-ionic-strength (0.10) medium containing ["CJmethionine. The monolayers were washed with medium of thesame ionic strength 13 h after
infec-tion, and then incubated briefly in medium ofionic strength 0.25. Anamount ofvirus equivalent to the yield for 25 cm2 of monolayer was subjected to sedimentation ina15to30%sucrosegradient contain-ing0.2MNaCI,0.05MTris, pH7.4,0.001MEDTA,
0.3%fetal calfserum.Centrifugationwasfor58minat 36,000 rpm and 23C in the SW41 rotor. Fractions
werecollected by drops from thebottom ofthe tube
and counted with I ml of water in 10 ml of a
dioxane-naphthalene-based scintillation fluid.
Sedi-mentation isfrom rightto left.
VOL.13, 1974 1031
on November 10, 2019 by guest
http://jvi.asm.org/
[image:2.494.251.443.242.491.2]PIERCE, STRAUSS, AND STRAUSS
period at 37 C, the inoculum was removed and 3 ml of Eagle medium containing 2% fetal calf serum was added.Mock-infected cells were treated similarly but without virus.
Adsorption of"C-labeled virus. For experiments
at4 C,petri plates were removed to the cold room and allowed to chill. Themedium was removed by aspira-tion, theplates were allowedtodrain tipped on edge, andanyresidual medium was removed. A 1- or 0.5-ml portion of PBScontaining 1% fetal calf serum and 103
to 104 counts permin of "4C-labeledvirus wasadded
to each plate. The plates were rocked at 15-min intervals to redistribute the inoculum. After 90min of adsorption, the inoculum was removed by aspiration,
and the monolayer was washed twice with 5-ml portions of the appropriate buffer. The plates were drained well, and the monolayersweredissolved in2
ml of 1%sodium dodecyl sulfate (SDS).
Monolayers to be pretreated with high-ionic-strength buffer before virus adsorption were treated similarly,butafterremovalofthe culture fluid5ml of high-ionic-strength buffer (usually 0.2M NaCl
con-taining 0.01 M phosphate, pH 7.4) wasadded. This bufferwasremoved, and themonolayerswerewashed once with 5 ml of 0.15 M NaCl containing 0.01 M
phosphate before the virus inoculum was added. Adsorption experiments at 37 C were similar in
design, but manipulations were performed at room
temperature and adsorption stepsat 37C. The
mono-layerswere also incubated for 10min at 37C inthe wash solutionsused toremove unadsorbedvirus.
Assay ofbound virus. Monolayers dissolvedin 1%
SDSwere precipitatedwith trichloroacetic acid (20% final concentration). The precipitates werecollected
on DA membrane filters (Millipore Corp.), dried thoroughly at 60C, and counted in 5 ml of toluene-fluor.
RESULTS
Effect of ionic strength upon the adsorp-tion of Sindbis virus to uninfected cells at 37 C. RadioactiveSindbisvirus wasadsorbedto
monolayers of chick cells for 75 min at 37 C fromPBSsolutionscontainingdifferent concen-trationsofNaCltovary the ionicstrength.After adsorptionthemonolayerswerewashedat37 C with either PBS of the same ionic strengthas
used for adsorption, or with a solution ofhigh ionic strength (0.25). The results presented in Fig. 2 show that the optimal ionicstrength for virus adsorption under these conditions is 0.15 to 0.17, where
approximately
20% oftheinput
radioactivity was adsorbed to the cells. Eighty
to 90% of the adsorbed virus was resistant to
elution with buffer of ionic strength 0.25. This probably represents virus that has successfully initiated an infectioncycle, because the
particle
to plaque-forming unitratio is 3 to6under the conditions ofvirus preparation and
adsorption
to cells
(unpublished
results).0
wLJ m 0 (I)
a
4)ru500
a-u
0.10 0.15 0.20 0.25
IONIC STRENGTH OF ADSORPTION FIG. 2. Adsorption of "C-labeled Sindbis virus to
chick monolayers in solutions of different ionic
strengths. Sin'dbis virus, labeled with
[14Cjmethio-nine, was diluted into buffers of differing ionic
strength. These buffers consisted of PBS (9), but the
NaCIconcentration wasvariedsothat the finalionic
strength ranged from 0.1 to 0.25; the buffers also contained 1%fetal calfserum. Viruswasadsorbedto monolayers of chickcells in60-mm petriplates from 0.5 mlofbuffer (containing 4,400counts/min)for75 min at 37C. The inocula were removed and the
monolayers incubated for20minat37C in 2.5 ml of
buffer of the same ionic strength (a) or of ionic
strength 0.25 (0). The monolayers were then
dis-solved in 1% SDS, precipitated with trichloroacetic
acid,and assayed forradioactivity.
Ationicstrengthsgreaterthan0.17,relatively little adsorption occurred, but nearly all of the virus that did adsorb resisted elution with buffer of ionic strength 0.25. At ionic strengths less than 0.15, virus adsorbed well, but 50% of this could be eluted subsequently with high-ionic-strength buffer. Thus, mu'h of the virus adsorbed at low ionic strength is "loosely" bound, whereas most of the virus adsorbed at
ionic strengths greater than 0.15 is "tightly" bound.
Effect of ionicstrengthupontheelution of virus adsorbed to cells at 4 C. "C-labeled Sindbis virus was adsorbed to monolayers of
chick cells from PBS(ionic strength 0.17) for 90
min at 4C. The monolayers werethen washed with sodium chloride-phosphate solutions of various ionic strengths, and the radioactivity
I 00
1032 J. VIROL.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:3.494.260.451.73.310.2]EFFECT OFIONIC STRENGT
remaining adsorbed to the monolayer was as-sayed. The results areshown in Fig. 3.
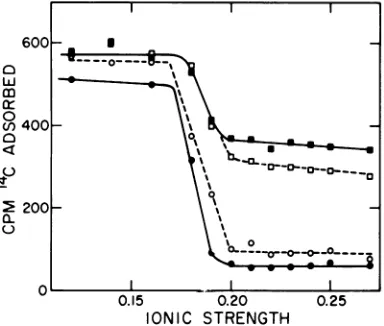
Under these conditions, about 50% of the virus is adsorbed to the cells. As the ionic strength of the wash solution is raised above 0.17, however, much of the adsorbed virus is eluted. The dependenceupontheionicstrength
of the wash solution is very steep. When the monolayerswerewashedwithasolution of ionic strength 0.20 approximately one-half of the adsorbed virus is eluted, indicating that much ofthe virus adsorbed at 4C is only "loosely" bound. Thus, only20to25%ofthe applied virus is adsorbed to cells in a manner resistant to washing with an ionic strength of 0.20; after washing with an ionic strength of 0.2, an
amountofvirus is left that isequivalenttothat which can bind and initiate infection at 37 C.
In some experiments, the amount of virus adsorbed to the cells is greater when the wash
6C
00
a
m
0
°f4C
0
2 20
(-)
0.15 0.20
IONIC STRENGTH 0.25
FIG. 3. Elution of Sindbis virus adsorbedtochick cells by washing with buffers varying in ionic
strength. "C-labeled Sindbis virus was diluted into
PBS (9) containing1%fetalcalfserumand adsorbed
to monolayers of chick cells outof 1 ml (containing 900counts/min) for 90 minat4C. The inocula were
removed, and the monolayers were washed twice at 4C with sodiumchloride-phosphate buffers ofuarious
ionicstrengths. These wash buffers contained 0.01 M phosphate, pH 7.4, and NaCl from 0.1 M (ionic strength 0.12) to 0.25 M (ionic strength 0.27). The monolayers were then assayedfor bound radioactiv-ity. Symbols: U, uninfectedcells,notprewashed; O,
uninfected cells, prewashed with sodium chloride-phosphate of ionic strength 0.2followed by sodium chloride-phosphate ofionicstrength0.17beforevirus adsorption; *, cells 7.2 h after infection by Sindbis
virus, notprewashed; 0,cells7.2hafter infection by
Sindbisvirus, prewashed with sodium chloride-phos-phate of ionic strength 0.2followed by sodium chlo-ride-phosphate of ionic strength 0.17 before virus adsorption.
'H ON SINDBIS ADSORPTION 1033
solution hasanionicstrength of 0.25 than when the ionic strength is 0.2. This effect is not
always seen.
The behavior of virus boundtoinfectedorto
uninfected cells is similar in response to ionic strength of the wash solution, but less virus is bound to infected cells. This effect is further explored in the following sections.
Prewashing the cells, either infectedor unin-fected, with sodium chloride-phosphate solu-tions of ionic strength 0.20 to 0.25 had little effectonthesubsequent adsorption and elution ofSindbis virus.
Decline in tight binding of the virus after Sindbis infection. The experiment shown in Fig. 3 demonstrates that the amount of virus bound to infected cells orto uninfected cells is approximately the same when the monolayers arewashed with buffers of ionicstrength 0.17or
less after virus adsorption. However, the
amount ofvirus "tightly" bound, in a fashion resistant to washing with ionic strength 0.2, is markedly less for infected cells. In the
experi-ment in Fig. 3, the amount of virus tightly bound to cells infected for 7 hby Sindbis virus was only about 20% of that which was tightly bound touninfected cells.
Todetermine the time of loss oftightbinding by infected cells, chick cells wereexamined for
20004P* 10-_l9
w
mi
HOURS AFTER INFECTION
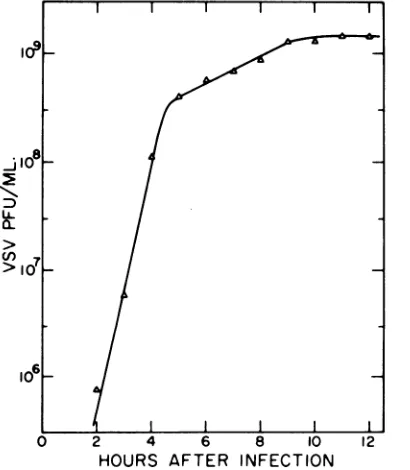
FIG. 4. Adsorption of Sindbisvirustochickcellsat
various times after infection with Sindbis virus. '4C-labeledSindbisvirus inPBS containing 1%fetal calfserum wasadsorbed tomonolayers of chick cells at various times after Sindbis virus infection. The adsorption period was 90 min at 4 C out of 0.5 ml containing 5,400 counts/min. The monolayers were then washed twice at 4C with sodium chloride-phosphate butfers of ionic strength 0.17(0) or 0.27, (0), and the radioactivity remaining bound to the
monolayerswasassayed. Agrowth curveof the virus isshownfor thesameexperiment (A).
II
0---0-)o
-'
10~~ ~ ~
~~0-?--O--O--olI II I
on November 10, 2019 by guest
http://jvi.asm.org/
[image:4.494.48.240.284.447.2] [image:4.494.252.441.374.540.2]PIERCE, STRAUSS, AND STRAUSS
their ability to adsorb Sindbis virus at various timesafter Sindbis infection. Radioactive virus wasadsorbedto the cells for 90min at 4 C, and the cells were then washed with sodium chlo-ride-phosphate solutionsthat were either0.17 or 0.27 in ionicstrength. The results are shown in Fig. 4. Also plotted is a growth curve of the virus performed at the same time to indicate the course ofthe infection.
Most of the loss in tight binding occurs between 2and4h afterinfection, with a slower, continueddecline thereafter. At 3 h after infec-tion, fewer than 10 PFU/cells have been re-leased into the culture fluid. Thus, the reduc-tion in binding beginswhen less than 1% of the 8-h virus yield hasbeen produced. Virus is still beingactivelyproduced at 8 h; cells continue to yield virus underthese conditions until about 12 h after infection.
In the experiment of Fig. 4, there is also a progressive declineinthe total amount of virus bound bythe infectedcells.Thetotal amount of virus bound byinfected cells has been variable fromexperiment to experiment, butmost often results comparable to Fig. 3 have been ob-tained, where the total amount of virus bound to infected cells is only slightly less than that bound byuninfected cells.
The decrease in binding of Sindbis virus to Sindbis virus-infected cells is accompanied by anincrease in thebindingof anunrelated virus, VSV, (Table 1) implying that loss of Sindbis bindingis notduetochangesincellsurfacearea butrathertoconfigurational changes occurring at the cell surface.
Binding of VSV to cells. VSV, a bullet-shaped virus, is another enveloped virus that matures by budding, but is quite different in structure from Sindbis virus. For the purposes
TABLE 1. Binding of 14C-labeled VSVtochick cells after Sindbis virusinfectiona
4C-VSVadsorbed
(counts/min)
Chick cells
After0.17' After0.25'
wash wash
Uninfected... 620 410
Sindbis-infected ... 1,010 520
aVSV(11,000counts/minin1ml)wasadsorbedfor
90min at 4 Ctochickmonolayersofeitheruninfected
cells, or cells5h after infection with Sindbis virus. The monolayers were then washed with buffers of ionicstrength0.17or0.25,and theamount ofbound viruswasassayed.
b Ionic strength.
ofcomparison, the binding of VSV to uninfected andinfected cells as a function of ionic strength wasexamined. For reference, a growth curve of VSV under our experimental conditions is shown in Fig. 5. This may be compared with the growth curve of Sindbis virus in Fig. 4.
The binding of VSV to cells as a function of ionic strength is shown in Fig. 6. In contrast to the results with Sindbis virus, VSV binds equallywelltocells infectedfor 8h withVSVas it does to uninfected cells. In addition, the dependence upon ionic strength for elution of the virus is not as marked.
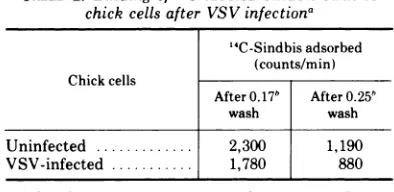
Sindbis virus, however, binds somewhat less readilytoVSV-infectedcells than to uninfected cells (Table 2). This difference in binding is small in comparison with thatbetween Sindbis virus-infected and uninfected cells, especially when the ionic strength of the wash buffer is 0.25.
DISCUSSION
Sindbisvirusappears to adsorbtochickcells in two different ways, which can be referred to as "loose" binding and "tight" binding. At 4 C loosebinding occurs at anionic strength of 0.17 or less with bothinfected and uninfected cells,
[image:5.494.264.469.368.603.2]HOURS AFTER INFECTION
FIG. 5. GrowthcurveofVSV.Monolayers ofchick
cellswere infected with VSVat a multiplicity of17. After various times at 37C the number of plaque-forming units ofvirus released into theculturefluid wasassayed.
1034 J.VIROL.
on November 10, 2019 by guest
http://jvi.asm.org/
900
m
0
°l)600
a
c:
E 300
a-0.15 0.20
IONIC STRENGTH 0.25
FIG. 6. Effect of ionic strength upon elution of VSV adsorbed to chick cells. VSV,labeled with [14C]-methionine, was diluted to 5,550counts per min per ml in PBS containing 1% fetal calfserum. One-ml portions were adsorbedfor 90 min at 4 C to
[image:6.494.42.239.58.237.2]mono-layers ofuninfected cells (0) or to cells which had been infected for8 h by VSV (0). The monolayers were then washed twice with sodium chloride-phos-phate buffers of various ionic strengths, and the amountofradioactivityremainingboundwasassayed.
TABLE 2. Binding of "C-labeled Sindbis virus to chick cells after VSVinfectiona
'4C-Sindbis adsorbed
(counts/min)
Chick cells
After0.17b After0.25b
wash wash
Uninfected ... 2,300 1,190 VSV-infected ... 1,780 880
aSindbis virus (4,400 counts/min in 1 ml) was
adsorbed for 90 min at 4C to chick monolayers of
either uninfected cellsorcells8 hafter infectionwith
VSV.Themonolayers werethen washed with buffers of ionicstrength0.17 or0.25, and theamountofvirus boundwasassayed.
b Ionicstrength.
and is readily reversed
by
exposure to ionic strengthsin excess of0.2.At 37 C, adsorptionof virus is complicated by penetration events. However, a loose binding does occur when adsorption occurs at ionic strengths less than 0.15. At an ionicstrength of0.17, loose binding either does not occuror is unstable.Atight bindingofvirus tocells can also occur at 4 C. Virus bound in this way remains at-tached even when the cells are washed with buffers of high ionic strength (although very littlevirus will attach at ionic strengths greater than 0.17). In the case of uninfected cells,
tightly bound virusaccounts for approximately 50% ofthe virus adsorbed, butonlyabout 10 to 20% of the virus adsorbed to Sindbis virus-infected cells is tightly bound. Virus adsorbed at 37 C that resists elution into buffers ofhigh ionic strength is probably virus that has eclipsed and penetrated.
The sites on the cell surface responsible for tightbindingofSindbisvirusdecline earlyafter infection, beginning at a time when only asmall fraction of the final virus yield has been pro-duced. Loose binding of virus is affected rela-tively little. We have considered thepossibility that loss of these sites is due to saturation by progeny virus particles, but this seemsunlikely because the loss beginsearly and loosebinding is not appreciably affected. In addition, one would expect a similar phenomenon to occur with VSV, which also releases progeny virus into the medium, but it does not. The loss of tight binding sites coincides with several other eventsreflectingchangesinthe cellsurface at 3 to 5 h afterinfection. Cells becomeagglutinable at this time by several of the plant lectins (4), cellsacquiretheabilitytohemadsorb red blood cells (7), budding virus can be detected in the electron microscope (3), and the cells will ad-sorb increased quantities oftheunrelated virus VsV.
The loose binding ofSindbis virus may be a nonspecificadsorptionofthevirus,unrelatedto virusinfection. Alternatively, adsorption ofthe virus may be a two-stage event. Virus is first loosely bound, then a certain fraction of this bound virus undergoes a second event that converts the viruses to being tightly bound. Tight binding of the viruses could be due to attachment of the virus to several receptors simultaneously. In this case, the loss of tight binding after Sindbis infection could be due eitherto a general reductionofvirus receptors, makingattachmenttomultiplesites less proba-ble, or to an alteration in the clustering of the sites on the cell surface.
Waite and Pfefferkorn (22) have shown that the interaction of Sindbis virus with cells is sensitive to ionic strength inanother way. Virus does not completethe budding process at ionic strengths less than 0.15, although the earlier
events inthe viruslife cycleoccur normally. In
addition, Pfefferkorn and Clifford (19) have shown that the virus is insoluble at ionic strengths less than 0.15. It is unclear at the present time what relationship exists between these phenomena and the ionic-strength de-pendence of virus adsorption, but it is clear
I I
_0
,
1035
on November 10, 2019 by guest
http://jvi.asm.org/
[image:6.494.40.237.347.443.2]PIERCE, STRAUSS, AND STRAUSS
that ionic surface charges play an important role in the interaction of Sindbis virus with the host cell.
ACKNOWLEDGMENTS
We acknowledge the excellent technical assistance of' Edith Lenches and Sharman Christoph. This work was
-;upported bygrantGM06965 from the Public Health Service.
grantGB31763X from the National Science Foundation, and
by a grant from the Merck Foundation. J. S. P. was a
P'ostdoctoralFellow of the DamonRunvon MemorialFund for
CancerResearch.
LITERATURE CITED
1. Adams, M. H. 1959. Bacteriophages. Interscience Pub-lishers,Inc. NewYork.
2. Bachtold,J.G., H. C. Bubel, and L. P.Gebhardt. 1957. The primary interaction of' poliomyelitis virus with hostcells of tissue culture origin. Virology 4:582-589. 3. Birdwell, C. R., E. G. Strauss, and J. H. Strauss. 1973.
Replication of Sindbis virus III. An electron
micro-scopic study of virus maturation using the surface replica technique. Virology 56:429-438.
4. Birdwell, C. R., and J. H. Strauss. 1973. Agglutination of Sindbisvirusand of cells infected withSindbisvirus by plant lectins. J. Virol. 11:502-507.
5. Brown, D. T., M. R. F. Waite, and E. R. Pf'efferkorn. 1972. Morphology and morphogenesis of Sindbis virus
as seen with freeze-etching techniques. J. Virol.
10:524-536.
6. Burge, B.W., and E. R. Pfefferkorn. 1966. Isolationand characterization of conditioned-lethalmutantsof Sind-bisvirus. Virology 30:204-213.
7. Burge, B. W., and E. R. Pf'efferkorn. 1967. Temperature-sensitive mutantsof Sindbisvirus: biochemical
corre-lates ofcomplementation.J. Virol. 1:958-962. 8. Darnell, J. E., and T. K. Sawyer. 1960. The basis for
variation insusceptibility topoliovirus in HeLa cells. Virology 11:665-675.
9. Dulbecco, R., and M. Vogt. 1954. One-step growthcurve
ofwesternencephalomyelitis virusgrown in uitroand
analysis of the virus yields from single cells. J. Exp. Med. 99:183-199.
1(1. Eagle, H. 1959. Aminoacid metabolism in mammalian cell cultures. Science 130:432-437.
11. Fenwick,M. L., and P. D. Cooper. 1962.Early
interac-tionsbetweenpoliovirus and ERK cells:some observa-tions on the nature and signif'icance of' the rejected particles. Virology 18:212-223.
12. Gottschack. A. 1959. Chemistry of' virus receptors, p.
51-61. InF. M. Burnet and W. M. Stanley (ed.). The viruses,vol.III. Academic PressInc.. NewYork.
13. Hebert, 'r. T. 1963. Precipitation of plant viruses by
polyethyleneglycol. Phytopathology53:362-365.
14. Joklik, W. K.. and J. E. Darnell. 1961. The adsorption and early fate of' purified poliovirus in HeLa cells. Virology 13:439-447.
15. Kanarek. A.D.,and G. W. Tribe. 1967. Concentration of' certainmyxoviruses with polyethvleneglvcol. Nature (London) 214:927-928.
16. Marcus, P. I., and V. G. Schwartz. 1968. Monitoring molecules of the plasma membrane: renewal of' sialic acid-terminating receptors, p. 143-147. InL. A.
Man-son (ed.), Biological properties of' the mammalian
surface membrane. The Wistar Institute Press, Phila-delphia.
17. Morgan, C., and C. Howe. 1968. Structure and
develop-mentofvirusesasobserved in the electron microscope. IX. Entry of parainfluenza I (Sendai) virus. J. Virol. 2:1122-1132.
18. Newbold,J. E.. and R. L.Sinsheimer.1969.Theprocess
of infection with bacteriophage OX174.XXXII. Early
stepsin the infectionprocess:attachment, eclipse, and
DNApenetration. J. Mol. Biol. 49:49-66.
19. Pfefferkorn, E. R., and R. L. Clifford. 1963. Precipitation andrecoveryof Sindbisvirus from solutionsoflowionic strength. Virology 21:273-274.
20. Simon,L.D., and T. F. Anderson. 1967. The infection of Escherichia coli by T2 and T4 bacteriophagesas seenin
the electron microscope. I. Attachment and
penetra-tion.Virology 32:279-297.
21. Waite, M. R. F., D. T. Brown, and E. R. Pfefferkorn. 1972. Inhibition ofSindbis virus release by mediaoflow ionic strength:anelectron microscope study. J. Virol.
10:537-544.
22. Waite, M. R.F., and E. R. Pfefferkorn. 1970. Inhibition of Sindbis virus production by media of low ionic strength: intracellular events and requirements for reversal.J. Virol. 5:60-71.
1036 J.VIROI.