0022-538X/78/0026-0673$02.00/0
Copyright©1978 AmericanSociety for Microbiology Printedin U.S.A.
Effect of UV Irradiation
on
the Expression of Vaccinia Virus
Gene
Products
Synthesized in a Cell-Free System Coupling
Transcription
and
Translation
WALTER BOSSART, DONALD L. NUSS,* AND ENZO PAOLETTI
DivisionofLaboratories and Research, New York State Department of Health, Albany, New York 12201
Received for publication 7 December 1977
The effect of UV irradiation on the expression of the vaccinia virus genome
was investigated in a cell-free system coupling transcription with translation.
Exposure ofvaccinia virus to an increasing dose of irradiation resulted in differ-ential reduction in the syntheses ofvirus-specified polypeptides in the coupled
system,with sensitivity being proportional tothesize ofthe geneproduct.This
suggeststhateachtranslationallyfunctional mRNA species produced in vitro by vaccinia virus cores issynthesized from an individualpromotersite.
Vaccinia virus is alarge, DNA-containing vi- peptides bypolyacrylamide gelelectrophoresis.
ruswith a genomecomplexityofabout1.2x 108 Inthisreportwedescribe theeffects of increas-daltons(4, 6, 7,9). Synthesisof viralRNApro- ing UV irradiation of vaccinia virus upon the
ceedsreadilyunder invitro conditionsat alevel fmalexpressionofindividual viralgene products
which indicates that 40 to 50% of the entire in the
coupled
transcription-translation
system. genome is transcribed (19). This result suggeststhat the RNA synthesizedunder in vitrocondi- MATERIALS AND METHODS
tions couldeasilycode for asmany as 150 to 200 UV
irradiation.
Purified vaccinia virus particles proteins in the size range of104 to105
daltons. (strainWR), preparedasdescribedby Joklik (10), atAlthough in vitro-synthesized vaccinia virus aconcentration of 45 optical density(OD260)units per
RNA has generallybeen described as a family ml in 10 mMTris(pH 7.5) wereexposedto incident
of molecules with an average sedimentation UV irradiation of 5 ergs/s per mm2
(supplied
by avalue ofbetween 8 and 12S (11, 26), the purifi- 253.7-nm-wavelengthsourcesituated30cmabove the
cation and partial characterization of a virion- sample). Exposures were conducted with continuous
associated speciesofhigh-molecular-weightvac- stirring at room temperature in a plastic cap7 mm in
cinia RNAhaverecently been described. 17,18). diameter and2.5mmindepth, with the thickness of
ciniaRNA haverecentlybeendescribed (17,18). the virus particle suspension measuring less than 1
The results derived from anumber ofdifferent mm.
experimental approaches are consistent witha Invitro RNA and protein synthesis. A cell-free
precursor-product relationship between the protein-synthesizing system was prepared from
Ni-high-molecular-weight RNA and the virion-re- black wheat germ by the method of Roberts and
leased 8 to 12SmRNA (16-19). Paterson(21). RNAsynthesiswasfollowed
by
deter-The technique of UV transcription mapping mining the incorporation of[3H]UTP (NewEngland
developed by Sauerbier andcolleagues (22, 23) Nuclear Corp.) into trichloroacetic acid-precipitable
originally with bacterial systems has recently material which was collected on Whatman GF/C
beensuccessfullyemployed to determineth glass-fiber filters (14). Conditions for in vitro RNA
been successfuly
employee
todetermine
the synthesis in the absence of the translation system wereorder of tranScription of several animal virus asfollows.A
400-,ul
reaction mixture contained 50 mM genomes (1, 3, 8). We decided to apply this Tris-hydrochloride (pH 8.4); 10 mM MgCl2; 10 mMtechniquetothe vaccinia in vitrotranscription
dithiothreitol;
0.05%NonidetP-40;
2 mMeachATP,
system inanefforttodetectthe
transcription
of GTP, and CTP; 0.2 mM UTP; 40 ul of 0.5-mCi/mlmultiplegenes froma commonpromoter,oneof [3H]UTP; and 0.72 OD2we unit equivalent of virus.
thepossiblemodesoftranscription suggested by Incubation was at
37°C.
Conditions used for thethe synthesis of a high-molecular-weight pre- syntheses of RNA and protein in the coupled
tran-sumptiveprecursor tovacciniamRNA (16-19). scription-translation system are described in detail in
Sincegood resolution of individual invitro-syn- Results. Protein synthesis was monitored in this
sys-thesized viral mRNA's
been,
has notachieved,tem
bydeterminingtheincorporationof[S]methio-wecoupledthesizedscriptionofthe
mRNAievmed
viralnine
(New EnglandNuclearCorp.)into polypeptides
we coupled thetranscriptionof theviralgenome asdescribed by Mans and Novelli (13). Globin andto translation in a wheat germ protein-synthe- reovirus mRNA'swere obtained from C. Baglioni and
sizing system and resolved the resulting poly- A. J.Shatkin, respectively.
673
on November 10, 2019 by guest
http://jvi.asm.org/
674 NUSS, J. VIROL.
Polyacrylamide gel electrophoresis. Samples 800
containing [3S]methionine-labeledpolypeptidesfrom the coupledtranscription-translationsystemwere pre-pared foranalysis byaddinganequalvolume ofwater W
and 2 volumes of2xsample buffersothat the final
!
/concentrations ofsample bufferingredientswere0.1 O 600 M dithiothreitol, 2% sodiumdodecylsulfate, 0.08M C
Tris-hydrochloride (pH 6.8), 10%glycerol, and 0.4% o bromophenol blue. Thesampleswereboiled for 2 min 0 and applied to 15% sodium dodecyl sulfate-polyac- 400 rylamideslabgelspreparedaccordingtoAndersonet
4L0_
al. (2) by the method of Laemmli (12).After
electro-phoresis, gels were fixed and stained with 0.025%
(wt/vol) Coomassie brilliant blue in 40% (wt/vol)
methanol-7.5%(wt/vol)aceticacid, usuallyovernight. , 200 Gelsweredestained in severalchangesof 40%(wt/vol) u
methanol-7.5% (wt/vol)acetic acid and then dried in / vacuoandautoradiographedonKodak BB54 medical E
X-ray film. Anappropriateexposure timewaschosen a inwhich the film gavealinear responsetotheamount
ofradioactivity. Autoradiographswere scanned with 0 60 120 180 240
aBeckman model R-112scanningdensitometer.The MINUTES
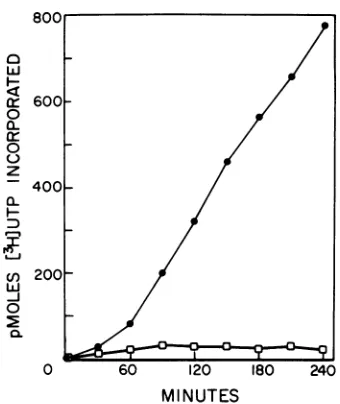
radioactivity incorporated into individual polypep- FIG. 1. RNA synthesis directed by vaccinia virus
tides wasestimatedbydeterminingtheareaunder the in a wheat germ cell-free translation system. The peaks in the autoradiograph tracings or measured reaction mixture contained
206
wheatgerm
extract,directly by cutting out the polypeptide bands and 15 mM creatine phosphate, 11 U of creatine
phos-detesrining
prev(ously
theamountofradioactivityas phokinase per ml, 16 mM N-2-hydroxyethyl pipera-described(15).zine-N'-2-ethanesulfuric
acid(pH7.5),
10mM dithi-othreitol, 110mMpotassium acetate, 0.3mM sper-RESULTS midine, 0.01% Nonidet P-40, 0.01 mMS-adenosyl-L-Syntheses of vaccinia virus RNA and methionine, 4 mMmagnesium acetate, 0.05 mM each
polypeptides in the wheat germ cell-free amino acids minus methionine, 1.7 mM ATP, 0.75
system. In considering thecoupling of vacinia mM GTP, 0.5 mM CTP,
200
mCi ofr3HJUTP
pervirus-directed RNAsynthesis totranslation, we 1tmolat a final concentration of 0.1mM;and1 OD20
first
dunit
ofpurified vaccinia virus per ml ofreactionfirst
determined
the level andrateof RNA sy- mixture. Incubation was at22.50C,
and13H]UTP
thesis under standard conditions used for
protein
incorporation was determined as described in thesynthesis
inthewheatgerm
translationsystem. text.Symbols:El,
r3HUTP
incorporation inthe ab-RNAsynthesisbegan slowlyandthenproceeded senceofvirus; 0,f3HJUTPincorporation directed bylinearly
under these conditions forover4h(Fig.
addedvaccinia virusparticles.
1).Although the initialrateof RNA
synthesis
inthe translationsystem at22.5°C andpH7.6was since no delay was observed in the synthesis of
approximately 50% that observed under stan- proteins directed by exogenous vaccinia RNA
dard transcription conditions at
370C
and pH (Fig. 2). Rather, the lag probably reflects the8.4
(data
notshown),
the final level of RNA time required for the energy-dependentextru-synthesiswasgreater in the translation system. sionof RNA from the viruscores(11).
This is because RNA
synthesis
nornally
ends The abrupt cessation in[3S]methionine
in-within30 to 40minwhen
transcription proceeds
corporation after 2 h of linear virus-directedintheabsence ofan
energy-regenerating
system proteinsynthesisappears to be due to thecon-(18). sumption or inactivation of some factor(s) in the
[3S]methionine
incorporation into polypep- wheat gern extract itself. Ofall
the reaction tides under conditions whichsupport bothtran- ingredientstested,only the wheatgerm
extractscriptionandtranslationin thewheat
germ
sys- permitted proteinsynthesistoresume. Thisef-temwas
dependent
upon theadditionof either fect cannot be attributedto RNase contamina-mRNA or vacciniavirus (Fig. 2).After addition tion of the system since addition of fresh RNA of virusparticlestothe reactionmixture,there failed to stimulate protein synthesis and3H-la-was a 30-to45-minlag inthe incorporation of beled vacciniamRNAwasstable, as determined
[3S]methionine
into polypeptides,followed
by by measuring acid-insoluble radioactivity, foralinear increaseinproteinsynthesisfor approx- severalhours in thetranslation system (data not
imately
2 h, atwhich time[wS]methionine
in- shown).corporationendedabruptly.Thelag phasewas Inpreliminary experiments,acomparisonwas
not a characteristic ofthe translation system, made of the level of translation directed by
on November 10, 2019 by guest
http://jvi.asm.org/
[image:2.501.285.455.64.266.2]8 efficient indirectingprotein synthesis than
vac-cinia RNA purified from the standard in vitro
/ transcription system. Thus, there is no
indica-tion thatgross quantities oftranslationally
in-O 6 active mRNA are
synthesized
and releasedby
/ vaccinia virus cores in the coupled system. At
the same
time,
these measurements do not ruleoutthepossibilitythat afraction of these RNA
/k
species
arenonfunctional.[3S]methionineincorporation in the
virus-di-rected
coupled
system wasoptimized
withre-spect to each reaction ingredient (Table 1). As
previously determined for the translation of
ex-e 2- ogenousvaccinia RNA (25), the translation of
vaccinia virus RNA synthesized directly in the
coupled system was also dependent upon the
methyldonorS-adenosylmethioninebeing
pres-ent
during
RNAsynthesis.
Nonidet P-40 was0 60 120 180 240 required for RNA synthesis to proceed; however,
[image:3.501.49.224.55.276.2]ithadlittle effectonthe subsequenttranslation
MINUTES of the RNA over a concentration range of 0.01
FIG. 2. Protein synthesis in the wheat germ cell- to0.1%.RNAsynthesiswas alsodependentupon freetranslationsystemdirectedbyvacciniavirus or thepresence of
Mg2s
iwiththeoptimumconcen-exogenous vaccinia RNA. [uS]methionine incorpo-
tratio
fre
protein ynthe
theocouped
-ration intoprotein wasdetermined, asdescribed in
the text, under reaction conditions similar to those tem beig 2 mM above the total nucleoside
described in the legendtoFig. 1, except that unla- TABLE
1.
Optimalconditionsofproteinsynthesisin beled UTPwaspresentataconcentrationof0.5mM,the
wheatgerm
cell-free
system coupling
and the reaction mixture contained45,uCi of["S]7-
thenwheati
byvcU-frus
particleitmethionine. Vaccinia virusRNAwasextractedfrom
transcrition
by vaccinia virusparticleswith
an in vitro transcription reaction by phenol
extrac-tion of the virus-free supernatant after removal of Reaction mixture constitutents Concn
virusparticles by centrifugation. The RNA wasfur-
Vaccinia virus"
1OD2w
unit/mi
therpurified bygelfiltrationonSephadex G-50equil-ATP.
1.70
mM
ibrated with0.01M sodiumacetate(pH5.0),0.05 MGTP*
0.75mM
NaCl, and 0.5% sodiumdodecyl sulfate, followed by
CTP
.. 050mMthreeethanolprecipitations (asthe sodium salt) to UTPc
0.10
mM remove traces of sodium dodecyl sulfate. Symbols:Total nucleoside
triphos-O,
f'S]methionine
incorporationdirected by addedphatesc
. 3.05mMvacciniavirusparticles; *,rS]methionineincorpo- Nonidet P
40.
0.01-1.1%
rationdirectedby exogenous vaccinia RNA;EL level
S-adenosyl-L-methionineb
.. 0.01mMof
J`S]methionine
incorporationobserved in the ab- Creatinephosphatec
.15.00 mMsenceof added virusorRNA. Creatine
phosphokinase'
11.00U/mi
HEPES-KOH (pH7.6)cd 20.00mM
vaccinia RNA extracted from an mi vitro tran- Mg acetateb
5.00
mMscription
reaction with the level of translation Kacetate"b
... 135.00mMobtained with othermRNA
species,
e.g.,globin
Amino acids minusmethio-and reovirusmRNA's.In
vitro-synthesized
vac- ninec 0.05 mM eachcinia mRNA formed functional initiation com- Spermidine" .. ... 0.03 mM
plexes efficiently in both the wheat germ and Dithiothreitol" ... 13.00 mM
thereticulocytecell-free system
(25).
Moreover, Wheatgermextract"b
.. 20.0OD20 units/mlthe level of amino acid
incorporation
obtained a The effect ofvarying theconcentrationofindivid-per microgram of input RNA suggested that ualreaction constituents on the level of
[3'S]methio-exogenous vaccinia RNA was
approximately
nineincorporationin thecoupledsystemprogrammed
70% as efficientasglobinmRNAin stimulating byvaccinia virusparticleswas determined.
protein synthesis (data not shown). From cal- "Reagent which showed a
clear
concentrationop-culationsof the amountof RNA
synthesized
in timum.thecoupled system, based onthelevel of [3H]- cReagentfor
which
noclearoptimumwasobserved,
UTPincoportionintRN
(Fi.itapparsbut
theconcentrationshownissaturatingforproteinUTPincorporation into RNA (Fig. 1), it appears
synthesis.
that the RNAsynthesized directly in the cell- dHEPES, N-2-hydroxyethyl
piperazine-N'-2-free translation system is only slightly more ethanesulfonicacid.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:3.501.246.439.353.554.2]676 BOSSART,
NUSS,
AND PAOLETTI J. VIROL.triphosphate level in the reaction mixture. Ad- 1 2 3 4 5
dition of nucleosidetriphosphatesoramino acids to concentration levels in excessof that shown
in Table 1 resulted in no increase in protein
synthesis. The saltoptimumforprotein
synthe-sisin thecoupled systemwasestablished as135 G
-mM potassiumacetate. When thechloride salt
ofpotassium was used, however, the optimum
was muchlower,presumablydue tothe inhibi- N S
-toryeffect of thechloride ion
(23,
25).
Distinct N-optima
were also observed for the amounts of -P 45virusparticles andwheatgermextractaddedto
the reaction mixture.
Analysis of
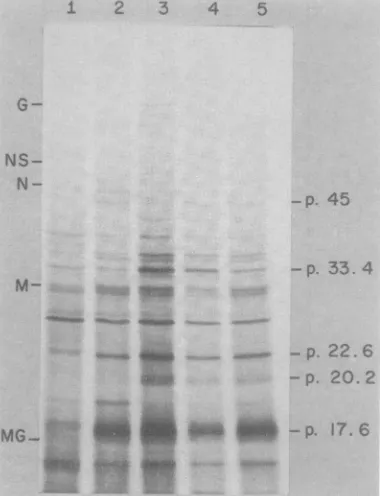
polypeptides synthesized
in -p. 33. 4the
coupled
system. 35S-labeledpolypeptides
M-synthesizedinthe
coupled
systemwereanalyzed
by
sodiumdodecyl
sulfate-polyacrylamide gel
blectrophoresisasshown inFig.3. Polypeptides
synthesized
in the wheatgermextract,directed - Pby RNApurifiedfrom the in vitro
transcription
- p. 20. 2reaction,areshown in lane1.
Approximately
30 ___to35
prominent
bandsareobserved,
ranging
in ' gjg 9 i - p 17. 6molecular weight from 10,000 to 50,000. When
proteinsynthesiswasdirectedby RNA
resulting
fromtranscription byvirus particles
directly
inthe translation system, the same
polypeptides
bands were made, but a greater proportion of FIG. 3. Sodiumdodecylsulfate-polyacrylamidegel
the higher-molecular-weight polypeptides was analysis of 35S-labeled translation products
pro-the m . s s . * .
rgrammed
by
exogenousvaccinia RNAorbypurified observed with molecularweights
in excess of virusparticles. Theautoradiograph shows the poly-65,000. Therelative synthesis ofindividual poly- peptidesresulting from translation ofexogenousvac-peptideschanged
dramatically
when the system cinia RNA (lane 1) and the translation productswas programmed by the virus rather than by synthesizedin thecoupled system atincreasingvirus
exogenous RNA; e.g., notice the changein the concentrations: (lane 2)0.5OD26ounit/ml;(lane 3) 1 levels ofsynthesis ofp-17.5, p-20.2, p-22.6, and OD26o unit/ml;
(lane 4)
1.5OD260units/ml;
and(lanep-45. (cf. lane 1 with lanes 2 to 5). We also 5) 2OD260 units/ml The lettersontheleftdenote the
observed that the virus concentration affected locationsofprotein markers, which include the
vesic-the relative synthesis ofindividualproteins (cf. ular stomatitis virus glycoprotein (G), minor protein
lanes 2
through 5).
Thisphenomenon appeared
(NS),
nucleocapsidprotein
(N),
andmembrane matrixlanes2through5).Thispprotein (M) with respective molecular weights of
to be dependent upon the actual virusparticle 66,000, 52,000, 47,000, and 30,000. MG denotes the concentration andnotupon therelative amounts positionofmyoglobin (molecularweight,
17,000).
The of viral RNA made in the system. A similar numberson therightindicatethecalculated molec-effect onthesynthesisofindividualpolypeptides
ularweights ofseveralpolypeptidebandsofinterest. was notobserved when theamountofexogenousvaccinia mRNA was variedover aconcentration upon theexpressionofviral geneproducts
range similar to thelevelofRNA
synthesized
in in the coupledsystem.High-molecular-weightthe coupledsystem
(data
notshown).
Also,
the RNAsaresynthesizedinvitro by vacciniavirus,gel profiles ofpolypeptides synthesized in the and these RNAs have a number of properties
coupledsystem did notchangewith timeduring consistent with their being precursors to 8 to
thereaction, i.e.,asmRNAsynthesisincreased, 12S mRNA (17, 18).Thisfinding stimulatedus
for reactionscontainingagivenconcentration of to investigate the mechanism of vaccinia virus virusparticles.Thus, it appears that the relative transcriptionbyusing thecoupled
transcription-synthesisand/orrelative translation of individ- translation system as a means ofresolvingand ual mRNA's is affected by the virus particle quantitating the viral geneproducts after expo-concentration rather than themRNAconcentra- sure of the virus to UV irradiation. Conse-tion. Whether this effect has any relation to a quently, we first determined the effect of UV control mechanism
normall_
operating in the irradiation on the infectivity ofthe virus (Fig. infectedcellsisnotpresentlyknown. 4A).Infectivity decreasedat anexponentialrate, Effect of UVirradiationofvacciniavirus witha37%
survival doseof60ergs/mm2.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:4.501.275.465.70.318.2]larly, RNA synthesis, as measured either in the resolved into three polypeptide bands having
standardinvitrotranscriptionreaction or in the sensitivities similartothose of theirimmediate
coupled system, decreasedwith single-hit kinet- neighbors; i.e.,theyfailedtoshow theresistance
icsafterexposureto UVirradiation, as did pro- exhibited by the unresolved band. The nature
teinsynthesisinthe coupled system directed by and significance of thispuzzling phenomenaare
virus particles exposed to increasing doses of
irradiation (Fig. 4B). Similar results were ob- '0 A 0 B
tainedwhether or not exposure of virus particles 8c
to UVirradiationwas preceded by sonic oscil- , 60
lation to disrupt aggregates. z E
When the viral polypeptide products which 4 0 40
were synthesized in the coupled system pro- w \
grammed byirradiated virus particles were an-
-alyzed by polyacrylamide gelelectrophoresis, it
was observed that the individual
polypeptides
20were differentially inhibited in their synthesis 0 20 60 0 100 200 300
(Fig.5). Thisexperimentwasrepeated a total of UV DOSE(sec)
five times, and in each case visual inspection FIG. 4. Effect ofUV irradiationon vacciniavirus
suggested that the sensitivity of the individual infectivity andon the rates ofRNA
synthesis
andproteinswas directly related to their molecular protein synthesis in thecoupled system directedby
weights. On severaloccasions, a diffuse band of UV-treated virusparticles. (A)Effect of UV
irradia-approximately
55,000 molecularweight
ap- tiononinfectivity. Purified
virusparticles
wereex-peapproxitel
55,000b
moleula
weight
ap-V
posedtoUVirradiation,
asdescribed in the text, andpeared to beconsiderably more resistantto UV the infectivity was subsequently determined on
L-cell
inactivationthan did other polypeptide bands in monolayers. (B) Reduction in RNA synthesis
(0)
andthatmolecular weight range. When gel electro- protein synthesis (0) programed by virus particles
phoresis conditions were altered to give maxi- which had been exposed to UV irradiation under
mum resolution in this region, the band was identical conditions.
1
2
3
4
5
6
7
8
9
G-
-p. 66
-p.
55
N-
-p.
45
J~I4~.
-p.
37.5
M
l£tZ
-p.
29.2
-p. 26
-p. 22.6
-p.
20.2
-p.
17.6
FIG. 5. Sodium dodecyl sulfate-polyacrylamide gel analysis of translation products synthesized in the coupledsystemprogrammed byvirusparticles exposedtoincreasing doses ofUV irradiation. As inFig. 3, the
lettersontheleftofthisautoradiograph show the positions of the marker polypeptides, and the numbers on therightdenotethecalculated molecularweights ofseveralprominent polypeptidebands. Lane 1,Translation productsprogrammedbyvirusparticles which receivednoUV irradiation.Lanes 2 to9,Translationproducts
programmedby virusparticlesexposedtothefollowing UV doses:40, 100, 140, 200, 300, 400, 600,and1,000s. Eachwellreceivedidenticalamountsofthecoupled cell-freeextract.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:5.501.240.437.127.236.2] [image:5.501.94.385.348.585.2]678 BOSSART, NUSS, AND PAOLETTI J. VIROL.
unclear since thedifferences in the relative
sen-sitivities of all other polypeptide bands remained
the same under the varying conditions of gel
0010/
analysis. In addition, it should be mentioned thatexposure of virus to UV irradiation resulted neither in the appearance of new polypeptides ' synthesized in the coupled system, nor in any 0.008 evidence of increasedearlyquitting (Fig. 5). w
Quantitation of the major discretepolypeptide
!Z*
bands over a molecular weight range revealed
thatthe reduction insynthesisofthesepolypep- o 0006
tides after exposuretoUVirradiation exhibited
!Z/
single-hitkinetics (Fig. 6) forseveral represent- > .
ative polypeptides. In addition, whentherates
of inactivation for the synthesis of 17polypep- z-tideswereplottedversusthemolecularweights >
of these polypeptides, a linear relationship re- /
sulted (Fig. 7). This relationship suggests that
0.002
-the mRNA's coding for the individual viral translationproducts synthesizedin thecoupled systemaretranscribed fromindividualpromoter
sites; i.e., they appear not to be derived from O I
polycistronicmRNA precursormolecules which 0 2 4 6 8
are the result oftranscription ofseveral genes MOLECULAR WEIGHT x 104
fromasinglepromoter site
~~FIG.
7.Relationship
between the rates ofinacti-froma
singlepromotersite.vation
and themolecularweightsoftranslationprod-uctssynthesizedin thecoupled systemprogrammed
100i by UV-treatedvirusparticles. From survivalcurves
suchasshown inFig. 6,theinactivationratesfor17
80 \\sR\^ polypeptide bandsweredetermined andareplotted
against the calculated molecular weights of the
re-- \ \\ \\ spectivepolypeptides.
60\ \ X \DISCUSSION
O -\ \ \ \ 8\ The in vitro
synthesis
of RNAby
vacciniaz virusparticles and the translation of the
result-< 40 ingtranscription products were successfully
cou-pled
inthewheatgermcell-freeprotein-synthe-Wr \ \ \ \ sizing system. Since evidence forthe synthesis
of a
high-molecular-weight
virion-associated
RNA species which may be the precursors to vaccinia virus8 to
12S
mRNAhasrecently
been reported(17,
18),we decided to use this coupled system as a means ofanalyzing the effect of UV20\ irradiation on the synthesis of the viral gene
products. Although coupling oftranscription to
0
\\ translation provides only an indirect method of analyzing and quantitating the survival of the viral genetranscripts after exposure of virus to 0) 200 400 UV irradiation, it is the only method immedi-ately available for this task, since resolution of
UV DOSE (sec) individual viral mRNA's has not been
satisfac-FIG. 6. Survivalcurves for severalrepresentative
torily
achieved. Inactivation of virus infectivityvirus-programed polypeptides. Thisfigure shows the 4A),a nsctinan of trslation effect ofincreased UVirradiation of virus particles
(Fig.
4A),transcription, and resultingtranslationon thesyntheses in the coupled system of five poly- in thecoupled system (Fig. 4B) did follow
single-peptides of different molecular weights. The calcu- hit kinetics after exposure ofthe virus to UV latedmolecularweights of the five polypeptides are irradiation.Analysisof thepolypeptides synthe-17,600
(LJ),
22,600 (A),29,000
(0),45,000
(0), and sized atthe direction of virus particles exposed80,000 (-). toincreasing doses of irradiation showed a
on November 10, 2019 by guest
http://jvi.asm.org/
[image:6.501.73.267.333.580.2]ferential inhibition of synthesis of individual the translation products to be identical to the
polypeptides (Fig. 5 and 6). Finally, a linear translation productsof 8 to 12S in vitro mRNA
relationship between the rates ofUV inactiva- (manuscript inpreparation). Additional
experi-tion and the molecular weights of individual ments in progress suggest that the sequences
polypeptidessynthesizedin thecoupled system translated arelocated at the 5' ends oftheselong
wasobserved (Fig. 7). We obtained no evidence transcripts. Third, a detailed comparison of the
whichwouldsuggestthe transcription of multi- polypeptides synthesized in thecoupled system
plegenes from a commonpromoter site. While programmed by virus particle with the
virus-this work was in progress, similar resultswere specific polypeptides synthesized in infected
reported byPelham (20) for a coupled system, cells has not yet been completed. However, a using amessenger-dependent reticulocyte cell- comparisonofthe cell-freetranslation products
freetranslationsystem. ofvaccinia invitro-synthesizedmRNA with the
There are,however,several limitationsinher- cell-freetranslationproducts of vaccinia mRNA
entinthesesystems.First, there is currentlyno isolatedfrompolyribosomesofinfected cells has
way ofdetermining the actual relationship be- revealed that many of the polypeptides have
tweenthe relative sizes of the polypeptides and similar migration rates on polyacrylamide gels
the corresponding mRNA molecules coding for (J. Cooper and B. Moss, personal
communica-thosepolypeptides; i.e., just because polypeptide tion). Furthermore, since evidence for a
high-A is twice the molecularweight of polypeptide molecular-weight presumptiveprecursor mRNA
B, itdoesnotnecessarily follow that themRNA has so far been obtained only under in vitro
coding for
polypeptide
Ais twice the sizeofthe transcription conditions (16, 17-19), wefeeljus-mRNA coded for
polypeptide
B. This uncer- tifiedinusingthe system in its present state oftainty most
probably
affects the resultsplotted
characterizationtoinvestigate possible modes ofin Fig. 7. Second, we have little information transcriptionas itproceeds in the purified viral
concerning therelationship between the relative particles.
abundanceofindividual mRNA'ssynthesized by Additional studiesarerequired both to
char-the virus
particle
and the relativesynthesis
of acterizethecoupledsystem, which should provepolypeptides in the coupled system as deter- a usefultoolin elucidating controlmechanisms
mined byquantitation after
polyacrylamide gel
operativeininfectedcells, andtodetermine theelectrophoresis.
Boone and Moss (5) have re- functional significance of the virus-associatedported thatasmallportionof thecomplexityof high-molecular-weightRNA.
in vitro vacciniamRNAispresentat a concen-
ACKNOWLEDGMENTS
tration of about 150-fold in excess of other
se-quences (J. Virol., in press). This may explainquences(JVirol.inpress).This
mayeFoundation
Walter Bossart is a recipient of a Swiss National SciencePostdoctoral Fellowship. This work wassup-why only 30 to 40 polypeptides, ratherthan a ported by Public Health Service grant 1RO1 GM23853-01
possible 200 to 300 vaccinia polypeptides, are from theNational Institute of General MedicalSciences.
detectedin cell-freetranslation systems.
Alter-nately,the
polypeptides synthesized
inthecou- LITERATURECITED
pledsystem may representthe translation
prod-
1. Abraham, G., and A. K. Banerjee. 1976. Sequentialuctsofonlyafraction of the bulk oftranscripts transcriptionof thegenes of vesicularstomatitisvirus.
normallysynthesized b the, .r il
a...
Proc. Natl. Acad.Sci.U.S.A. 73:1504-1508.normally
synthesized
by
the virus particles,
a 2.Anderson,
C. W., P. R. Baum, and R. F. Gestland.portion of which may be translationally non- 1973. Processingofadenovirus 2-induced proteins.J.
functional.Inthis
regard,
acomparison
ofiniti- Virol.12:241-252.ation
complex
formation and thelevel of amino 3. Ball,LA., and C. N. White. 1976. Order of transcriptionacid incorporation stimulated by vaccinia ofgenes of vesicular stomatitis virus. Proc. Natl.Acad.
Sci.U.S.A.73:442-446.
mRNAwith that observed for other functional 4. Berns, K. I., and C.Silverman. 1970. Natural occur-mRNAspeciesdoessuggestthat the RNAsyn- rence of cross-linked vaccinia virus deoxyribonucleic
thesized andreleased
by
vaccinia viruscoresis acid. J. Virol. 5:299-304.contaminated with nonfunctional 5. Boone, R.F.,andB.Moss.1978.Sequence complexity
not grossly contammatea wltn nontunctlonal and relative abundance of vaccinia virus mRNA's syn-mRNA (25; unpublished data). However, we thesized in vivo and in vitro. J. Virol.26:554-569.
cannotruleoutthepossibilitythat thepolypep- 6. Gangemi, J. D., and D. G.Sharp.1976.Use ofa
restric-tides expressed in the coupled system are the tionendonucleaseinanalyzingthegenomesfromtwo productsprodutsofofsequnces oundvery ear he 5'endsequencesfoundverynearthe 5' end
Geshelin,
different strains of vaccinia virus. J. Virol. 20:319-323.P.,and K.I.
Berns. 1974. Characterizationof several long transcriptional units, whereas and localization of the naturallyoccurringcross-linksin
other sequences that are
normally
expressed
vacciniavirusDNA.J.Mol. Biol.88:785-796.only after processing of these long transcripts 8. Goldberg, S., J. Weber, and J. E. Darnell. 1977.The
aresilent inoursystem. In this respect,wehave
definition
ofalargeL
iral
taping
bpy
effects
ofultraviolet
recently translated
purified
virion-associatedirradiation.
Cell 10:617-621.high-molecular-weightvaccinia RNAandfound 9. Grady,L.J.,and E.Paoletti. 1977.Molecular
on November 10, 2019 by guest
http://jvi.asm.org/
ity of vaccinia DNA and the presence ofreiterated atedRNA of vaccinia: apossible precursor to8-12S sequencesinthe genome.Virology79:337-341. mRNA.J.Biol.Chem.252:872-877.
10. Joklik,W. K. 1962. Thepreparationandcharacteristics 19. Paoletti, E., and L. J. Grady. 1977. Transcriptional of highly purified radioactively labelled poxvirus. complexity of vaccinia virus in vivo and in vitro. J. Biochim.Biophys. Acta61:290-301. Virol.23:608-615.
11. Kates, J.,and J.Beeson.1970.Ribonucleicacidsynthe- 20. Pelham,H. R.B.1977. Use ofcoupledtranscription and sisinvacciniavirus. I.The mechanism ofsynthesisand translation to study mRNA production by vaccinia release of RNA invaccinia cores. J.Mol.Biol.50:1-18. cores.Nature(London)269:532-534.
12. Laemmli, J. K. 1970. Cleavage of structural proteins 21. Roberts, B. E., and B. M. Paterson. 1973. Efficient duringthe assemblyof theheadofbacteriophageT4. translation of tobacco mosaic virus RNA and rabbit Nature(London)227:680-685. globin 9S RNAinacell-free system fromcommercial 13. Mans, J.R., and G. D. Novelli. 1961.Measurementof wheatgerm.Proc. Natl. Acad.Sci.U.S.A.70:2330-2334. theincorporationofradioactiveaminoacids into pro- 22. Sauerbier, W., and A. R. Brauetigam. 1970. A simple tein by a filter-paper disc method. Arch. Biochem. method of isolating RNA frombacteria. Biochim.
Bio-Biophys.94:48-53. phys.Acta 199:36-40.
14. Nuss, D.L., andE. J.Herbst. 1975. Stimulationof in 23. Sauerbier, W., R. L. Millette, and P. B. Hackett.1970. vitrotranscription ofT,DNAby the polyamine sper- Theeffects ofultraviolet irradiation on the transcrip-midine.Arch.Biochem.Biophys.169:513-521. tionofT4DNA.Biochim. Biophys.Acta209:368-386. 15. Nuss, D.L., and G. Koch.1976.Variationinthe relative 23. Weber,L. A., E. D. Hickey, P. A.Maroney, and C. synthesisofimmunoglobulinGandnon-immunoglobu- Baglioni. 1977. Inhibition of protein synthesis byCl-. linG proteinsincultured MPC-11cells withchangesin J. Biol. Chem.252:4007-4010.
the overall rate of polypeptide chain initiation and 25. Weber, L. A., E. D. Hickey, D. L. Nuss, and C. Bag-elongation.J. Mol.Biol. 102:601-612. lioni.1977.5'-Terminal7-methylguanosineandmRNA 16. Nuss,D.L., andE.Paoletti.1977.Methylgroupanalysis function: influence of potassiumconcentration on trans-ofvirion-associated high-molecular-weight RNA syn- lation in vitro. Proc. Natl. Acad. Sci. U.S.A. thesized in vitro by purified vaccinia virus. J.Virol. 74:3254-3258.
23:110-116. 26. Wei, C. M., and B.Moss. 1974. Methylation of newly 17. Paoletti,E. 1977. In vitrosynthesisofahighmolecular synthesized viral messenger RNA by an enzyme in weight virion-associated RNA by vaccinia. J. Biol. vaccinia virus. Proc. Natl. Acad. Sci. U.S.A.
Chem.252:866-871. 71:3014-3018.
18. Paoletti, E. 1977. High molecular weight
![FIG.MINUTESexogenousfreetherationdescribedbeledandtionO,threeremoveanvirustheribratedNaCl,methionine.sencevacciniarationof J`S]methionine 2](https://thumb-us.123doks.com/thumbv2/123dok_us/1528989.105493/3.501.246.439.353.554/fig-minutesexogenousfreetherationdescribedbeledandtiono-threeremoveanvirustheribratednacl-methionine-sencevacciniarationof-j-s-methionine.webp)