0022-538X/78/0027-0551$02.00/0
Copyright i 1978 AmericanSocietyforMicrobiology PrintedinU.S.A.
Involvement of
Mouse
Mammary
Tumor
Virus in
Spontaneous and Hormone-Induced Mammary Tumors
in
Low-Mammary-Tumor
Mouse
Strains
R.MICHALIDES,*L. VAN DEEMTER,R.NUSSE,G. ROPCKE,AND L.BOOT
DivisionofVirology, Antoni van Leeuwenhoekhuis, The Netherlands Cancer Institute,Amsterdam,
TheNetherlands
Received forpublication10March 1978
The involvement of themousemammary tumorvirus (MTV) in spontaneous
andhormone-inducedmammary tumorsin low-mammary-tumor mouse strains
wasstudied by comparing the amounts of MTV RNA and MTV DNA sequences in mammary tumors and other tissues of mice with and without hormonal
treatments. The following results were obtained. (i) Mammary tumors which
appearedinC3H miceas aresult of an infection with MTV contained more MTV
DNAcomparedwith noninfected organs; these mammary tumorsalsocontained
moreMTV RNA thanwas present inlactating mammary gland cells. (ii)
Hor-monal stimulation byadministration of excessive amounts ofprolactin via
hy-pophyseal isograftsinC3Hf and020miceresulted inanincreasedexpression of
MTV RNA inthemammaryglands.Thiselevated level of MTV RNAexpression
was, however, not maintained in the hormone-induced mammary tumors. (iii)
SpontaneousmammarytumorsinBALB/c mice contained similar levels of MTV
DNA and MTVRNA sequencesaswerefound in other cells of theseanimals.
Twotypes ofmousemammary tumor viruses
(MTV)arepresent in inbred strains ofmice,the
endogenous and exogenous mouse MTVs (1).
The exogenous MTV isusuallytransmitted via
the milkat nursing and is instrumental in the
earlydevelopment of mammarytumors in
sus-ceptible mice. The endogenous MTV is in its
proviral form transmitted as a gene and is in
some caseslinkedtothelateonsetof mammary tumors(26).
These routesof transmission of the MTV are
reflectedinthe MTV DNA
proviral
content ofmurinecellular DNA (15, 16).Allstrains ofMus
musculus contain 6 to 10 MTV
proviral
DNAcopiesof theendogenous MTVvariant in their
DNA. Mammarytumorswhichappearas a re-sult ofaninfectionbythe exogenous MTV con-tain inaddition anumberofproviralDNA cop-iesof the exogenous MTV variant in their DNA.
OnlytheEuropeanhigh-mammary-tumorstrain
GR s/a (17) contains 24 MTV DNA
copies
in the DNA ofallits cells, 16 of which belong to the Mtv-2 gene which controls the early and frequent appearance of mammarytumorsinthis strain (14a).The question whether MTV is involved in spontaneous, late-onset mammary tumors in low-mammary-tumor strains has not yet been solved. MTV variants have been isolated from spontaneous mammary tumors of C3Hf and
from mammaryglands of old BALB/c mice and havebeen shown to be oncogenic by injection in
susceptiblemice(13).Theiractualparticipation
in the spontaneous mammary tumors in these
strains, however, has not been determined. In
anotherlow-mammary-tumor strain, 020,MTV
antigens becamedetectableuponaging or
treat-mentwith X irradiation and urethane (2). This
ledBentvelzenetal.topostulate that MTV was
involvedin the formation ofall kinds of
mam-marytumorsinthemouse (2).
Another way ofinducingmammary tumors in
the low-mammary-tumor mouse strains is by
implantation ofmultiple hypophyseal isografts
in these mice(18).Prolactin is then continuously
released, and the excessive amountofthis
lac-togenic hormone causes high-mammary-tumor
frequenciesinalmost allmousestrains (5, 6).
The present study deals with the possible
involvementof the mouse MTV inspontaneous
and hormone-inducedmammary tumors in
low-mammary-tumor mousestrains. Tothis end we
comparedtheamountsofMTV DNAandMTV
RNA of mammary tumors, mammary glands, and otherorgans of thefollowingmousesystems: (i) C3H micecarryingtheexogenous, milk-trans-mitted MTVversusthe C3Hfstrain, freed from thatvirus byfosternursingnewbornC3H mice on mothers of a low-mammary-tumor mouse strain (C3Hf mice carry only an endogenous 551
on November 10, 2019 by guest
http://jvi.asm.org/
MTV variant transmitted via thegametes); (ii) hormone-treated 020 or C3Hf miceversus un-treatedcontrols; (iii)spontaneousmammary tu-morsof BALB/c miceversus mammary tumors ofBALB/c mice infected with MTV.
MATERIALS AND METHODS Mice.Mice ofhighlyinbred strainsmaintained at TheNetherlands Cancer Institute were used in these
experiments.As aruleallexperimentalanimals were
keptinplastic cages(threemice percage).Hormone treatment was carried out as describedpreviously(5) asfollows. Fourhypophysealisograftswereimplanted,
twointoeachkidneyof C3Hf femalesat anageof 6to 8weeks;onehypophysiswasimplantedinto the left
kidney ofcastrated 020 males at the age of 6 to 8
weeks, estrone (0.25 mg/liter) being added to the
drinking waterthroughoutthewhole lifespanof the mouse,while 3mgof progesteronewasgiven subcu-taneously once every week.
Mammarytumors,lactatingmammarygland, livers,
andspleenswereremovedfrom 5to10mice and used for nucleicacidextraction.
Virus.Thepropagationof MTVof theMm5mt/cl cell linehas beendescribedpreviously (10).The me-dium was removed from the rollerbottles at 3-day intervals, spun at 8,000 x gat 40C toremove cell debris, andstoredat40C.Twolitersof this medium was then concentrated 20-fold with an Amicon con-centrator, type HlP100 (Amicon, Lexington, Mass.).
This concentratewasthenlayeredover a5-ml cushion of 20% (wt/vol) sucrosein TNEbuffer(0.01M
Tris-hydrochloride, pH 6.8;0.15MNaCl;and 3 mMEDTA) andspunfor1hat100,000xgat40CinaBeckman
ultracentrifuge. Theresultingpelletwasresuspended
inTNEbufferandlayeredover apreformed20 to 60%
(wt/vol) sucrosegradientinTNE. After 16hof
cen-trifugationat100,000xgat40C,the virus band visible betweenbuoyant density valuesof1.16 to 1.20g/ml
wasisolated and concentratedbyultracentrifugation
and used either for thesynthesisofanMTV-specific 3H-labeled complementary DNA (cDNA) or for the isolation of MTV70SRNA. Rauscherleukemiavirus wasobtained fromplasmaofBALB/cmice infected with this virus andpurified by equilibrium
centrifu-gationin sucrose.
SynthesisofMTV-specific [3H]cDNA.
[3H]thy-midine-labeledDNAcomplementarytothe 70S RNA of MTV was prepared from MTV of the cell line
Mm5mt/cl.The MTV[3H]cDNAwassynthesized by usingaslightmodification of the method describedby Tayloretal.(25) withaDNase-digestedendproduct
of calf thymus DNA as primer in the endogenous
reverse transcriptase reaction. The reaction mixture of 500 ,d contained: 0.05 M dithiothreitol; 0.15%
(vol/vol) Nonidet P-40 (Shell, Rotterdam); 15 mM
potassium chloride; 20 mM magnesium chloride; 2.5 mg of calfthymusDNAprimerper ml (25);1.6mM eachdATP, dCTP,anddGTP;0.05mM
[3H]TTP
(50 Ci/mmol, The Radiochemical Centre, Amersham, U.K.);62.5mMTris-hydrochloride (pH 7.8);andan MTVconcentratecontaining0.6mg ofproteinfrom2 liters of tissue culture fluid of theMm5mt/clcell line. Thereactionwascarriedoutfor16hat37°C,and the[3H]cDNA was then further processed as described previously (15).
Preparation of cellular DNA. DNA was ex-tracted frompools of mammary tumors, livers, and
spleens from5 to 10animals.Tissuewasfinely minced,
homogenizedfor 30 s at maximal speed with aSorvall
homogenizer in TNE buffer containing 5% sucrose, and centrifuged at 3,000 x g for 10 min at 40C. The
resultingpellet was resuspended in 20 volumes of TNE buffercontaining1%sodiumdodecyl sulfateand 1 M sodium perchlorate. This preparation was then ex-tracted four times with TNE buffer-saturated PC mix
(phenol-chloroform 7:1, containing 0.17%
8-hydro-quinoline)and twice withchloroform-isoamylalcohol (100:1). After the addition of 1 volume of cold ethanol to the aqueous phase, high-molecular-weight DNA wasspooledout,dissolvedin3 mMEDTA and 0.4N
NaOH,and incubated at room temperature for 16 h. DNA wasfragmented by ultrasonification (Branson Sonifier, highestsetting),usingtwo 30-spulses, at a DNA concentration of approximately 50 ug/ml. A sedimentation value of 6 to 7Swasobtained for the DNAby thistreatment, asmeasured by alkaline su-crosegradient centrifugation.TheDNAsolution was neutralized byHCIafterthe addition of 0.1 volume of 1M Tris-hydrochloride (pH 8.3). DNA was then re-extracted withPCmix andprecipitated by adding 1
volume of cold ethanol. The DNA waspelletedby
centrifugation at 12,000 x g at 40C for 30 min and dissolved in 0.003 M EDTA (pH 8.3). The ratio of the
optical densityat 260nm to the optical density at 280
nm(OD2w,/2w)of the DNA was >1.80.
Preparation of cellular RNA and viral RNA.
Cellular RNAswereisolatedfrom the 3,000 x g
cyto-plasmic supernatant fractions remaining from the preparation procedure ofcellularDNA. Sodium
do-decyl sulfate (1%, wt/vol)wasadded to this
superna-tant, and this preparation was then extracted four times with TNE buffer-saturated PC mix and twice withchloroform-isoamylalcohol (100:1). Two volumes ofethanolandNaClto 0.4M wereadded to this RNA preparation, which was thenkeptat-20°C for 16h. The RNA waspelleted bycentrifugation at 5,000 x g for 30 min at-10°C, and the resulting pellet was
dissolvedin 3 mMEDTA, pH7.4. RNAsampleswhich
contained contaminating DNA as detected by the Burton assay (8) were treated with20,ugofDNase-1 perml(SigmaChemicalCo., St. Louis,Mo.) for 2 h at roomtemperatureand werere-extractedwith PCmix.
TheOD20/280ofthe RNA was >2.00. The RNA from
MTV and Rauscher leukemia virus was isolated as describedpreviously (14).
Molecularhybridizations. (i)Kinetic analysis.
Cellular DNA and MTV[3H]cDNA wereincubated for variousperiods oftimeat650Cin reactionmixtures
containing0.01MTris-hydrochloride(pH 7.8), 0.75 M
NaCl, 2 mM EDTA,0.05% sodium dodecyl sulfate, 0.01% diethylpyrocarbonate (Sigma), 10,000 cpm of MTV [3H]cDNA perml, and 5 mg ofcellularDNA per ml. All hybridization mixtures wereoverlayered
with paraffin oil to prevent evaporation. The DNA:DNAhybridizationreactions wereinitiatedby
boilingthesamplesat1000Cfor10min, after which thesamples weretransferredto65°C. Samples(100
,d)weretaken aftervariousCot values andassayedfor
on November 10, 2019 by guest
http://jvi.asm.org/
S-1resistanceasdescribedpreviously (3).
Cellular and viral RNAwereincubatedwith MTV
[3H]cDNA for aconstant period of timeat600C in similar hybridization mixtures, butnowwith
increas-ingamountsof murine cellular RNAorviral RNA in areaction volume of 25
pl.
ThefinalRNA concentra-tion in thehybridization mixtureswasadjusted by theaddition of yeast RNAto 30iLg ofRNA perml for viralRNA-MTV [3H]cDNA hybrid formations andto 120mg/ml for reaction mixtures containing cellular RNA. Theextentofhybridizationwasassayed by S-1
resistance aftera16-hhybridization reaction forCrot
values of 1.3x 105and lowerorafter 120 h forCrot
values >1.3 x105.Theresults arecorrectedto
stan-dard annealing conditions (7).
(ii)Hybridization of hIlabeled MTV 70S RNA and MTV [3HJcDNA. 70S RNAwas isolated from
MTV of the Mm5mt/clcelllineasdescribed before
(14) and labeled with '25I by using the methods of Prensky (20). The iodinationprocedure resulted inan
MTV [InI]RNA with a specific activity of 9 x l0r
cpm/ug. Amountscorrespondingto5,000cpmofthis MTV [t2I]RNA were incubated with increasing amountsof MTV[3H]cDNA (specific activity,2x107 cpm/,ug) in 10-pl hybridization mixtures containing 0.01MTris-hydrochloride (pH 7.8),1MNaCl, 2 mM EDTA, 0.05% sodium dodecyl sulfate, 0.01% diethyl-pyrocarbonate, and 3 mgofyeast RNAperml. The
hybridizationwasperformedtoacDNACot value of
0.2. Thehybridization mixtureswerethentransferred to 2 mlof 2x SSC (lx SSC, 0.15 MNaCl-0.015 M sodium citrate, pH 6.8). The sample wasdivided in twoparts,and50,ugof RNase A (Sigma,previously heatedat800Cfor15mintodestroy DNaseactivity)
was addedto oneportion. Bothportionswere
incu-batedat370Cfor 45minand thenassayedfor trichlo-roaceticacid-precipitable radioactivity (15), whichwas
counted for10min inaPackardgammacounter.
RESULTS
Characteristicsof themousestrains. Ta-ble 1liststhe mousestrainsusedin thisstudy, the hormnonal treatments, and the mammary
tumorincidences obtained. Thehighmammary
tumorfrequency in themousestrainC3His due
totheexogenousMTV which istransmitted via the milk. Removal of this exogenous MTV by fosternursing newborn C3Hmiceon a low-mam-mary-tumorstrain without themilk-transmitted MTV, such as C57BL, leaves a C3Hf mouse strain carrying only the endogenous MTV
var-iant.This endogenous MTV isexpressed in the C3Hf miceatlaterageandislinkedtothe late onset of mammary tumors in this strain (26). Theincidence ofmammarytumorsinthis C3Hf strain is dramatically increased in mice which have obtained ahypophyseal isograft (5). The 020 mouse strain seldom develops any mam-marytumors. Hypophyseal isografts implanted incastratedmales of this strainandcontinuous administration ofestrone and progesterone in-duce, however,mammarytumorsinthisstrain. The BALB/cmousestrainhasalowfrequency
of mammarytumors, which appear lateinlife. Animalsof thisstrainwhich havebeeninjected withMTV isolatedfrommammaryglands of old BALB/c females develop early appearing mam-marytumors (13).
Characterization of the single-stranded MTV
[3HJcDNA.
'The MTV [3H]cDNA was hybridizedtoitscomplementary MTV70SRNAtodeterminetheproportionoftheprobe which ishybridizabletoMTV 70SRNA. Atotal of 93% ofthisprobewashybridizedtoMTVRNAata
Crotvalue of2,whereas3%of theprobe hybrid-ized to Rauscher leukemia virus RNA at that
Crot value (Fig. 5a). The CrOtI/2 value of the MTV RNA:MTV [3H]cDNA hybrid formation
was4x 10-2.
Theproportion of the MTV 70S RNA that is represented in the MTV[3H]cDNA was
deter-minedbyhybridizing
1251-labeled
MTVRNAtoincreasing amounts of MTV [3H]cDNA as de-scribedabove. As shown inFig.1,72% of the 70S
['"I]RNA
from MTVwasresistanttoRNaseATABLE 1. Characteristicsofmousestrains
%
Man-Strain Exogenous MTV Treatment mary tu- Avg day
mors
9C3H(C3H/HeA) + - 79 305
9 C3H(C3H/HeA) + Forcedbreeding 93 255
9C3Hf - 7 655
9 C3Hf - +Ha 98 313
9 020(020/A) - 0
6 020 - +H 98 300
BALB/c(BALB/cHeA)b - Forcedbreeding 16 453
BALB/Cb +MTV(BALB/c) Forced breeding 90 198
aThese
animals
weretreatedwith hormones as described in thetext.bThese
data are from G. Ropcke, Ph.D. thesis, University of Amsterdam, Amsterdam, 1975, and from reference 13.on November 10, 2019 by guest
http://jvi.asm.org/
554 MICHALIDES ET AL.
t
t70
10 o
40- 30-20
10 0
1 10
[image:4.501.71.240.60.248.2]DNA:RNA ratio
FIG. 1. Hybridization between '25I-labeled MTV RNA and MTV[3H]cDNA. Hybridizationreactions contained5,000cpmofMTV['251]RNAand
increas-ing amounts ofMTV[3HJcDNA. The RNA:DNA
hybridswereperformedto acDNA Cotvalueof0.2 and were assayed for RNase A resistance as de-scribed in the text. Symbols: 0,percentage of 125I-labeled viral RNA that entered into an RNase
A-resistantfI251-RNA:DNAhybridat agivenratioof
MTV [3H]cDNA to MTV
['25IIRNA;
0, intrinsicRNase A resistanceof'251-labeledMTVRNA.
afterhybridizationwitha22-foldexcessof MTV
[3H]cDNAprobe,indicatingthatatleast 72%of
thenucleotidesequencesof MTV70SRNAwere
representedin the
[3H]cDNA
probe.Thecurvein Fig. 1 showed furthermore that the MTV
[3H]cDNA
probedidnotcontainpreferentiallytranscribed sequences of the MTV 70S RNA,
because 50% of the maximal
hybridization
was achieved at a 1:1 DNA-RNA ratio. The iodina-tionconditions forlabelingthe MTV RNA and theuse ofdiethylpyrocarbonate
in thehybridi-zationmixture may result in breakdown of
high-molecular-weight RNA,whichmightbea reason
that no
complete
hybridization values were reachedathighDNA-RNA ratios.Thermalsta-bility of the hybrids formed between MTV
[3H]cDNA
and various RNAs and DNAs was measuredto see whether anydifference in the outcome ofthe hybridizations could be due to qualitative differences between the hybrids formed. Figure 2 shows that the Tm's (i.e., the temperature at which50% of theradioactivelylabeled DNA is released from the RNA:DNA hybrid) of the hybrids formed between mam-mary gland RNA of O3H, C3Hf, and 020 and MTV
[3H]cDNA
areidentical tothe Tm (750C)of thehybrid forned betweenMTV cDNAand MTV 70SRNA.Also,nodifferenceinTm values of thehybridsformed between MTV
[3H]cDNA
and DNAs ofmammarytumors and organs ofC3H,C3Hf, 020, and BALB/c were found (data notshown), indicating no differences in the hy-bridized sequences.
Quantitation of MTV DNA sequences in normal and tumor tissues ofvarious mouse strainsby reassociation kinetics. The reas-sociation kinetics of MTV cDNAandDNA from mammary tumors and livers of C3H mice are shown in Fig. 3. More MTV DNA sequences were present in the DNA of C3H mammary tumorsthan in liver cells ofthe same mice. As
is known, these liver cells did not become
in-fected with theexogenous MTV (9). The
Cot1/2
value for the reassociation of C3H mammary tumor DNA with MTV [3H]cDNA was 250, compared with aCotI/2
of1,700forthe BALB/cunique sequence DNA. These data indicated
thatthereareapproximately six to seven copies
related to the MTV DNA per haploid genome in C3H mammary tumors. The
Cot1/2
valueforthe reassociation between C3H liverDNA and
MTV
[3H]cDNA
was 750, suggesting the pres-ence of two to three MTV DNA copies perhaploid genome in a C3H cell which is not
infected with the exogenous MTV.
Mammary tumors which appeared as a result of hormone treatment, involving a continuous
release ofthemammatropic hormone prolactin,
contained thesamenumber of MTV DNA
cop-iesas was present in the DNA of livercellsof these animals, as shown in Fig. 3 and 4. The
CotI/2
valuesfor the reassociations of DNAs from mammarytumorsandliversofhormone-treated C3Hf and 020 mice with MTV[3H]cDNA
amountedtoapproximately700, suggesting the presence of two tothree MTV DNA copies per
I
I
a
bL
0 a
20-
40-60 a
80~~~~~~~~~~~~~~~~
50 60708090100 5060708090100 temperature(t)
FIG. 2. Thermalstabilityprofileof the hybrids be-tweenMTV[3H]cDNAand variousRNAs.Hybrids
betweenMTV[3H]cDNAand RNAswereperformed
toCrot valuesatwhich maximalhybridizationhad
occurred, andportionswerethenkeptfor15minat
differenttemperatures andsubsequently assayedfor S-Inuclease resistanceasdescribed inthetext. The
hybridscontained MTV[3HJcDNAandRNAsfrom (a)MTV(A) and mammaryglandsof020(A) and
(b)mammaryglandsofC3H(0)andC3Hf(0).
J. VIROL.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:4.501.266.456.458.571.2]MTV IN SPONTANEOUS AND HORMONE-INDUCED TUMORS
.*IN
80
N
2 60
I
[image:5.501.65.416.57.299.2]log Cot
FIG. 3. HybridizationofMTV[3H]cDNAwith murinecellular DNA. Hybridization reactions contained 10,000 cpmofMTV[3H]cDNAand 5 mgofcellular DNA per ml and wereperformedasdescribed inthe text.
Samples (100
i1)
weretaken at variousCotvalues andassayedforS-inucleaseresistance. Thehybridization mixtures contained DNA ofmammary tumors ofC3H(0) and C3Hf+H (A) (treated with hormones asdescribedin thetext),ofthe organs(spleensandlivers)ofthesameanimals(0,A),andof calf thymus(0). The hybridization between unique 3H-labeled NIH-Swiss DNA and BALB/corgans DNA isgiven as a
referencecurve(x).
0
40
log Cot
FIG. 4. Hybridization ofMTV
rH]cDNA
with DNA ofmammary tumors of020 mice treated with hormones(0),oflivers andspleensofthese020 mice(0), andof calf thymus (O). ThehybridizationswereperformedasdescribedinthelegendtoFig.3.
haploidgenome in these tissues. Mammary tu-morswhichappearedspontaneouslyinBALB/c mice contained also the same number ofMTV DNAcopiesperhaploidgenome as was present inthelivercells(datanotshown).
Theseresults indicate thatmammarytumors
which appear as aresult of infection with the exogenous MTV contain an extra number of MTVDNAsequences.Mammarytumorswhich appearspontaneouslyorbyhormoneinduction 555 VOL. 27,1978
on November 10, 2019 by guest
http://jvi.asm.org/
[image:5.501.105.401.378.571.2]556 MICHALIDES ET AL.
contain the same numberof MTV DNAcopies asispresent in other cells of the mouse.
Hybridization ofMTV
[3H]cDNA
tocyto-plasmic RNA from normal andtumor
tis-sues. The amount of MTV RNA in the
cyto-plasmic RNA of variousmurine tissueswas
de-termined bycomparing the hybridization
kinet-icsof MTV
[3H]cDNA
and MTV RNA with the kinetics of thehybrid formation between MTV [3H]cDNA andcellularRNA.TheCrOtI/2
values,i.e., the
Crot
valueat50% hybridization, of thehybrid formation between MTV[3H]cDNAand cellular RNA were then related to the
CrOti/2
value of the hybrid formation between MTV [3H]cDNA and MTV RNA. The ratio of these
CrotI/2
values indicates the fraction of thecellu-lar RNA being related to MTV RNA (4).The hybridization kinetics between MTV[3H]cDNA
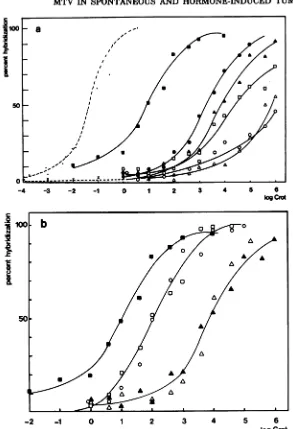
and cellular RNA from C3H tissues are shown inFig. 5Aand B; the corresponding
Croti/2
values are given inTable2. Mammary tumors of C3Hmice contained 11-fold more MTV RNA than
wasfoundinthelactatingmammaryglands;the
spleens contained much less MTV RNA. The
mammary tumors of C3Hf mice, without the exogenousMTV,contained the same amount of
MTV RNAas wasfoundinthelactating
mam-maryglandsofthese mice. These levels of MTV
RNAweremuch lower than those in C3H mice in which the exogenous MTV variant is present.
Hormone-inducedmammarytumorsofC3Hf
or020mice contained less MTV RNAcompared
with the lactating mammary glands of
corre-sponding tumor-free animnals (Fig. 5 and6 and
Table 2). Lactating mammary glands of
hor-mone-treated C3Hffemales contained
approxi-mately60-fold more MTV RNA compared with
nontreated C3Hf females. This clearly showed that hornone treatmentstimulated the expres-sion of MTV inthe mammary glands of C3Hf
females. Thesame wasfound in mice of the 020
strain.Untreated 020 mice did not contain
de-tectableamounts of MTV RNA intheir lactating
mammaryglands,whereas the mammary glands
ofhormone-treated 020 mice contained a very
low, but still detectable amount ofMTV RNA (Fig. 6). Mammary glands of 6-month-old 020
females, which had been infected with
exoge-nousMTVbybeing foster nursed on C3H moth-ers, containedalarge amount of MTV RNA.
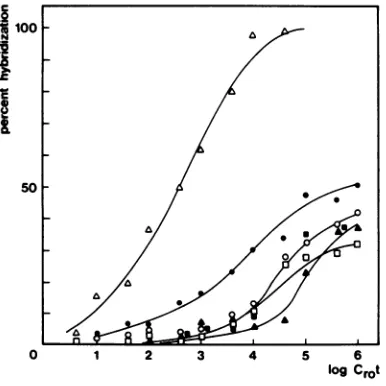
Spontaneous mammary tumors of BALB/c
females containedhardly detectable amounts of MTV RNA, like the other tissues of BALB/c mice, including the lactating mammary glands (Fig. 7), as also has been reported previously (27). When newborn BALB/c mice are infected with MTV (BALB/c), they obtain mammary tumorsatahigh frequency. These virus-induced
mammary tumors contained large amounts of MTV RNAas shown inFig. 7. The spleens of these MTV-infected BALB/c mice also con-tained detectable amounts of MTV RNA.
DISCUSSION
The present results can be summarized as follows.(i)Virus-induced mammarytumors con-tain more MTV DNA copies in their cellular genome thanarepresent in the genome of cells ofnoninfected tissues (Fig.3). The former also containlarger amounts of MTV RNA than are found in thelactatingmammaryglandsofthese animals (Fig. 5). (ii) Hormone-induced mam-marytumorscontain thesamenumber of MTV
DNAcopies in their cellular genomeas do the
othertissues of the animals.(Fig.3and4). The
hormone treatment results in an increased
expressionof MTV RNA inthelactating
mam-mary glands, but this enhanced level of MTV
RNA expression is notmaintained in the
hor-mone-induced mammarytumors (Fig.5and6).
(iii) Spontaneousmammary tumorsofBALB/c mice donotcontain enhancedamountsof MTV DNA or MTV RNA (Fig. 7).
It hasalready been shown in the early thirties (1) that the exogenous MTV causes mammary tumors in susceptiblemice. The mechanism of the transformation ofamammaryglandcellinto amammary tumorcellbythe infection ofthis exogenous MTVstill remainstobesolved, how-ever.Mechanismsmightincludeatransforming
gene product as has been found for type C sarcoma viruses (11, 23), a disturbance of the
cellular regulation system by the insertion of
MTV DNA sequences into host genome, or an escape from the host control system by the
synthesisof new, viralproteins.It is clear from
this and other studies (9, 15, 16, 27) thatthese virus-induced mammary tumors contain en-hancedamountsof MTV DNA and MTV RNA sequences(Fig. 3and5).
Theinvolvement of MTV inhormone-induced
and spontaneous mammary tumors would
re-quire an expression of the endogenous MTV
DNAsequences inthese tissues. We measured
thisbydeterminingthelevels ofMTVDNA and
MTV RNAsequences in mammarytumorsand in other tissues ofthe mice. Increasedamounts of theseMTV DNAandMTVRNAsequences in thehormone-inducedmammarytumors
com-paredwith other tissues of mice would indicate
an involvement of MTV in the formation of these tumors. In neither hormone-inducednor
spontaneously occurring mammary tumors did
we detect enhancedamounts ofMTV RNAor
MTV DNAsequences. This would suggest that these tumors originated without any
on November 10, 2019 by guest
http://jvi.asm.org/
ilo-
a1100 8 <1
50~~~~~~~~~~~~~
-4 -3 -2 -1 0 1 2 3 4 5 6
ogCrOt
l~oo
b
0A~~
A~~~
[image:7.501.104.397.60.489.2]A~~~
FIG. 5. (a)Hybridization ofMTV[3HJcDNAwith viral andmurinecellularRNA.Thehybridizationswere
performedasdescribed in thetext,containing increasingamountsofviralorcellular RNA ataconstant
RNA concentrationwhich wasadjusted bythe additionofyeastRNA. Allhybridizationpointshavebeen
adjustedtothe 93%hybridizationbetweenMTV[3H]cDNAandMTVRNAataC,otvalueof4whichwa-sput
as10l0%, aftersubtractionofthepercentbackgroundhybridizationtoyeastRNA, whichwasbelow 4%. The
hybridizationmixtures contained RNA
firom:
MTV(...),Rauscher leukemia virus(x---x);mammary tumors ofC3H (a), C3Hf+H (a), and C3Hf (A); spleens ofC3H (O), C3Hf+H (O), and C3Hf (A\). (b) Hybridizationsbetween MTV[3H/cDNA
andRNAfrom:mammarytumorsofC3H(M,C3Hf (A),lactating mammaryglands ofC3N(O),C3Hf+H(O),andC3Hf(A).Thecurvesformammaryglands ofC3H(O)andC3Hf+H(O) arethesame,asarethecurvesof C3Hfmarnmaryturnors(A)andC3Hfmammzaryglands(A).
mentof MTV.However, sincewemeasured the amountof MTV expressioninthe tissues of the mouseatthe time of sacrifice of theanimals, it still remains possible that a transient expression of theendogenous MTVis instrumental in the transformation of the mammary gland cells.
Thispossibilityhasalso beensuggestedforthe
involvement of type C RNA tumor viruses in
theoriginof radiation-inducedleukemia in mice
(12). The enhanced amounts of MTV RNA found in thelactatingmammaryglandsof hor-mone-treated020 and C3Hf mice couldaccount
on November 10, 2019 by guest
http://jvi.asm.org/
558 AL.
TABLE 2. Amountsof MTV RNA in various tissues of different mouse strains
Mouse Tissue Ct/2 % MTV
strain value' (x10-2)
C3H Mammary tumor 12 33
Lactating mam- 130 3 marygland
Spleen 21,000 0.019
C3Hf Mammary tumor 9,100 0.048
Lactating mam- 9,100 0.048 marygland
Spleen 690,000 < C3Hf+H' Mammarytumor 2,500 0.16
Lactating mam- 130 3 marygland
Spleen >106 < 020+Hc Mammarytumor >106 <
Lactating mam- 530,000 0.00008 marygland
Spleen >106 < These
C,OtI/2
values of the hybridformation
be-tweencellularRNAandMTV[3H]cDNAweretaken fromFig.5and 6.bThe percentage of MTV RNA is determinedby relating the
Crot1/2
values of the hybrid formation betweencellularRNAand MTV[3H]cDNAtothat of the MTV RNA:MTV [3H]cDNA hybrid formation. <,Undetectableamountof MTV RNA.cTheseanimalshave beentreated with hormones
asdescribed in thetext.
0 1 2 3 4 5 6
log Crot
FIG. 6. Hybridization of MTV C3H]cDNA with RNAsfrom thefollowingtissuesof020 mice: mam-maryglands of6-month-old mice (0), of 12-month-old mice (U), of6-month-old mice treatedwith hor-mones asdescribed in thetext(0), of020fC3Hmice at6 monthsofage(A), spleens of6-month-oldmice (A);andmammarytumorsof020 micetreated with hormones(0).Forhybridization conditions,see leg-endtoFig.5.
100 U
o 1 2 3 4 s 6
logC,ot FIG. 7. Hybridization of MTV
J3H/cDNA
withRNAs ofBALBIc, mammaryglands at 12months
(a), spleens and livers (A\), and
spontaneously
ap-pearingmammarytumors(A);BALBIcmice
infected
with BALBIc MTV, mammary tumors /!), and
spleensand livers(O).Forhybridization conditions,
seelegendtoFig.5.
for this transient MTV
expression.
Itappeared
from thisstudy
thatacontinuousexpression
of this MTVgene(s)
is notrequired
to maintain themammary tumorcells inatransformedstate,because the mammary tumors contained less MTV RNA than the mammary
glands
of the hor0one-treated mice(Fig.
5and6).The number of MTV DNA
copies
inmam-marytumorandliver cell DNA was estimated
by
companng thereassociation kinetics between MTV[
H]cDNA
andDNAsfromthese tissues. Thistypeofanalysi
os(o);notpreclude
thepos-sibility
that themammary tumorDNA hasac-quired
asmallnumber ofoneto twoadditional MTV DNAcopies. Other,
moresensitiveassays, suchasanalysis
ofviral sequencesby
restrictionenzyme
cleavage
and theblotting procedure
ofSouthern
(22),
couldprovide
ultimate answers tothisquestion.
Comparison
of MTVRNAexpeV elS in mammaryglands
between hormeone-treated and unhormeone-treated mice showedaclearstim-ulatory
effect ofprolactin
on the MTV RNAexpression
(Fig.5and6). The effect ofprolactin
instimulating
theMTVRNAecpressioninvivo seemssimilartotheaction ofglucocorticoids
in vitro(19, 21).Inin vitro studiesnoeffectonthe MTVRNAexpression
has been observedby
the addition of prolactin alone (19), butprolactin
increased the MTV RNAexpression
in combi-nation with dexamethasone and insulin (24).quiscombination ofhoerones mimics the situ-ation inhocnone-treated more
sensight
also beJ. VIROL.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:8.501.263.457.74.269.2] [image:8.501.63.256.385.578.2]possible that prolactin only stimulates the
growth of aselect population of cells that are
expressingMTV RNA.
Nodetectable amount of MTV RNA was ob-served in mammary glands of 12-month-old BALB/c mice, although infectious MTV has
been isolated from mammary glands of old
BALB/c mice (13). In our studies we usedpools ofmammary glands from five animals, and it
canthereforenotbe excluded that some cells of
these glands contain complete MTV particles.
ACKNOWLEDGMENTS
Wethank J. vanGrondelle,R.Bakhuysen,and J. Arends fortechnicalassistanceand P.Emmelot for criticalreadingof themanuscript.
LITERATURE CITED
1. Bentvelzen, P. 1972. The biologyof mouse mammary tumor virus.Int. Rev.Exp. Pathol.11:259-297. 2. Bentvelzen, P.,J.H.Daams, Ph. Hageman,J.
Cala-fat, and A. Timmermans.1972.Interactions between viral and genetic factors in the originofmammary tumor in mice. Natl. Cancer Inst. Monogr. 48:1089-1094.
3. Benveniste,R.E., andE. M.Scolnick. 1973.RNA in mamnuaian sarcoma virus transformed nonproducer cells homologoustomurineleukemia virus RNA. Vi-rology51:370-382.
4. Birnstiel,M.L, B. H. Sells, andI. F.Purdom.1972. Kinetic complexity of RNAmolecules. J. Mol. Biol. 63:21-39.
5. Boot, LM., andG.R6pcke.1966.Studieson hypophys-icalisograftsin mice. I.Biologicalaspects. Cancer Res. 26:1492-1496.
6. Boot, LM., G.Rdpcke, andH.G. Kwa.1972.Hornonal factors intheoriginof mammary tumors, p. 275-282. In P.Emmelot andP.Bentvelzen(ed.),RNA viruses and hostgenome inoncogenesis.NorthHolland Publishing Co., Amsterdam.
7. Britten, R.,D. E.Graham,and B. R.Neufeld.1974. Analysis of repeatingDNAsequencesby reassociation. MethodsEnzymol.29E:363-419.
8. Burton,K.1956.Astudyof the conditionsand mecha-nism of the diphenylamine reaction for colorimetric estimation of deoxyribonucleic acid. Biochem. J. 62:315-323.
9.Drohan, W.,R.Kettmann,D.Coicher, and J. Schlom. 1977. Isolation ofthemouse mammary tumor virus sequences nottransmittedasgerminalprovirus in the C3Hand RIIImousestrains.J.Virol.21:986-995. 10.Fine,D.L,J.K.Plowman, S.P.Kelly,L 0.Arthur,
andE.Hellman.1974.Enhancedproductionofmouse
mammary tumor virus in dexamethasone treated 5-iododeoxyuridinestimulatedmammarytumorcell cul-tures.J.Natl.Cancer Inst.52:1881-1886.
11.Frankel,A.E., andP. J.Fischinger.1976.Nucleotide sequences inmouseDNA and RNAspecificfor Molo-ney sarcoma virus. Proc. Natl. Acad. Sci. U.S.A. 73:3705-3709.
12.Haas,M. 1977. Transient virusexpressionduringmurine
leukemia induction by X-irradiation. J. Natl. Cancer Inst.68:251-257.
13. Hageman,Ph., J.Calafat, and J. H. Daams.1972.The mousemammary tumor viruses, p.283-300. In P. Em-melot and P. Bentvelzen(ed.), RNA viruses and host genome inoncogenesis.NorthHollandPublishing Co., Amsterdam.
14. Michalides, R., andJ. Schlom. 1975. Relationshipin nucleicacidsequences between mousemammarytumor virus variants. Proc. Natl. Acad. Sci. U.S.A. 72:4635-4639.
14a.Michalides,R., L van Deemter, R.Nusse, andR. van Nie. 1978.Identificationof theMtv2gene,responsible fortheearlyappearance of mammary tumors in the GR mouse,bynucleicacidhybridization.Proc. Natl. Acad. Sci. U.S.A.75:2368-2372.
15. Michalides, R., G.Vlahakis, andJ.Schlom. 1976.A biochemical approachtothe study of thetransmission of mousemammarytumorvirusesin mousestrains RIII andC3H. Int. J. Cancer 18:105-115.
16. Morris,V.,E.Medeiros, G.M.Ringold,J. M.Bishop, and H.E. Varmus. 1977.Comparison of mouse mam-mary tumor virusspecificDNA ininbred wild and asian mice, andin tumors andnormalorgans frominbred mice.J.Mol.Biol.114:73-91.
17. Muhlbock,0.1965.Note on a new inbred mouse strain GR/A. Eur.J.Cancer1:123-124.
18.Mubibock,O., andL. M. Boot. 1959.Inductionof mam-mary cancer in mice without the mammam-mary tumor agent byisograftsofhypophyses.Cancer Res. 19:402-412. 19.Parks,W.P.,J.C.Ranson, H. A. Young, and E.M.
Scolnick. 1975. Mammarytumor virusinductionby glucocorticoids:characterization of specific regulation. J.Biol. Chem.250:3330-3336.
20.Prensky,W. 1976. TheradioiodinationofRNA andDNA to high specific activities. Methods Cell Biol. 13:121-132.
21. Ringold,G.M.,K R.Yamamoto, G.M.Tomkins,J. M.Bishop, andH.E.Varmus. 1975. Dexamethasone mediated induction of mouse mammary tumor virus RNA:asystemforstudying glucocorticoid action.Cell 6:299-305.
22. Southern,E. 1975. Detection ofspecificsequences among DNA fragmentsseparated by gel electrophoresis. J. Mol. Biol.98:503-517.
23. Stehelin, D.,R.V.Guntaka,H. E.Varmus, andJ. M. Bishop.1976.Purificationof DNAcomplementaryto nucleotide sequencerequired for neoplastic transfor-mationof fibroblastbyavian sarcoma viruses. J.Mol. Biol. 101:349-365.
24. Svec,J., andJ.Links. 1977.Mousemammarytumor virusproductionstimulated by hormones and polya-minesincelisgrown insemi-syntheticin vitro condi-tions. Int. J. Cancer19:249-257.
25.Taylor,J.M.,RF.Illmensee,andL.Summers.1976. Efficient transcription of RNA into DNA by avian sarcoma virus polymerase. Biochim. Biophys. Acta 442:324-330.
26. VanNie,RK,and A.A.Verstraeten.1975.Studiesof genetic transmissionofmammarytumorvirusbyC3Hf mice. Int.J.Cancer16:922-931.
27. Varmus, H. E., N. Quintrell, E. Medeiros, J. M. Bishop,R C.Nowinski, and N. H.Sarkar. 1973. Transcriptionofmousemammary tumorvirusgenes in tissues from high and low tumor incidence mouse strains. J. Mol. Biol. 79:663-679.
on November 10, 2019 by guest
http://jvi.asm.org/
![FIG. 1.MTVRNaseRNAhybridsresistantscribedcontainedlabeledanding Hybridization between '25I-labeled MTV and MTV [3H]cDNA](https://thumb-us.123doks.com/thumbv2/123dok_us/1525880.105164/4.501.266.456.458.571/fig-mtvrnasernahybridsresistantscribedcontainedlabeledanding-hybridization-i-labeled-mtv-mtv-cdna.webp)
![FIG. Cotperformedhormones 4. Hybridization of MTV rH]cDNA with DNA of mammary tumors of 020 mice treated with (0), of livers and spleens of these 020 mice (0), and of calf thymus (O)](https://thumb-us.123doks.com/thumbv2/123dok_us/1525880.105164/5.501.65.416.57.299/cotperformedhormones-hybridization-mammary-tumors-treated-livers-spleens-thymus.webp)