Copyright X 1991, American Society for Microbiology
Human
Immunodeficiency Virus (HIV)-Infected
Tumor
Xenografts
as an
In
Vivo Model for Antiviral Therapy: Role of Alpha/Beta
Interferon in Restriction of
Tumor
Growth
in Nude
Mice Injected
with
HIV-Infected
U937 Tumor
Cells
PATRIZIAPUDDU,1 CHIARA LOCARDI,1 PAOLA SESTILI,1FRANCO
VARANO,1
CARLO PETRINI,1 ANDREA MODESTI,2 LAURA MASUELLI,2 ION GRESSER,3 ANDFILIPPOBELARDELLIl*Laboratory of Virology, Istituto Superiore diSanita, VialeReginaElena299, 00161 Rome, Italy';Departmentof Experimental Pathology, University ofRome,00161 Rome, Italy2; andInstitutdeRecherches Scientifiques surleCancer,
94802 Villejuif Cedex, Paris, France3 Received5 November1990/Accepted24January 1991
The host factors involvedin therestrictionoftumorgrowthwerestudiedin nudemicetransplanted witha
cloned line of chronically human immunodeficiency virus (HIV)-infected U937 cells. HIV-infected and uninfected U937 cellsexhibitedthe samegrowth patternsinculture. However, HIV-infected cells werenot tumorigenicwheninjected subcutaneously in nude mice, whereaslargesolidtumors wereobserved in mice
injected withuninfected U937cells. Injectionof nudemicewithantibody toalpha/betainterferon(IFN-a>)
enabled HIV-infected U937cells togrow progressively in approximately 90to 100% ofmice. HIV-infected U937 cells formed solid tumors in the majority (60 to 90%) ofeither immunosuppressed (splenectomized, irradiated,and
anti-asialo-GM,-treated)
orgeneticallyimmunodeficient(bg/nu/xid)nude mice. InmicetreatedwithantibodiestoIFN-alwithestablishedHIV-positive tumors,adirect correlationwasfoundbetween p24
antigenemiaand tumorsize. Treatmentofestablished HIV-positive U937 cell tumorswith human IFN-a or
mouseIFN-a$/ resultedinaclear-cut inhibition ofbothtumorgrowth and p24HIVantigenemia. Incontrast,
treatmentwith tumornecrosisfactor alpha markedly inhibitedtumorgrowth but didnotsignificantlydecrease
serum p24levels. 3'-Azido-3'-deoxythymidine treatment didnot affect either tumorgrowth orthe levelsof
serump24 antigen. Thesedataindicate thatendogenous IFN-oc$ isacrucialfactor in the restriction of both
tumorgrowth and p24 antigenemia in mice injected with HIV-infected tumor cells. Moreover, the results suggestthat thedevelopment of HIV-1 p24 antigenemiainathymicimmunosuppressedmicemayrepresentan
interestinginvivomodelforanti-HIVtherapy.
Althoughconsiderableprogress has been achievedinour
knowledge of the epidemiology of AIDS and its etiologic agent(humanimmunodeficiency virus[HIVI), little is known about the pathogenesis of infection. In particular, it is important to establish suitable in vivo model systems not onlytostudy someaspectsof thepathogenesisof AIDS but also to evaluate prophylactic measures, such as vaccines,
andtodefine effective antiviral therapies.
Genetically immunodeficient (SCID) mice transplanted withhematolymphoid cells from humanfetalliver, thymus, and lymph node (22, 25), adult human peripheral blood leukocytes(24),orhumanbonemarrowcontaining hemopoi-etic stemcells (17) have beenproposed as possible animal models for AIDS. The suppression of HIV infection in
3'-azido-3'-deoxythymidine (AZT)-treated SCID mice
en-grafted with human hematolymphoid organs has recently
been reported (23), suggesting that such models can be of someinterest instudyingnewantiviralcompounds.Inthese experimental systems, HIV expression is apparently
re-stricted to the virus-permissive transplanted human cells, and neitheramplification of HIV expressionnor viral anti-genemia is observed.
In this study, we have investigated some host factors
involved in the restriction oftumor growth in nude mice transplanted withacloned line ofchronicallyHIV-infected
U937cells, andwehave defined theexperimentalconditions
* Correspondingauthor.
for obtaining p24 antigenemia in mice with established HIV-positivetumors. Thedatapresented demonstratethat injection of antibodies to mouse alpha/beta interferon
(mu-IFN-olp)in nudemicetransplantedwith HIV-infected U937 cells results in both tumor growth and p24 antigenemia. Moreover,these dataindicate thatnudeimmunosuppressed mice treated with anti-IFN-a/1 antibodies and transplanted withHIV-infected U937 cells mayrepresentauseful model
forstudyingthe action of anti-HIV substances in vivo. MATERIALS AND METHODS
Cells and culture conditions. The human histiocytic lym-phoma cell line U937(31) (kindly provided by A. Siccardi, Milan, Italy) was cultured in RPMI 1640 medium supple-mented with 10% heat-inactivated fetal calf serum. Cells
wereseededat2x 105/ml in fresh medium and reachedacell densityofapproximately2 x 106/ml 3to4days thereafter. Primary infection of U937 cells. A total of 3 x 106 U937 cellswere treated with Polybrene (2 ,ugIml)for 1 h at 37°C. Cellswerecentrifuged, and the cellpelletwasinfected with 50 jil of HIV (HLTVIII B strain), correspondingto 15,000
cpmofreversetranscriptase activity. Afterincubation for 1
hat37°C,the cellswereresuspendedataconcentrationof 5
x 105/ml in RPMI 1640 medium containing 20% fetal calf
serum. HIV-infected U937 cellswereclonedbythe dilution
methodapproximately80daysafter HIV infection. All of the 15 clones tested released HIV in the cell medium, as
determined by p24 assay, even though some differences
2245
on November 10, 2019 by guest
http://jvi.asm.org/
were observed in the amounts of released virus (data not shown). These data indicated that virtually all U937 cells were HIV infected. In the experiments to be described, we used a cloned HIV-infected cell line (clone 10) capable of releasing greater amounts of HIV than can the uncloned HIV-infected cell population.
HIV-infected U937 cells exhibited a markedly lower
expression ofCD4, HLA-DR, and HLA-DP antigens than did uninfected U937 cells. No significant differences were observed in the expression of a panel of other membrane
antigens (OKM1, TecMG2, TecMG3, LeuMl, LeuM3,
OKM14, OKM13, OKM5, and HLA-DQ).
Mice. Male or female Swiss nude mice were purchased
fromCharles River Laboratories (Calco, Italy) at 4 weeks of ageand kept for 1 week before use. bg/nu/xid mice (17) were
obtained from Harlan Sprague-Dawley (Indianapolis,
Ind.).
Splenectomy and irradiation. The splenectomized,
irradi-ated, and anti-asialo-GM1-treatednude mice were prepared as described by Caretto et al. (4). Briefly, splenectomy was
performed via a 5-mm-long incision on the left upper flank. The mice received a sublethal total-body irradiation of 450 radsfrom a137Cs source providing a dose rate of 50 rads/min and were referred to as SI nude mice. Twenty-four hours after splenectomy and irradiation, a few mice were also
injected intravenously (i.v.) with 0.2 ml of
anti-asialo-GM1
antiserum (Wako Chemicals GmbH, Dusseldorf, Federal
Republic ofGermany) (SIA nude mice) (14). This treatment eliminates all natural killer cell activity (7, 11).
Reagents.Polyclonalsheep
anti-mu-IFN-odp
(sheep 1) wasprepared andpurified (13) and had a neutralizing titer of 6.4 x 106 against 4 to 8 U of
mu-IFN-ao/r
(13).Partially purified
mu-IFN-a/p
(107 U/mg of protein) wasprepared asdescribed elsewhere (32). In some experiments, we used as control material a protein preparation derived from the culture supernatant of C243 mouse cells; this controlpreparationwassubjected to the same procedures as wereused toprepare the partially purified
mu-IFN-ot/&
(32). Human natural IFN-ot(hu-IFN-a; 5 x 106 U/mg of protein) was kindly provided by K. Cantell (Helsinki, Finland).Recombinant human tumor necrosis factor alpha (hu-TNF-ot; specificactivity, 7 x 107 U/mg) was a generous gift of W. Fiers and J. Tavernier (Ghent, Belgium). Cytokines wereinjected peritumorally (0.2 ml per injection).
AZT waspurchased from The Wellcome Research Labo-ratories (Beckenham, Kent, England) through the courtesy of M. Martorelli (Wellcome Italia SpA, Pomezia, Italy). Mice were treated with AZT either by oral administration (1
mg/ml
in the drinking water) as described elsewhere (23) orby peritumoral injection (480/mg/kg/day [maximal tolerated dose]).
Antigen capture assay. Sera were tested for HIV p24
antigen by an antigen capture enzyme-linked immunosor-bent assay (Cellular Products Inc., Buffalo, N.Y.).
AssayofIFN inserum. IFN was assayed by inhibition of thecytopathic effect ofvesicular stomatitis virus on L cells in monolayer cultures (0.2 ml per well) in Falcon
mi-croplates. Units are expressed in our laboratory units; 1 of our units is the equivalent of 4 mouse research reference units.
Evaluation oftumor growthin nude mice transplanted s.c. with U937 cells. Nude mice were injected subcutaneously
(s.c.)in the shoulder with noninfected or HIV-infected U937 cells in 0.2 ml of saline. The diameters of tumor nodules were measured with calipers, and the mean surface area of the tumor perexperimentalgroup was determined.
Electron microscopy. The tissue fragments were fixed in
A
2U
I1-3
ouninfected a HIV-
infected
in vitro
1 TI
1 2 3 4 5
days
B
I
4
2-ouninfected
* HIV- infected
invivo
10 20 30 40 50
days
FIG. 1. Growth of uninfected and HIV-infected U937 cells in culture and transplanted s.c. in nude mice. (A)Cells wereseededat 2x 105/mlin RPMI medium supplemented with10% fetal calfserum as described in Materials and Methods. At different days aftercell seeding, cells were counted and their viability was determined by the trypan blue dye exclusion method. Values represent the means of duplicate samples. The variability of replicate samples was always less than 10%. Cell viability was more than 90%. (B) Six-week-old male Swiss nude mice(eight per group) wereinjected s.c. with107 uninfected or HIV-infected U937 cells. Tumor growth was evaluated as described in Materials and Methods.
cacodylate-buffered 2.5% glutaraldehyde, postfixed in os-mium tetroxide, dehydrated in graded alcohols, and then embedded in Epon 812.
The cells were collectedby gentle centrifugation, and the pellet was resuspended in 2.5% glutaraldehyde in phosphate buffer (0.1 M) at pH 7.2 at room temperature and placed at
4°C
for 12 to 16 h. The cell suspension was pelleted at low speed, washed in the same buffer,postfixed
with1%
OS04,
rapidly dehydrated in a series of alcohols, and embedded in Epon 812.Ultrathin sections were stained with uranyl acetate and lead hydroxide and observed under a Philips
CM10
electron microscope.RESULTS
Growth properties of uninfected and HIV-infected U937 cells in culture and when
injected
s.c. in nude mice. Cloned HIV-infected U937 cells (clone 10) exhibited in vitro growth curves very similar to those of uninfected control cells (Fig.1A).
These chronically infected cloned U937 cells released higher levels of HIV in the culture medium than did the parental uncloned HIV-infected cell population, as deter-mined by reverse transcriptase and p24 assays of the culture supernatants (data not shown). Large solid tumors devel-oped in mice transplanted with control uninfected U937cellsL
on November 10, 2019 by guest
http://jvi.asm.org/
[image:2.612.364.523.73.330.2](Fig. 1B). In contrast,s.c.tumorswerenotobserved innude mice after injection of107HIV-infectedU937cells (Fig. 1B). Some initialtumorgrowthwasobserved insomemice (10to
30%) injected s.c. with1.5 x 107 2x 107HIV-infected U937 cells, but these tumors generally regressed 3to4weeks after tumor cell injection (data not shown).
Effects of
injection
ofantibodies tomu-WN-a/j3 on tumorgrowth in nude mice transplanted s.c. with uninfected or
HIV-infected U937 cells. It was of interest to determine whether the injection of antibodies to mu-IFN-a/o would result in any change oftumorgrowth in mice transplanted s.c. with uninfected and HIV-infected U937 cells. The injection ofantibodies to IFN-a/I in mice transplanted s.c.
with uninfected U937 cells resulted in significantly greater
tumor growththanin mice treatedwith control sheepgamma
globulin (Fig. 2A). Treatment with anti-IFN antibody of mice transplanted with HIV-infected U937 cells resulted in the developmentof rapidly growing solidtumors in allmice (Fig. 2B and C). Although small tumors were initially
de-tected afterinjection of large numbers ofHIV-infectedU937 cells (1.5 x 107) in some control treated mice, these s.c.
tumors regressedat alaterstagein this experiment(Fig. 2B). Development ofHIV p24 antigenemia in
anti-IFN-aIj8-treated nude mice transplanted s.c. with HIV-infected U937 cells and correlation withtumorgrowth. All ofthe anti-IFN-treated mice with established U937 HIV-positive tumors
exhibited detectable serumlevels of p24on day 40(Fig. 3).
p24 levelsranged between 800 and 3,600 pg/ml. Incontrast,
only 2 of10 mice treated with normal sheepgammaglobulin
and transplanted with HIV-infected cells developed solid tumors, and none of them exhibited detectable p24 antigen-emia. Evenwhen solid tumors did develop in mice treated with normalsheepgammaglobulin,nodirect correlationwas
observedbetweentumorgrowth and serump24levels (Fig.
4A).
Figure 4B shows the kinetics of the direct correlation betweentumor growth and p24 antigenemia inan
anti-IFN-treated mouse transplanted with HIV-infected U937 cells. These data suggested that endogenous IFN-a/, was
in-volvednot only intherestrictionoftumorgrowthbut also in thein vivo inhibition ofHIV expression.
Growth of HIV-infectedU937 cells inimmunosuppressedor
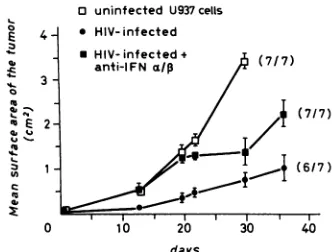
genetically immunodeficient nudemice. Itseemed of interest to determine the growth of HIV-infected U937 cells in immunosuppressed or genetically immunodeficient nude mice. As shown in Table 1, s.c. injection ofbglnulxid mice withHIV-infected U937cells resulted inthedevelopmentof s.c. tumors in 60% ofthe injected mice. Likewise, HIV-infectedU937 cellsformedsolid tumorsin90% of both SIA nudemice andanti-IFN-treated mice; 60%of the SIA nude mice transplanted with HIV-infected U937 cells exhibited detectable serumlevels of p24 antigen. In this experiment, treatment of SIA nude mice with antibodies to IFN-ac, resultedin a very rapid development of solidtumorsin100% of the mice injected with HIV-infected cells, and all mice exhibited a p24 antigenemia (Table 1). Figure 5 shows the kineticsof tumorgrowthin SIAnude micetransplanteds.c.
with controlandHIV-infected U937 cells. Therateoftumor
growthwaslower for HIV-infected cells than for uninfected U937 cells. Treatment of SIA nude mice with antibody to
IFN-aJp
resulted inahigherrateof growth ofHIV-infected U937 tumors than in mice not treated with antibody, al-thoughthe growthof thesetumorswasstill less rapid than inmice injected with uninfected U937cells (Fig. 5).
Electron microscopy studies ofHIV-positive U937 cell
tu-mors. Figure 6A shows the morphology of HIV-infected
A
E
1. a
I0 o
IE
a c0-,
o normal serum
*anti-IFNri/u
10 20 30 40 50 60
days
B
a
120- 100-S
80-% 60-u
I.. 40-a V"
20-U
mrmalserum
*anti-IFNaip
(00/10)
(2/10)
\ ~r ~~~ I
10 20 30 40 50
days
c
E
a
c to
S.. U)
to
q
2-0
O no
* an
irmal serum I (10/10)
iti-IFNax/
(2/10)
10 20 30 40 50 60
days
FIG. 2. Effect of antibodies to
IFN-cx/p
on tumorgrowthinnude mice transplanted s.c. with uninfected orHIV-infected U937 cells. Five- to six-week-old female Swissnude mice (10 pergroup) wereinjected s.c. with107 uninfected U937 cells (A) or 1.5 x107 HIV-infected U937 cells (B and C). Each mouse was treatedwith 0.2 ml of a 1:20 dilution of either antibody to
IFN-a/P
(320,000neutralizing
units) or normal sheep gammaglobulin asfollows:
Timeoftreatment
3hbefore tumorcellinjection
time0,withtumorcells Day7
Day14
Day21
Day28
Route i.v.
S.C.
s.c. (peritumorally)
s.c. (peritumorally) s.c. (peritumorally)
s.c.(peritumorally)
ro
on November 10, 2019 by guest
http://jvi.asm.org/
[image:3.612.341.529.90.558.2]80- so
,
60 604,o40 40 ~.
20- 20
0 0
Control Anti-IFN
serum ct/p
FIG. 3. Correlation between tumor growth and HIV p24 antigen-emia in anti-IFN-treated nude mice transplanted s.c. with HIV-infectedU937cells. Five-week-old male nude mice (10 per group) were treated with either antibodies to
IFN-oi/,
or control sheep gammaglobulin as described in the legend to Fig. 2. Mice were injecteds.c. with 1.5x107HIV-infected U937 cells. The percentage ofmice with tumor was assessed 40 days after U937 cell injection. Sera were taken on day 40, and the levels of p24 antigen were determined as described in Materials and Methods.U937 cells seen by electron microscopy after 3 days of
culture. Some HIVparticleswere found to be released from the cell membrane of in vitro-passaged U937 cells. Figures 6B and C illustrate some examples of the electron micros-copy analysis of an HIV-positive U937 cell tumor derived
from an anti-IFN-a/13-treated mouse. Extensive HIV
pro-ductionwith a large number of virus particles budding from
the U937 cell membrane was observed (Fig. 6C). Large
numbers ofHIVparticleswere detected in the intercellular spaces(Fig. 6B)and in close association with blood vessels
(datanotshown).
Effects of hu-IFN-aL,mu-IFN-aod,hu-TNF-a, and AZT on tumor growth and p24 antigenemia in immunosuppressed mice with established HIV-infected U937 cell tumors. The results presented above indicated that immunosuppressed nudemice injectedwith HIV-infected U937 cells and treated
with antibodies to IFN-a/, might represent a useful model
forthedevelopment ofHIV-1p24 antigenemia and therefore
A
E
Ch Q0
[image:4.612.93.275.75.190.2]'-Q.
TABLE 1. Subcutaneous growthofHIV-infected U937cellsin immunosuppressed(SIA) orgeneticallyimmunodeficient
(bg/nu/xid) nude micea
No. ofmice withtumorsc/total No. of mice
Anti-IFN- with
detect-Mice
e.Xsa/pb
Uninfected HIV-infected ableserum U937cells U937 cells p24levelsdNude - 10/10 0/10 0
bglnulxid - 8/8 6/10 NDe
Nude + 10/10 9/10 9
SIA nudef - 8/8 9/10 6
SIAnude + ND 10/10 10
aFive-tosix-week-oldmale mice wereinjecteds.c.with107uninfectedor
HIV-infectedU937cells.
bTreatments withanti-IFN antibodieswereperformedasdescribed in the legendtoFig.2
cCalculated35 to 40days after U937 cellinjection.
dSerafrom10miceinjected with HIV-infectedU937cellswereharvested
onday 28 and tested for p24 levelsasdescribed in Materialsand Methods. Detectable p24serumlevels were those >100pg/mnl.
IND, Notdetermined.
fForpreparation of SIA mice,seeMaterials and Methods. The micewere
injecteds.c.withU937cells1day after i.v.injection ofanti-asialo-GMI.
for studies to determine the relative value of anti-HIV therapies.
We investigated the effects of hu-IFN-a, mu-IFN-ao/,
hu-TNF-a, and AZT on the p24 antigenemia in mice with established HIV-infected U937 cell tumors. Preliminary in vitro studies were performed to determine the possible effectsofthesesubstances on HIVproduction inchronically infectedU937cells under cultureconditions. Figure7shows
theeffects of hu-IFN-a, mu-IFN-a/,, hu-TNF-a, and AZT on HIV production in chronically infected U937 cells in
culture, as determined by measuring the p24 levels in the culture supernatants. Hu-IFN-a exhibiteda marked
inhibi-tory effect on HIV yield, whereas hu-TNF-a significantly increased HIV production. Neither AZT nor
mu-IFN-ac/p
affected HIV production in vitro (Fig. 7). Hu-IFN-a and TNF-a exhibited someinhibitoryeffect on cellmultiplication (i.e.,a 20to30% inhibitionofcellgrowth in the presence of
B
U E Cm
0
t-4, :E
a
S.
U)
0 10 20 30 40 50
days
6E
I-C 0. a
E
z
4'
I-a I0
IU)
days
FIG. 4. Analysis oftherelationship between tumor growth and HIV p24 antigenemia in nude mice transplanted s.c. with HIV-infected U937 cells andtreated withnormal sheep serum (A) or antibodies toIFN-a/,(B). Five-week-old male nude micewere treated with either control sheep gammaglobulin or antibodies to
IFN-a/p
as described in the legend to Fig. 2. Mice were injected s.c. with 1.5 x 107 HIV-infectedU937 cells. At different times after tumor cell injection, orbital venous blood was taken from tagged individual mice and serum samplesweretested for the p24 antigen as described in Materials and Methods. Values represent the surface area of each tumor and the serum p24antigenlevels ofonerepresentative tumor-bearingmousetreated as indicated above.Inn-.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:4.612.321.559.106.198.2] [image:4.612.138.490.527.668.2]E44
I,
11 3 3-L.
E 2
a_i
Co
- 1
c
O uninfected U937 cells
* HIV-infected
*HIV-infected+
anti-IFN a/p7
//
i (7/7).., (6/7)
..
..
.~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
0 10 20 30 40
days
FIG. 5. Subcutaneous growth of uninfected and HIV-infected U937 cells in SIA nude mice and effect of antibodies toIFN-a/,.
Five-week-old male nude mice (seven pergroup) were
splenecto-mized, irradiated,andtreated with
anti-asialo-GM,
antibodies(4)asdescribed in Materials and Methods. At 24 h after i.v. injection of
anti-asialo-GM,,
themicewereinjecteds.c. with 107uninfectedorHIV-infected U937 cells. Onegroupof micewasalsotreated with
antibodiestoIFN-a/, asdescribed in the legendtoFig. 2. Numbers in parentheses indicate the numberof mice with tumor/totalnumber of mice injected (determined40days aftertumorcellinjection).
100 to 500 U/ml), whereas mu-IFN-a/, and AZT were
ineffective (datanotshown).
We then performeda series ofin vivo experiments with
these substances. SI nudemice weretreatedwith
anti-IFN-al
antibody and subsequently injected s.c. withHIV-infected U937 cells. On day 14, allof themice haddeveloped solid tumors and were treated with hu-IFN-a or AZT or
were leftuntreated. Treatmentwith hu-IFN-axresulted in a
marked reduction oftumorgrowth and even tumor
regres-sion in comparison with controlmice (Fig. 8A). Incontrast,
AZTtreatment (1 mg/ml in thedrinkingwaterfor 3 weeks) did not inhibit tumorgrowth. On day 38, mice were
sacri-ficed, solidtumorswere removed,and serumsamples were
taken for p24 titration. None of the mice treated with hu-IFN-a exhibited detectable serum p24 levels (<100 pg/
ml), whereas three ofthefourcontrol mice and three of the four AZT-treated mice exhibited serum p24 levels ranging
between 400 and 1,350 pg/ml. One control and one
AZT-treated mouse exhibited very small tumors (mean surface
area, <150 mm2) and had no detectable serum p24 levels
(<100pg/ml). Inasubsequentexperiment, AZTwas
admin-istered peritumorally eachday for 2weeks (480 mg/kg/day [maximal tolerated dose]). No significant change in tumor growth or in p24antigenemia was observed inAZT-treated
miceincomparison with controlmice (datanotshown). Inanotherexperiment, establishedHIV-positive U937cell
tumors were treated with mu-IFN-a/,, hu-TNF-a, or a
control preparation. Both mu-IFN-a/, and hu-TNF-a stronglyinhibited tumorgrowth (Fig. 8B). Allof the TNF-treated tumors exhibited detectable levels ofp24
antigene-mia, whereas none of the mice treated with mu-IFN-a/,B
exhibiteddetectablelevels ofserump2438daysaftertumor
cellinjection. Thesedatasuggestedthat p24antigenemia did
notsimplyreflecttumorgrowth and thatcytokines such as IFN and TNFmay exhibitopposite effects onHIV produc-tion inthis invivomodel.
The electron microscopy analysis of HIV-infected U937
tumors 7 and 18 days after the initiation of therapy (two
tumors per group) revealed a clear-cut reduction of HIV
particles intumorstreatedwitheitherhu-IFN-axor
mu-IFN-a/,1. Areasoftumornecrosis weredetectedinHIV-infected
U937 tumors treated with
hu-TNF-a,
hu-IFN-a,
andmu-IFN-a/,B intheabsence of obvious host cell infiltrates
(data
notshown).
DISCUSSION
The results ofthese
experiments
demonstrate that injec-tion of antibodiestomu-IFN-a/1
innude micetransplanted
s.c.with HIV-infected U937 cells resultsinthe
development
of HIV-positive U937 cell tumors and
p24
antigenemia.
Usually, neither tumor
growth
norp24
antigenemia
was observed in control micetransplanted
with HIV-infected U937cells.Inanti-IFN-a/W-treated
mice,
adirectcorrelationwas observedbetweentumor
growth
and HIVp24
antigen-emia,
indicating
thatendogenous
IFN-&x/I
represents animportantfactorin therestriction of bothtumor
growth
and HIVproductionin this system.Although
HIV-infectedU937 cellsdidgrowto someextentinimmunosuppressed
(SIA)
or genetically immunodeficient(bg/nu/xid)
nudemice,
treat-mentwith antibodiestoIFN-a/1
seemed essentialtoobtain progressive tumor growth and HIVproduction.
TreatmentofSIA nudemice withantibodiestoIFNresults in the
rapid
development(latency
time,
8 to 10days)
ofHIV-positive
U937 cell tumors and in
p24
antigenemia
in 100% of thetumor-injected mice.
We have previously shown that
injection
ofantibodies toIFN-a/p
markedly increased tumorgrowth
in micetrans-planted with different
syngeneic
tumors(9)
andabrogated
the resistance to the
multiplication
ofFriend erythroleuke-mia cells in the livers ofallogeneic
mice(10,
12).
In nudemice, it had been
previously
shownthatinjection
of antibod-iestomu-IFN-a/,3
markedly
increasedtumorgrowth
inmice transplanted with tumor cellspersistently
infected with different viruses (28,29)
as well as with humanprostatic
tumors(28). Itis alsoof interestthattreatmentof mice with
antibody
toIFN-oa/,
abrogated
theresistance of X-irradiatedF1hybrid and
allogeneic
mice to bonemarrowgrafts (1).
Inview oftheinterest in in vivo murine models forAIDS,
it is reasonable to suggest that
HIV-positive
U937 cell tumorsimplanted
inanti-IFN-a/,-treated
nude mice withestablished p24
antigenemia
may represent a useful modelforthedefinition ofnew
protocols
for antiviraltherapies.
Inparticular,
this system allowed usto compare the effects of hu-IFN-aandAZT(Fig. 8A).
Hu-IFN-awashighly
effective ininhibiting
both tumorgrowth
andp24
antigenemia,
whereas AZT treatment did notaffect the
growth
of estab-lishedHIV-positive
U937 cell tumors(Fig. 8A)
and did notinhibitp24
antigenemia.
Hu-IFN-aprobably
actsdirectly
on establishedHIV-positive
tumors, ashu-IFN-a
showsavery lowcross-reactivity
on mouse cells(2).
In this context, it isof interest that addition of
hu-IFN-a
to thesechronically
infectedU937 cells in culture resulted inamarkedinhibition of HIV
production,
whereas AZT did not affect HIVpro-duction
(Fig. 7).
These data are in agreement with thosereported by Poli and co-workers
using
otherchronically
HIV-infected cell lines(27).
Although HIV-infected U937 cells in culture were resis-tant to the
antiproliferative (data
notshown)
and antiviral (Fig. 7)effectsofmu-IFN-a/,, injection
ofmu-IFN-a/p
washighly effective in
inhibiting
both tumorgrowth
andp24
antigenemia. Thisfinding
suggests that host antitumor and antiviral mechanisms were also mostlikely
activatedby
mu-IFN-a/,B inimmunosuppressed
nude mice.Although
nudemicehavebeen
widely
utilized for in vivoinvestigation
ofmany human tumors, there isample
evidence thatthey
display
residualimmunity
against
xenotransplants
evenafteron November 10, 2019 by guest
http://jvi.asm.org/
[image:5.612.94.262.78.204.2]A
2>A-,.._. i
v
lo *k I
a~~~~~~~~~~~
S~~~~sl
'
4
l~~~ta
r1
,
.
FIG. 6. Electron microscopy of HIV-infected U937 cells in vitro and when transplanted s.c. in anti-IFN-a/p-treated nude mice. (A)
Morphology of HIV-infected U937 cells in culture. Cellswere harvested 3 days after cellseeding and processed for electron microscopy
analysis asdescribed inMaterials and Methods. Some HIV particlesareobserved in association with the cell membrane and foundtobe
released inthe extracellularspace. Magnification, x8,820. (B) HIV-infected U937 celltumorestablished inan
anti-IFN-a/P-treated
mouse.Thetumorwasdissected 40 days aftertumorcell injection and processed for electronmicroscopy analysis asdescribed in Materials and Methods.Invivotreatmentswereperformedasdescribed in thelegendtoFig.2.Note the cluster of virusparticles in the extracellularspace. Magnification, x14,400. (C) Typicalpatternof extensive virusproduction inanHIV-infected U937 celltumordissected froman
anti-IFN-a/a-treatedmouse.Notethe differentstagesof virus formation in theU937 cell plasma membrane. Budding with crescent-shaped nucleoid surroundedbyanenvelopewithfuzzysurfaceprojectionsandimmaturevirus withring-shapednucleoidprofilesandmatureforms sectioned
longitudinally andperpendiculartothecoreaxis canalso beseen. Magnification, x50,000. 2250
rf ,111
on November 10, 2019 by guest
http://jvi.asm.org/
[image:6.612.75.547.67.635.2]0
0
-10
[image:7.612.69.293.72.265.2]hu-IFNa mu-IFNa/13 AZT TNFa
FIG. 7. Effects ofhu-IFN-a,
mu-IFN-a/p,
hu-TNF-a,and AZT on HIV production in chronically infected U937 cells in culture. HIV-infectedU937cellswereseededat2 x 105/ml inthe presence orabsence of different substances. On day 3 of culture, cells were diluted 1:2 with fresh medium with or without IFN or AZT to maintainthe initial concentration. Supernatants werecollected on day6of culture and testedforp24. Treatments wereperformedas follows: hu-IFN-a, 100U/ml; mu-IFN-a/1, 100U/ml; andAZT, 1F±M. Values represent the means of duplicate samples. Standard deviations did not exceed10%.
immunosuppression (reference 30 and references herein). In fact, despite their nominal athymic state, nude mice have
some residual Tcells and exhibit immune reactions (16, 18, 19, 33). The numbers of B cells, natural killer cells, and macrophages are similar to or even higher than those in
euthymicmice of thesamestrain(5, 15). Recovery of natural
killer cell and B-cell activity has been described after the initial suppression induced either by whole-bodyirradiation
orby injectionof anti-asialo-GM1 antibody. Likewise,
mac-rophage functions appear to be unaltered in SIA or
geneti-cally immunodeficient nude mice. In this regard, Balkwill
and Proiettipreviously reportedthatmu-IFN-a/ exerteda
potentantitumoractivityin nudeandnude/beigemice trans-planted with human tumors (3). The fact that SI nude mice with establishedHIV-positive U937 cell tumors respond to
mu-IFN-o/ treatmentby reducedtumorgrowthand inhib-itedp24 antigenemia is inaccord with these findings.
The question still to be answered concerns the antiviral
action ofmu-IFN-a/,BonHIV-infected humanU937tumors.
Mu-IFN-a/, exertednoeffectonthese cells inculture(Fig. 7), and there is no evidence to suggestthat the important species specificity of mu-IFN-a/3 isnotconserved in vivo. Thus, it seems unlikely to us that mu-IFN-oa/, directly inhibitedHIV replicationin these humantumors. The anti-viral effectof mu-IFN-a/3 maybeindirect and secondaryto theIFN-inducedreductionintumorgrowth.Thefindingthat TNFinhibitedtumorgrowthand didnotinhibitp24 antigen-emia would seem to argue against this hypothesis, but the conditionsmaynotbesimilar,since TNFalsoenhances HIV multiplicationin thesecells(Fig. 7; 6, 20, 26). Lastly,itmay
be thatIFN-a/, induces theproduction bymousetissuesof
another cytokine that is not species specific and exerts
anti-HIV activity.
Afew mouse models for AIDS have beenproposed (17, 22-25), and the advantages and disadvantages of these
Days
FIG. 8. Effects ofcytokines (hu-IFN-a, mu-IFN-a/,, and
hu-TNF-a) andAZTon tumorgrowthinnude mice withestablished HIV-positive U937 cell tumors. Five-week-old female nude mice weresplenectomized and irradiated (SI nude mice)as describedin
Materials andMethods.Micewereinjectedi.v.with 0.2 mlofa1:20 dilution (containing 320,000 neutralizing units)ofanti-IFN-a/p
an-tibody.Three hourslater, the micewereinjecteds.c.with1.5x 107
HIV-infected U937cellsin 0.2ml of salinecontaininga1:20dilution ofanti-IFN-a/, antibody. The mice were further treateds.c. with
thesameantibodydilutiononday5. (A)Onday 14, themicewere
divided into three groups and treated with AZT (1 mg/ml in the
drinkingwaterfor3weeks)orhu-IFN-a(105U permousein 0.2 ml
s.c.; five daily injections per week; 3 weeks oftreatment) or left
untreated(NT). Onday 38,the mice weresacrificed, solidtumors
wereremoved, andserum samples were tested forp24 antigenas
described in Materials and Methods. Theserump24levelswereas
follows:
Treatment
None hu-IFN-a AZT
Serump24(pg/ml), 4mice/group 1,350, 1,300,550, <100 <100, <100, <100, <100
500, 400,600, <100
(B)On day21 oftumorgrowth, the mice were divided into three
groupsand treateds.c.with 0.2ml of controlpreparation (five daily injections perweek; 2 weeks oftreatment), hu-TNF-a (2 ,ug per mouse ondays21 and 22),ormu-IFN-a/,B (105Uper mouse; five
daily injections perweek; 2 weeks oftreatment). On day 38, the
mice were sacrificed, solid tumors were removed, and serum
sampleswere tested forp24 antigenas describedin Materialsand
Methods. Theserump24levelswere asfollows: Treatment
Controlpreparation mu-IFN-ap
hu-TNF-a
Serump24(pg/ml), 4mice/group 2,800, 1,400, 1,000,100 <100, <100, <100, <100 4,100, 500, 500, 100
A
I.,0E
1-cQ
ci
(-ci
:p
s
$.
0
on November 10, 2019 by guest
http://jvi.asm.org/
[image:7.612.343.527.73.380.2]models have recently been discussed (8, 21). In particular, the SCID-hu mouse has been found to be permissive for replication of HIV after direct injection of virus into the engrafted human lymph node or thymus (22). HIV expres-sion was apparently restricted to some transplanted human cells (22). In this system,virus replicationwassuppressed if AZT was administered before virus inoculation (23). There-fore, thismodel seems to be very useful for therapy aimed at preventing infection after acute virus exposure. One major differencebetween the modeldescribed in this report and the existing SCID models is the fact that we have directly injected micewithchronically HIV-infected tumorcells that arecapable of rapidly growing in anti-IFN-treated mice and ofproducing high amounts of HIV in vivo. Both tumor size and serum p24 levels can be easily evaluated in individual mice, both beforeand in the course ofan antiviraltherapy. In conclusion, our results suggest that HIV-infected U937 cell tumors transplanted in anti-IFN-a/,-treated mice con-stitute a simple and useful model for the therapy of chronic viremia and for evaluating the in vivo effects of different substances on HIV-infectedcells.
ACKNOWLEDGMENTS
This work was supported in part by grants from Ministero della Sanita/ISS (IIProgetto diRicherche sull'AIDS 1989, contract 4207/ 01), bytheAssociazione Italiana per laRicerca sulCancro, and by theAgence Nationale deRecherches sur leSida/C.N.R.S.(contract 89N60/0811).
We are gratefulto M. Martorelli(WellcomeSpA, Pomezia, Italy) and to L. A. Shute (The Wellcome Research Laboratories, Beck-enham, Kent, England) for providing AZT. We thank G. Forni (Institute of Microbiology, University of Turin, Turin, Italy) for helping to establish in our laboratory the optimal experimental conditions for preparation of SIA nude mice.
REFERENCES
1. Afifi, M. S., V. Kumar, and M. Bennet. 1985. Stimulation of geneticresistance to marrow grafts in mice byinterferon-a/p. J. Immunol. 134:3739-3745.
2. Balkwill, F. R., and E. M.Moodie. 1984. Positive interactions between human IFN andcyclophosphamide oradriamycin ina human tumor model system. Cancer Res. 44:904-908. 3. Balkwill, F. R., and E.Proietti. 1986. Effects of mouse IFN on
human tumor xenografts in the nude mouse host. Int. J.Cancer 38:375-380.
4. Caretto, P., M. Forni, G. D'Orazi, S. Scarpa, P. Feraiorni, C. Jemma, A. Modesti, M. Ferrarini, S. Roncella, R. Foa, and G. Forni. 1989. Xenotransplantation in immunosuppressed nude mice of human solid tumors and acute leukemias directly from patients or in vitro cell lines. Res. Clin. Lab. 19:231-243. 5. Cheers, C., and R. Waller. 1975. Activated macrophages in
congenitally athymic "nude" mice and in lethally irradiated mice. J. Immunol. 115:844-847.
6. Folks, T. M., K. A. Clouse, J. Justement, A. Rabson, E. Duh, J. H. Kehri, and A. S. Fauci. 1989. Tumor necrosis factor a induces expression of human immunodeficiency virus in a chronically infected T-cell clone. Proc. Natl. Acad. Sci. USA 86:2365-2368.
7. Forni, G., M. Giovarelli, and A. Santoni. 1985. Lymphokine-activated tumor inhibition in vivo.I.Thelocal administration of interleukin-2 triggers non reactive lymphocytes from tumor bearing mice to inhibit tumor growth. J. Immunol. 134:1305-1311.
8. Gardner, M. B., and P. A. Luciw. 1989. Animal models of AIDS. FASEB J. 3:2593-2606.
9. Gresser, I., F. Belardelli, C. Maury, M. T. Maunoury, and M. G. Tovey. 1983. Injection of mice with antibody to interferon enhances the growth of transplantable murine tumors. J. Exp. Med. 158:2095-2107.
10. Gresser, I., C. Maury, M. T. Bandu, and F. Belardelli. 1990.
Importance of interferon a in the resistance of allogeneic C5781/6 mice to the multiplication of Friend erythroleukemia cells in the liver. Int. J. Cancer 45:364-371.
11. Gresser, I., C. Maury, C. Carnaud, E. De Mayer, M. T. Manoury, and F. Belardelli. 1990. Antitumor effects of inter-feronin miceinjected withsensitive and interferon-resistant Friend erythroleukemia cells. VIII. Role of the im-mune system in the inhibition of visceral metastases. Int. J. Cancer46:468-474.
12. Gresser, I.,C.Maury,F.Vignaux,0.Haller,F.Belardelli,and M.G. Tovey. 1988.Antibodyto mouseinterferon ot/ abrogates resistancetothemultiplication of Frienderythroleukemiacells in thelivers ofallogeneic mice.J. Exp. Med. 168:1271-1291. 13. Gresser, I., M. G. Tovey, M. T. Bandu, C. Maury, and D.
Brouty-Boye. 1976. Role of interferon in the pathogenesis of virus diseases in mice as demonstrated by the use of anti-interferon serum. I. Rapid evolution ofencephalomyocarditis virusinfection. J. Exp. Med. 144:1305-1315.
14. Habu, S., H. Fukui, K. Shimamura, M. Kasai, Y. Nagai, K. Okumura, and N. Tamaoki. 1981. In vivo effects of anti-asialoGM1. I. Reduction ofNK activity and enhancement of transplantedtumorgrowthin nude mice. J. Immunol. 127:35-38.
15. Herberman, R. B. 1978. Natural cell-mediated cytotoxicityin nudemice, p. 135-166. In J. Foghand B. C.Giovanella(ed.),
The nude mouse in experimentaland clinical research. Aca-demicPress, NewYork.
16. Hunig, H., and M. J. Bevan. 1984. Ability of nude mice to
generate alloreactive, xenoreactive, and H-2-restricted cyto-toxic T-lymphocyte responses, p. 7-11. In B. Sordat (ed.), Immune-deficientanimals. S.Karger,Basel.
17. Kamel-Reid,S.,andJ.E.Dick.1989. Engraftmentof immune-deficient mice with human hematopoietic stem cells. Science 242:1706-1709.
18. Kindred,B. 1978. The nudemouseinstudyingT-cell differen-tiation, p. 111-134. In J. Fogh andB.C. Giovanella(ed.),The nude mouse in experimental and clinical research. Academic Press,NewYork.
19. Kindred, B. 1979. Nude mice in immunology. Prog. Allergy. 26:137-152.
20. Locardi,C.,C.Petrini,G.Boccoli,U.Testa,C.Dieffenbach,S. Butto, and F. Belardelli. 1990. Increased human immunodefi-ciency virus(HIV)expressioninchronicallyinfected U937 cells upon in vitro differentiation by hydroxyvitaminD3: role(s) of interferon and tumor necrosis factorinthe regulation ofHIV production. J. Virol. 64:5875-5882.
21. Lusso, P., F. Di Marzo Veronese, B. Ensoli, G. Franchini, C. Jemma, S.E.DeRocco,V. S.Kalyanaraman, andR.C. Gallo. 1990. Expanded HIV-1 cellulartropism by phenotypic mixing
with murineendogenousretroviruses. Science 247:848-852. 22. McCune, J. M.,R.Namikawa,H.Kaneshima,L.D.Shultz,M.
Lieberman, and I. L. Weissman. 1988. The SCID-hu mouse:
murine modelfortheanalysisofhumanhematolymphoid differ-entiationandfunction. Science 241:1632-1639.
23. McCune, J. M., R. Namikawa, C. C. Shih, L. Rabin, and H. Kaneshima. 1989.Suppressionof HIVinfectioninAZT-treated SCID-hu mice. Science247:564-566.
24. Mosier, D. E., R. J. Gulizia, S. M. Baird, and D. B. Wilson. 1988. Transfer ofa functional human immune systemto mice withseverecombinedimmunodeficiency.Nature(London)335: 256-259.
25. Namikawa, R., H. Kaneshima,M. Lieberman,I. L. Weissman, andJ. M. McCune. 1988. Infection ofthe SCID-hu mouseby
HIV-1. Science 242:1684-1686.
26. Poli,G.,A.Kinter,J. S.Justement, J.H.Kehrl, P.Bressler,S. Stanley, andA. S. Fauci. 1990. Tumor necrosisfactor a func-tions inanautocrine mannerinthe induction ofhuman immu-nodeficiency virus expression. Proc. Natl. Acad. Sci. USA 87:782-785.
27. Poli, G., J. M. Orenstein, A. Kinter, T. M. Folks, and A. S. Fauci. 1989. Interferona butnotAZT suppresses HIV expres-sioninchronically infectedcell lines. Science 244:575-577. 28. Reid, L.M., N.Minato, I.Gresser,J.Holland, A.Kadish, and
on November 10, 2019 by guest
http://jvi.asm.org/
B. R. Bloom.1981. Influence of anti-mouse interferonserum on
thegrowth and metastasis oftumorcells persistently infected with virus and of humanprostatictumorsin athymic nude mice. Proc. Natl. Acad. Sci. USA 78:1171-1175.
29. Shouval, D., B. Rager-Zisman, P. Quan, and D. A. Shafritz.
1983. Role in nude mice ofinterferon and natural killer cells in inhibiting the tumorigenicity of human hepatocellular carcinoma cellsinfected with hepatitis B virus. J. Clin. Invest. 72:707-717. 30. Silobrcic,V., A. L. Zietman, J. R. Ramsay, H. D.Suit,and R.S. Sedlacek. 1990. Residual immunity of athymic NCr/Sed nude mice and the xenotransplantation of human tumors. Int. J.
Cancer 45:325-333.
31. Sunstrom, C., and K. Nilsson. 1976. Establishment and charac-terization ofahumanhistiocytic lymphoma cell line (U937). Int. J. Cancer 17:565-577.
32. Tovey, M. G., J. Begon-Lours, andI.Gresser.1974. A method for thelarge-scale production ofpotentinterferonpreparations. Proc. Soc.Exp. Biol. Med. 146:809-815.
33. Zietman, A.L.,H.D.Suit, J. R. Ramsay, V.Silobrcic, and R. S. Sedlacek. 1988. Quantitative studiesonthe transplantability of murine and human tumors into the brain and subcutaneous tissues of NCr/Sed nude mice. Cancer Res. 48:6510-6516.