JOURNAL OFVIROLOGY, Mar.1976,p.865-875
Copyright0 1976 AmericanSocietyforMicrobiology Vol.
17,No.3 Printed inU.S.A.
In
Vivo and In Vitro
Synthesis
of
Adenovirus
Type
2
Early
Proteins
JOSE L. SABORIO AND BO OBERG*
Department of Microbiology, The Wallenberg Laboratory, Uppsala University, Uppsala, Sweden
Received forpublication6October 1975
The synthesis of adenovirus type 2 (Ad2)-induced early polypeptides was
examined in vivoand in vitrobyacombinationofsodiumdodecyl
sulfate-polya-crylamide gel electrophoresis alone and specific immunoprecipitation followed
by sodium dodecyl sulfate-polyacrylamidegel electrophoresis. Analysis oftotal
[35S]methionine-labeled polypeptides synthesized in vivo at 3 h postinfection
allowed us todetect in infected cells at least 13 distinctpolypeptides that are
either absent or less conspicuous in extracts from mock-infected cells. These
Ad2-induced early polypeptides have molecular weights rangingfrom 72 x 103to
10.5 x 103 and haveaccordingly been designatedasE72KtoE10.5K. Nineofthe
in vivo synthesized early polypeptides can be precipitated specifically from
infected cell extracts by antisera with specificity against early adenovirus
proteins. In vitro translation of mRNA extracted from mock-infected cells and
from Ad2-infected cells was carried out in preincubated Ehrlich ascites cell
extracts. All the earlyAd2-induced polypeptides identified in theextractsfrom
infected cells labeled in vivo were also detected among the polypeptides
immunoprecipitatedspecificallyfromthe in vitro reaction mixtures programmed
by RNA extracted at4hpostinfection from Ad2-infected cells.
Human adenovirus type 2 (Ad2) is a virus
composed of DNA and protein. The DNA is
linear and double stranded and hasamolecular
weight of 23 x 106 (10). During productive
in-fection, the information encoded in the viral
genome is expressed according to a complex
program not yetfully understood. Two distinct
phases of gene expression can, however, be
clearly recognized: an early phase, which
in-cludes events occurring priorto and
independ-entlyofviralDNAreplication, andalatephase,
which depends on the onset of viral DNA
replication. Practically nothing isknown about
the mechanisms controlling the sequential
ex-pression oftheviralinformation, andthe switch
from the early to the late phases (for a review
see Philipson et al. [17]). Prior tothe onset of
viral DNAreplication, RNA sequences
comple-mentary to 45% of the asymmetric coding
capacity of the viral genome are found as
mRNA in the cytoplasm of infected cells (27).
This"early"RNAhasbeenmapped in the viral
genome (U. Pettersson, C. Tibbetts, and L.
Philipson, Cell, in press; 18, 20), and recent
experimentsindicate thatit might be composed
of a minimum of seven discrete species of
mRNA (6). Part of the information expressed
during the early phaseofproductiveinfection is
also expressed in Ad2-transformed rat cells (8,
9).Identification of the early virus-coded
func-tionsis, therefore, essentialto the
understand-ing of both the lytic infection and the role
played by the virus in the establishment and
maintenance of the transformed-cell
pheno-type.
In this article we report attempts to
charac-terize the early virus-induced polypeptides by
bothinvivoandinvitrostudies. The combined
use of high-resolution gel electrophoresis and
specific immunoprecipitation has allowedusto
detect at least 14 distinct early virus-induced
polypeptides which donot seemtobepresentin
mock-infected cells. Most ofthesepolypeptides
can be synthesized in vitro in cell-freeextracts
programmed with RNA extracted from infected
cells. An enhanced in vitro translation ofearly
virus-induced polypeptides is observed with
RNA extractedfrom infected cellstreatedwith
cycloheximide.
MATERIALS AND METHODS
Cells and virus infection. HeLa cellsweregrown
in suspension cultures in Eagle spinner medium
supplemented with 7% calf serum andinfected with 865
on November 10, 2019 by guest
http://jvi.asm.org/
Ad2 (1 PFU/200 particles) at a multiplicity of 104 virusparticles/cell as previously described (11).
Labeling andfractionation of cells. Ad2-infected and mock-infected cells were collected from growth medium, washed twice, and suspended in growth medium without aminoacidsata density of4 x 106
cells/ml. [36S]methionine (5 MCi/ml) was added for
various times, and the cells were lysed in isotonic buffer (20 mM Tris-hydrochloride, pH 7.6-0.15 M
KCl-5 mM Mg acetate) by the addition of 0.5%
Nonidet-P40 and fractionated into a nuclear and a cytoplasmic fraction bycentrifugation at 2,000 x g for 10min.Portions of thecytoplasmic extracts were used directly forimmunoprecipitationorprecipitated with 10% trichloroacetic acid for analysis of labeled poly-peptides by polyacrylamide gel electrophoresis. The nuclear pellets were digested with DNase in "high-saltbuffer"according to Penman (16). Portions of the digested nuclear suspensions were precipitated with 10% trichloroacetic acid for analysis of labeled
poly-peptides.
Invitrotranslation. Total cytoplasmic RNA was extracted fromAd2-infected and mock-infected cells
asdescribed previously(13) andtranslated in vitro in
preincubated S30 extracts from Ehrlich ascites cells.
The assayconditions and preparation of the samples
forimmunoprecipitationandsodium dodecyl sulfate
(SDS)-polyacrylamide gel electrophoresis have been
describedpreviously (14).
Immunoprecipitation. Immunoprecipitation was carried out by a double-antibody technique as de-scribed previously (14) but with ureatreatment omit-ted. Allsamples and antisera were centrifuged for 15 min at 16,000 x g prior to immunoprecipitation. Incubation of the sample with the first
immunoglobu-linG (IgG)wascarried outfor 1h at room
tempera-ture.The secondIgGwasadded toequivalence, and
the sample was left at 4 C for at least 2 h. The
precipitate was collected, washed, and prepared for
SDS-polyacrylamide gel electrophoresis asdescribed
previously(14).Rabbit serum with specificity against
the P antigen (21) was kindly provided by W. C.
Russell, Mill Hill, London. A second antiserum
against a crude preparation of early proteins was preparedbyinjecting rabbits with extractsofKB cells infected with Ad2inthe presence of40ggofcytosine
arabinoside/ml (E. Everitt, unpublished data). This
antiserumwasadsorbedfor2hat 37C withan extract
ofdisrupteduninfected KB cells beforebeingusedin
theseexperiments. Antiserumwith broad specificity
against the Tantigens (3)was kindlyprovided byR.
Gilden and was prepared from hamsters bearing
tumors induced by an Ad2-simian virus 40 hybrid.
This antiserum reacts strongly against the DNA binding proteins of adenoviruses (T. Linne and L. Philipson, manuscript in preparation). The IgG's
usedinthesecond stepofimmunoprecipitation were swine IgG directed against rabbit IgG (Dakopatts)
and rabbit IgG directed against hamster IgG
(Hoechst).
SDS-polyacrylamide gel electrophoresis.
Elec-trophoresis in 13% or 10 to 18%polyacrylamide slab
gels with a separation distance of 8 or 28 cm was
performedaccordingtoMaizel(12).After
electropho-resis the gelswere impregnated with 2,5-diphenylox-azole, dried, andexposed to RP-14 Kodak X-ray film (4). Electrophoresis was in all cases carried out until themarker, bromphenol blue, reached the bottom of the gels.
Materials. Cycloheximide, cytosine arabinoside, pactamycin, L-1-tosyl-amide-
2-phenylethylchloro-methyl ketone (TPCK),
N-a-p-tosyl-lysine-chloro-methyl ketone (TLCK), and DNase (EC 3.1.4.5) were from Sigma. [35S]methionine, 40 to 50 Ci/mmol, was fromAmersham.
RESULTS
Ad2-induced early polypeptides
synthe-sized in vivo. Ad2-infected HeLa cells were
pulse labeledfor 30 minwith [35S]methionineat
either 3 or 18 h postinfection and fractionated
intocytoplasmic and nuclear fractions.Labeled
polypeptidesinboth fractionswereanalyzedby SDS-polyacrylamide gel electrophoresis
fol-lowed by autoradiography of the dried gels.
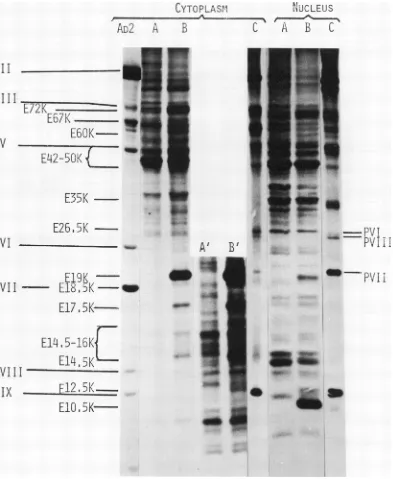
Figure 1 shows the pattern oflabeled
polypep-tidesfrom Ad2 and mock-infected cells labeled
under thesameconditions. At3hpostinfection
several bands are detected in infected cells
which are absent or less conspicuous in
mock-infected cells. In the cytoplasmicfraction (Fig.
1) at least 13 virus-induced polypeptides are
detectedwhen mock-infected and infected cells
are compared. These early polypeptides have been designated, according to their estimated molecular weight, as E72K, E67K, E60K,
E42-50K, E35K, E26.5K, E19K, E18.5K,
E17.5K, E14.5-16K, E14.5K, E12.5K, and
E10.5K. Thenumberofinduced
polypeptides
inthe region 42-50K and 14.5-16K is uncertain. In
the nuclear fraction, three virus-induced early
polypeptides are detected: E19K, E12.5K, and
E10.5K (Fig. 1). All thesepolypeptidesseem to
be true early products, since they are also
synthesizedincellsinwhich viral DNA
replica-tion is inhibited by cytosine arabinoside or
hydroxyurea (data notshown).
Immunoprecipitation
ofAd2-inducedearly
polypeptides
synthesized
invivo.Some oftheAd2-induced earlypolypeptidesshowninFig.1,
viz., E19K, E17.5K, and E10.5K, are always
easily detected among the high
background
ofhostcell
polypeptides,
whereas otherscanonly
be detected after findingthe
optimal
exposuretime forautoradiographyofdifferentsections of
thegelsand,eventhen,thedifferencesbetween
infected and mock-infected cell extracts might
be very faint
(polypeptides
E67K and E35K).An attempt was made to detect the
virus-inducedearlypolypeptides, withminimal
inter-ference fromhostcell
polypeptides,
by
immuno-precipitating
[35S]methionine-labeled
on November 10, 2019 by guest
http://jvi.asm.org/
Ad2EARLY PROTEINSYNTHESIS 867
CYTOPLASM
AD2
A
B
C
III
[67KE60K
--E35K
E26. 5K
V I
X
_--A
E19K
VII
-E18.5K
-n_
E17.5K--E14.5-16K[
E14,5K
VIII
Ix
F
12,5K-E10
.5K
..
A
l*B
PW
.0 r_
_4
mm
..
.,4
_ --mq- '-wPVT
_. _ .
---
PII I
Isp*
FIG. 1. SDS-polyacrylamidegelelectrophoresisof cellextractsfromAd2-infected and mock-infected HeLa cells.Ad2-infectedHeLacellswereharvestedat3and18hpostinfection andsuspendedatadensityof5 x 10'
cells/ml ingrowth medium lacking amino acids and calfserum. [35S]methionine (5ACi/ml)wasaddedto the
cellsuspensions,andincubation wascontinued at37C for30min. The cellswerecollected andfractionated
into cytoplasmic and nuclear extracts, and the extracts were analyzed by SDS-polyacrylamidegel electro-phoresis. Theslab gels wereimpregnated with 2,5-diphenyloxazole (4),dried, and exposedtoX-ray film. (A) Mock-infected cells; (B) early-infected cells; (C) late-infected cells.A'and B'are longerexposure times of A
andB, respectively.
NUC
LEUSA
B C
V
VOL.17, 1976
qm1.
E42-50K
I
on November 10, 2019 by guest
http://jvi.asm.org/
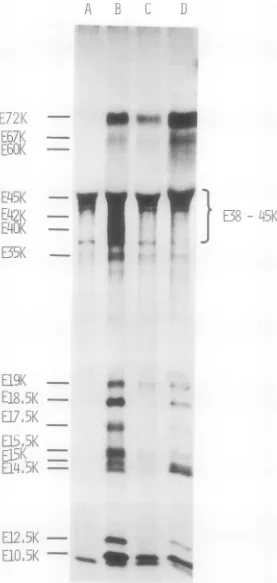
[image:3.491.44.437.78.557.2]mic extracts from infected cells with three different antisera with specificity against
virus-induced early proteins. Two controls were
in-cluded tocheck the specificity of the
immuno-precipitation; infected-cell extracts were
im-munoprecipitated by using serum prepared
from nonimmunized rabbits, and extracts from
mock-infected cells were immunoprecipitated
with all four antisera. The immunoprecipitates
were analyzed by SDS-polyacrylamide gel
elec-trophoresis followed by autoradiography. As
showninFig. 2, several polypeptides from early
4'
I
__m
IWh
W_
_4-0
w0
FIG. 2. Immunoprecipitation of[3("SJmethionine-labeledpolypeptides from Ad2-infectedandmock-infected
cellextracts. CytoplasmicextractsfromAd2-infectedandmock-infectedcells labeledasindicated in thelegend to Fig. I were immunoprecipitated by a double-antibody technique as described. The precipitates were analyzed by SDS-polyacrylamide gel electrophoresis followed by autoradiography of
2,5-diphenyloxazole-impregnated slab gels. The antisera used during the first step ofimmunoprecipitation were: A, IgG from
nonimmunized rabbits; B, rabbit IgG directed against Pantigen; C, rabbit IgG directed against a crude
preparation ofearlyproteins;D,hamsterIgGdirectedagainst Tantigen. Theseraused in the secondstepof
immunoprecipitationwereswineIgGdirectedagainstrabbitIgGand rabbitIgGdirectedagainsthamsterlgG.
W,,.
on November 10, 2019 by guest
http://jvi.asm.org/
[image:4.491.53.458.147.585.2]VOL. 17, 1976
infected cells are immunoprecipitated by the
three antisera with specificity against early
proteins (Fig. 2B-D), but not when IgG from
nonimmunized rabbits isused (Fig. 2A). Some
polypeptides are precipitated nonspecifically
from mock-infected cell extracts, but they do
not coincidewith those from infectedcells. One
exception is a polypeptide with molecular
weight around 45K, which is quiteconspicuous
in the extracts from mock-infected cells and
might contribute to the radioactivity found in
the same position in the extractsfrom infected
cells. The pattern obtained fromearly-infected
cells with P antiserum and an antiserum
pre-pared against crude preparations ofearly
pro-teins shows some difference, notably in the
regions 60K, 42K, 26.5K, and 19K. Altogether
one candetectninepolypeptides, atleastseven
ofwhich correspondtoearlyvirus-induced
poly-peptides detectedwithoutimmunoprecipitation
in Fig. 1: E72K, E67K, E60K, E35K, E26.5K,
E19K, and E17.5K. The two other early
poly-peptidesunraveledby immunoprecipitation are
designated E45K and E42K. As mentioned
before the apparently complexzone about 45K
might include host cell polypeptides. The T
antiserum precipitates polypeptides E72K,
E67K, E60K, and E45K. Notethat the
polypep-tidebetween E72K and E60KinFig. 2C (early)
migrates slightly slower than the corresponding
polypeptide in Fig. 2B and D (early). This
differencehas been noticed inseveral gels.
Late synthesis ofvirus-speciftic early
pro-teins. Cytoplasmic and nuclear extracts from
infectedcellslabeled with [35S
]methionine
at 18h postinfection were analyzed to determine
whether some ofthe virus-induced early
poly-peptides are also synthesized late during lytic
infection. Two early polypeptides, E72K and
E19K, seem to be present late in the
cytoplas-mic fraction (Fig. 1), although the amount of
polypeptide E19K is much lower than at 3 h
postinfection. In the nuclear fraction of
late-infected cells (Fig. 1), a small amount of
poly-peptide E10.5K is also detected. Polypeptides
E72K, E45K, and trace amounts ofE19K are
precipitated from cytoplasmic extracts of
late-infected cells by antisera prepared against a
crudepreparationofearly proteins (Fig. 2).
Pulse-chase experiments and effect of
TPCK and TLCK on the synthesis of
virus-induced early polypeptides. Two types of
ex-periments were carried out in an attempt to
detect any possible precursor-product
relation-ship between the virus-induced early
polypep-tidessynthesizedinvivo: (i) infected cells were
pulse labeled with [35S]methionine at 3h
post-infection for periods as short as 5 min and
Ad2EARLYPROTEINSYNTHESIS 869
chased for differenttimes, and (ii) infected cells
were pulse labeled at 3 h postinfection with
[35S
Imethionine
for 30 min in the presence ofeither TPCK orTLCKatup to 10-4 M, which
inhibited protein synthesis by 95 and 45%,
respectively. Labeled polypeptides from both
typesof experimentswereanalyzedasindicated
in Fig. 1 and 2. All the virus-induced early
polypeptidesweredetected aftera5-minpulse,
and no major changes were noticed during
chases up to 5 h. The patterns ofpolypeptides
synthesized in the presence ofeither TPCK or
TLCK were qualitatively identical to those
showninFig. 1and 2.
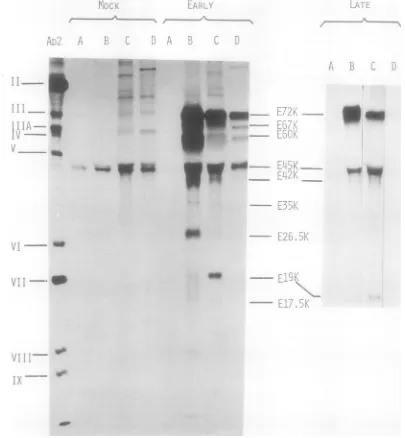
In vitro synthesis of virus-induced early
polypeptides. Total cytoplasmic RNAwas
pre-pared from mock-infected cells and from
in-fected cells at 4 h postinfection and translated
in vitro in a preincubated cell-free extract as
describedpreviously (14). Theinvitroproducts
labeledwith [35S]methionine wereanalyzed by
SDS-polyacrylamide gel electrophoresis
fol-lowedby autoradiography (Fig. 3). Comparison
ofthe polypeptides synthesizedinextracts
pro-grammed with RNA from mock-infected cells
(Fig. 3A) and with RNAfrominfected cells (Fig.
3B) reveals the presence, among the latter, of
three polypeptides which are absent or less
conspicuous in the first case. Although the
differences are faint, polypeptides with
mobili-ties corresponding to those of polypeptides
E72K, E14.5K, and E1OK synthesized in vivo
(Fig. 3A and B) can be detected in theextract
programmed with RNA from infected cells (Fig.
3B). These same three polypeptides are also
synthesized with RNA extractedat 4 h
postin-fection from infected cells incubated in the
presence ofhydroxyureaorcytosinearabinoside
(Fig. 3C and D).
In vitro translation of RNA from cells
incubated with inhibitors ofprotein
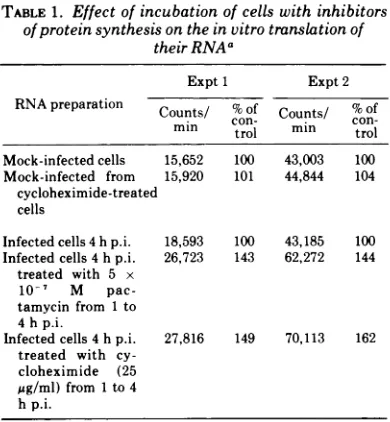
synthe-sis. Parsons and Green (15) found that the
addition of 25
gg
ofcycloheximide/ml toAd2-infected cell culturesat 1 hpostinfection results
inup to a 10-fold increase inthe percentage of
newly made RNA which hybridizes to viral
DNA. This RNAseems to include all the early
sequences found in untreated, infected cells
(15). An attempt was made to obtain RNA
preparations enriched with virus-specific early
sequences by incubating infected cells with 25
Ag
ofcycloheximide/mlfrom 1 to 4 hpostinfec-tion. In a parallel culture, infected cells were
incubated with 5 x 10-7 M pactamycinduring
thesametimeinterval. Totalcytoplasmic
RNA
was extracted from these cultures and from
mock-infected cells incubated under the same
conditions with both inhibitors of protein
on November 10, 2019 by guest
http://jvi.asm.org/
870 SABORIO AND
6BERG
m
m
-FIG. 3. SDS-polyacrylamidegelelectrophoresis of in vitro synthesized polypeptides. Total cytoplasmic RNA
from mock-infected andAd2-infected cells treated under various conditions was translated in preincubated
cell-freeextracts asindicated in Table1.The [35S]methionine-labeled productswereanalyzed by
SDS-polya-crylamidegelelectrophoresisfollowedby autoradiography. The RNApreparationswerefrom: A, mock-infected
cells; B, infected cells harvested at 4 h postinfection; C, infected cells incubated with 40 ug of cytosine
arabinoside/mlfrom 1 to 4hpostinfection; D, infected cells incubated with20mMhydroxyurea from 1 to4
hpostinfection;E,infected cellsincubated with5 x 10-IMpactamycinfrom1to 4hpostinfection; F,infected
cellsincubatedwith25Mgofcycloheximide/mlfrom 1 to4hpostinfection; G, mock-infectedcellsincubated
with25Agofcycloheximide/ml foraperiod of3h.
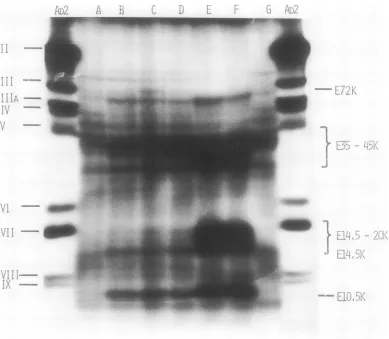
thesis. Table 1 shows that the RNA
prepara-tions obtained from infected cellstreated with
either pactamycin or cycloheximide are more
efficiently translated in vitro than those from
untreated, infected cells. Preincubation with
the inhibitors resultsinabouta50%increase in
the amount of radioactivity incorporated into
protein during in vitro translation. This more
efficient translation is not accompanied by a
uniform increase inthesynthesisof all
polypep-tides synthesized in vitro with the RNA from
infected, untreated cells but rather by an
in-crease in the synthesis of only a few
polypep-tides (Fig. 3E and F). A marked increase was
detected in polypeptide E10.5K and in two
zones including polypeptides with molecular
weights between14,000 to 20,000and 35,000to
45,000 (Fig. 3E and F). No quantitative or
qualitative differences weredetected during in
vitro translation of RNA from untreated or
cycloheximide- or pactamycin-treated
mock-infected cells(Table 1; Fig. 3G).
Immunoprecipitation of in vitro
synthe-sized
polypeptides.
Total cytoplasmic RNAwas extracted from mock-infected cells, from
infected cells at 4 h postinfection, and from
infected cells treated withcycloheximidefrom1
to4 orfrom 1to18hpostinfection. TheseRNA
preparations were translated in vitro, and the
products were precipitated with an antiserum
on November 10, 2019 by guest
http://jvi.asm.org/
[image:6.491.67.456.70.409.2]Ad2 EARLY PROTEIN SYNTHESIS 871
TABLE 1. Effect of incubationofcellswith inhibitors of proteinsynthesis on the in vitro translation of
theirRNAa
Expt 1 Expt2
RNA preparation Counts/ %Of
Counts/
%ofmn con- mn
con-mmn trol mm trol
Mock-infected cells 15,652 100 43,003 100
Mock-infected from 15,920 101 44,844 104
cycloheximide-treated cells
Infected cells4hp.i. 18,593 100 43,185 100
Infected cells4hp.i. 26,723 143 62,272 144
treated with 5 x
10-7 M
pac-tamycin from 1 to
4h p.i.
Infected cells4hp.i. 27,816 149 70,113 162
treated with cy-cloheximide (25
pg/ml) from1to 4
hp.i.
aCytoplasmic RNAat aconcentration of0.6mg/mlwas translated in a preincubated S30 ascites extract, and the
incorporated ["SImethioninewascountedasdescribed previ-ously (15).p.i.,Postinfection.
with specificity against virus-induced early
polypeptidesorwithserum fromnonimmunized
rabbits. Theimmunoprecipitateswereanalyzed
by SDS-polyacrylamide gelelectrophoresis
fol-lowed by autoradiography. The polypeptides
precipitated with the nonspecific serum have a
pattern identical to that shown in Fig. 2A. As
showninFig. 4A, theantiserumdirected against
early proteins precipitates a few polypeptides
from the in vitro translation directed by RNA
extracted from mock-infected cells. This
non-specificprecipitationis mostconspicuousinthe
region corresponding to polypeptide E45K. On
the other hand, the precipitates from the in
vitro translation programmed by the three
dif-ferentpreparationsof"early"RNAare
qualita-tively similar to each other (Fig. 4B-D) and
contain severalpolypeptidesnot present in the
products ofthe RNA from mock-infected cells
(Fig. 4A). Theseapparently virus-inducedearly
polypeptides migrate in the position
corre-sponding to polypeptides E72K, E67K, E60K,
E45K, E42K, E40K, E35K, E19K, E18.5K,
E17.5K, E15.5K, E15K, E14.5K, E12.5K, and
E10.5K.Thepolypeptidewith estimated
molec-ularweight of40K and a series ofpolypeptides
migrating between 16K and 14.5K seem to be
alsovirus-induced,although they havenotbeen
clearlyresolved in the in vivo products (Fig. 1
and 2). It is difficult to decide whetheror not
there is a virus-induced polypeptide
corre-sponding to E45K, sincea polypeptide
migrat-ing in that position is nonspecifically
precipi-tated from all the samples by serum from
nonimmunized rabbits. Translation of RNA
frominfected cycloheximide-treated cells shows
somedifferenceswhen RNA is extracted at 4 h
or 18 h postinfection. At the later time all
polypeptides but E72K, E67K, E60K, E15K,
E14.5K, and E10.5K show a strong decrease
(Fig. 4B and D).
DISCUSSION
The identification of Ad2-coded functions
expressed duringthe early phase ofproductive
infection poses some specialproblems. Theuse
of conditional lethal mutants to identify viral
functions has not been as successful with
ani-mal viruses as it hasbeen with bacteriophages.
Genetic studies with adenovirus mutants have
revealed a few complementation groups, and
only two complementation groups have been
ascribed to early regions of the ad5 gene,
namely, ts36and ts125 (7, 31), and three foradl2
(26). An alternative approachtoidentify
virus-coded functions wouldbetheinvitrosynthesis,
orisolationfrominfected cells, of RNA
comple-mentary to the viral genome, followed by in
vitro translation. This latter approach would
require a combination of in vivo and in vitro
studies, since the authenticity of any in vitro
synthesized polypeptide would be dubious
un-less it could be compared with an in vivo
synthesized polypeptide. However,
identifica-tion ofAd2-induced early polypeptides
synthe-sized in vivo is hampered by the dominant
synthesisofhost cell proteins during theearly
phase of productive infection. In this work a
polypeptide was considered as virus-induced
when detected in samples from infected cells,
but not from mock-infected cells, whether by
simple SDS-polyacrylamide gel electrophoresis
or by immunoprecipitation followed by
SDS-polyacrylamide gelelectrophoresis. Underthese
criteria, all polypeptides presented in Table 2
are virus induced. The only dubious case is
polypeptide E45K, which is precipitated from
mock-infected and infected cells by a
nonspe-cific serum (Fig. 2A). A catalogue of
virus-induced polypeptide previously detected is
pre-sented inTable 3. Polypeptide E72K probably
correspond to polypeptides ICSP-3 (21), El
(30), and the large DNA-binding protein (28)
detected byothers. Polypeptide E45K seems to
beequivalent to the smallDNA-bindingprotein
(27) and to the polypeptide called core 1 by
Bablanian and Russell (2). Polypeptide E19K
corresponds to polypeptide E2 (30) and
proba-blytop core2 (2),although in our results a clear
distinction can be made between p-VII and
VOL.17, 1976
on November 10, 2019 by guest
http://jvi.asm.org/
ea __s
0at
Vt
II
-f.f
FIG. 4. Immunoprecipitation of in vitro synthesized polypeptides. Total cytoplasmic RNAs from
mock-infected cells andfrominfectedcells treated underdifferentconditionsweretranslated in vitroasindicated in Table 1. Theproducts were immunoprecipitated andanalyzed by SDS-polyacrylamidegel electrophoresis
followedbyautoradiographyofthedried slabgels.Theserumused in thefirststepoftheimmunoprecipitation wasrabbitIgG directed againsta crudepreparation of earlyproteins. The RNAs translated werefrom:A,
mock-infected cells; B, infectedcells treated with25Mgofcycloheximide/mlfrom 1 to4hpostinfectionand
harvestedat4 h postinfection; C, untreatedinfected cells harvested at 4h
postinjection;
D, infected cells treated with25Mgofcycloheximide/ml from 1to18hpostinfectionandharvestedat 18hpostinfection.872
on November 10, 2019 by guest
http://jvi.asm.org/
[image:8.491.118.395.31.614.2]Ad2 EARLYPROTEINSYNTHESIS 873
TABLE 2. Adenovirus-induced early proteins detected in this study
In vivo In vitro
Immunoprecipi- Immunoprecipi-SDS-polyacrylamide station+SDS- tation +
SDS-electrophoresis polyacrylamide polyacrylamide gelelectropho- gel
electropho-resis resis
E72K E72K E72K
E67K E67K E67K
E6OK E60K E60K
E42-50K E45K E45K
E42K E42K
E40K
E35K E35K E35K
E26.5K E26.5K
E19K E19K E19K
E18.5K E18.5K
E17.5K E17.5K E17.5K
E14.5-16K E15.5K
E15K
E14.5K E14.5K
E12.5K E12.5K
E10.5K s E10.5K
E19K (Fig. 1). Polypeptide E10.5Kcorresponds
toE3 (30).
Van der Vlietetal. (29) have shown thattwo
virus-induced early proteins (molecular weight,
72K and 45K), synthesized at the permissive
temperatureincells infected withad5ts125, are temperature sensitive in vitro with regard to
their DNA binding properties and are very
likely virus coded. These authorssuggested that
thepolypeptide 45K isadegradation product of
the polypeptide 72K, and this is also indicated
by Rosenwirth et al. (20) and bytryptic
map-ping (Linne and Philipson, manuscript in
prep-aration). The hybridization experiments of
At-kins et al. (1) show that 72K, 55K, 42K, 19K,
15.5K, 15K, and 1lK are virus coded. It isnot
clear at present whether the otherearly
poly-peptides sofardetected arevirus codedorhost
cell polypeptides induced by the virus.
There-fore, any qualitative and quantitative
correla-tion betweenvirus-specific early RNA and early
proteins can only be tentative at the moment.
The pulse-chase experiments and the
experi-mentsinwhichtheearly polypeptides detected
in this study were labeled in the presence of
TPCK and TLCK suggest that all the
polypep-tides listed in Table 2 are primary translation
products. The combined molecular weight of all
distinct polypeptides amounts to 4.86 x 106,
about42% oftheasymmetric coding capacity of
the viral genome. Theoretically all these
poly-peptides could be virus-coded functions, since
during the early phase of infection RNA
se-TABLE 3. Adenovirus-inducedearlyproteins
detected inprevious studies
Reference Virusused Synthesis Proteins detected Russell andSkehel Ad5 In vivo ICSP-3 (64K), core
(22) 1 (46K)
Bablanian and Ad5 In vivo pcore 2(NC),a
Russell (2) ICSP-5 (NC)
Walter and Maizel Ad2 In vivo El (70K), E2
(30) (19K), E3 (11K)
VanderVlietet al. Ad5 In vivo 72K, 45K
(29)0
Shanmugametal. Ad2 In vivo 75K, 45K
(24)
Saborioetal.(23) Ad2 In vivo E70K, E45K, E19K, E17K, ElOK Atkins et al. (1) Ad2 In vitro 72K, 52K, 45K,
19K, 15.5K, 15K, 11K Saborioetal. (23) Ad2 In vitro E70K, E30-40K,
E19K, E17K, ElOK
aNC,Not calculated.
'DNA-bindingproteins.
quences complementary to up to 45% of the
coding capacity ofthegenome arefoundinthe
cytoplasm ofinfected cells (27). Tibbetts etal.
(27) and Sharp et al. (25) found that all the
early RNA sequences are present late in the
cytoplasm ofinfected cells. However, only four
ofthe early polypeptides, E72K, E45K, E19K,
E10.5K,seem tobesynthesizedlate (Fig. 1and
3).These results suggest thatsomeoftheearly
mRNA's might be stable but present in a
nontranslatable form in the cytoplasm of
late-infected cells.
Thevirus-induced earlypolypeptides
synthe-sizedinvitrowere nottranslatedwith thesame
efficiency from the various preparations of
"early"RNAused inthese studieseventhough
thesamenumberofcellequivalentsofRNAwas
used. From a quantitative point of view, the
most efficiently translated RNAs were those
obtained from infected cells treated with
cy-cloheximide orpactamycin (Table 1).
Preincu-bation ofinfected cells with either inhibitorof
proteinsynthesis resultedinRNA preparations
up to 50% more active during invitro
transla-tion than those obtained from untreated,
in-fected cells. However, immunoprecipitation of
the in vitro products followed by
SDS-polya-crylamide gel electrophoresis does not reveal
anymajordifferencesinthepattern of
polypep-tidessynthesized by the RNAs from untreated
andcycloheximide- orpactamycin-treated cells
(Fig. 4). At present we have no clue as to the
mechanism by which pretreatment of the
in-VOL.17,1976
on November 10, 2019 by guest
http://jvi.asm.org/
[image:9.491.248.443.63.290.2]fectedcellsresults inanenhancementof in vitro
translation, but the effect seems to be specific
for infected cells (Table 1). Two pieces of
information are relevant in this respect: (i)
recent experiments by Craig and Raskas (5) suggest that no increase in the mass of
viral-specific RNA occurs as a result of incubating
Ad2-infected cells with cycloheximide, and (ii)
Reichman and Penman (19) have shown that in
vitro initiation of protein synthesis is more
efficient in cell-free extracts prepared from
cycloheximide-treated cells than in extracts
from untreated cells and have postulated that
theinhibition of protein synthesis in vivo
stimu-lates the synthesis of a low-molecular-weight
RNA involved in the initiation process.
What-everthemechanism, it is clear fromour
experi-ments that the effect ofcycloheximideis more
pronouncedininfected cells andindependentof
the mechanism ofinhibitionofprotein
synthe-sis, since the same effect isobtained during in
vitrotranslation when 5 x 10-7 M pactamycin
is added at 1 h postinfection to infected cell
cultures. Further experiments are necessary to
understand the effect of these inhibitors of
protein synthesison Ad2-infectedcells.
ACKNOWLEDGMENTS
This investigation was supported by grants from the Swedish Cancer Society, J. L.S.waspartlysupported bya
postdoctoral fellowship from CONACYT, Mexico.
We are indebted to L. Philipson for many valuable discussions andtoAgneta Nordstromfordedicated technical assistance.
LITERATURE CITED
1. Atkins, J. F., J. B. Lewis, C. W. Anderson, P. R.
Baum, andR.F.Gesteland.1975.Mappingof
adeno-virus 2 genes by translation of RNA selected by hybridization, p. 293-298. In A. L. Haenni and G.
Beaud(ed.), Invitrotranscription andtranslation of
viralgenomes.INSERM, Paris.
2. Bablanian, R., and W. C. Russell. 1974. Adenovirus
polypeptide synthesis in thepresenceof non-replicat-ingpoliovirus.J. Gen. Virol.24:261-279.
3. Black, P. H., A. M. Lewis, N. R. Blacklow, J. B.
Austin, and W. P. Rowe. 1967. The presence of
adenovirusspecific antigensinhamstercells rendered
neoplastic by adenovirus 1-SV40 and adenovirus
2-SV40 hybrid viruses. Proc. Natl. Acad. Sci. U.S.A.
57:1324-1330.
4. Bonner, W. M., and R. A. Laskey. 1974. A film
detection method for tritium-labeled proteins and
nucleic acidsinpolyacrylamide gels.Eur. J. Biochem. 46:83-88.
5. Craig, E. A., and H. J. Raskas. 1974. Effect of cycloheximideonRNAmetabolismearlyinproductive infection with adenovirus2.J. Virol. 14:26-32. 6. Craig, E. A., S. Zimmer, and H. J. Raskas. 1975.
Analysis ofearly adenovirus 2 RNA using EcoR R1
viral DNAfragments.J.Virol. 15:1202-1213.
7. Ensinger,M.J.,and H. S. Ginsberg. 1972. Selection andpreliminarycharacterization of
temperature-sensi-tive mutants of type 5 adenovirus. J.Virol.10:328-339.
8. Gallimore, P. H., P. A.Sharp, and J.Sambrook.1974. Viral DNA in transformed cells. II. A study of the
sequencesofadenovirus2DNAinnine linesof trans-formedrat cellsusing specific fragments ofthe viral
genome.J. Mol.Biol.89:49-72.
9. Green, M.,J.T. Parsons, M.Pina,K. Fujinaga,H. Caffier, andI.Landgraf-Leurs. 1970.Transcription
of adenovirus genes in productively infected and in
transformed cells. Cold SpringHarbor Symp.Quant. Biol.35:803-818.
10. Green, M., M. Pina, R. C. Kimes, P. C.Wensink,L. A.
MacHattie, andC.A.Thomas,Jr. 1967. Adenovirus
DNA. I. Molecular weight and conformation. Proc. Natl. Acad. Sci.U.S.A.57:1302-1309.
11. Lindberg, U., andT.Persson.1972.IsolationofmRNA from KB-cells by affinity chromatography on
poly-uridylic acid covalently linked to Sepharose. Eur. J.
Biochem. 31:246-254.
12. Maizel, J. V. 1971. Polyacrylamide gel electrophoresis
ofviral proteins,p. 180-244. InK. Maramoroschand
H. Koprowski (ed.), Methods in virology. Academic
Press, Inc.,New York.
13. Oberg, B., and L. Philipson. 1969. Replication of poliovirus RNA studied by gel filtration and electro-phoresis.Eur. J.Biochem. 11:305-315.
14. Oberg,B., J.Saborio, T. Persson, E. Everitt, and L. Philipson. 1975.Identificationof thein vitro transla-tionproducts of adenovirus mRNA by
immunoprecipi-tation.J. Virol. 15:199-207.
15. Parsons, J. T., and M. Green. 1971. Biochemical studies on adenovirus multiplication. XVIII.
Resolu-tion of early virus specific RNA species in adeno 2-infected andtransformed cells. Virology 45:154-162.
16. Penman, S. 1969. Preparation of purified nuclei and nucleoli from mammalian cells, p.35-48.In K.Habel and N. P. Salzman (ed.), Fundamental techniquesin
virology.Academic Press, Inc., New York.
17. Philipson, L., U.Pettersson, and U.Lindberg. 1975.
Mapping ofearly and late viral RNA of adenovirus DNA, p. 22-25. In S. Gard and C. Hallayer (ed.), Molecular biology of adenoviruses. Virology Mono-graphs14.Springer-Verlag, Vienna.
18. Philipson, L., U. Pettersson, U. Lindberg, C.Tibbetts, B.Vennstrcm,and T. Persson. 1974. RNAsynthesis and processing in adenovirus infected cells. Cold Spring Harbor Symp. Quant. Biol. 39:447-456.
19. Reichman, M., and S. Penman. 1973. Stimulationof
polypeptide initiation in vitro afterprotein synthesis inhibitionin vivo inHeLa cells. Proc. Natl.Acad.Sci. U.S.A. 70:2678-2682.
20. Rosenwirth, B., K. Shiroki, A. J. Levine, and H.
Shimojo. 1975. Isolationandcharacterizationof ade-novirus type 12 DNA binding proteins. Virology
67:14-23.
21. Russell, W. C., and B.Knight.1967.Evidence fora new
antigen within the adenoviruscapsid. J. Gen. Virol. 1:523-528.
22. Russell, W. C., and J. J.Skehel. 1972.Thepolypeptides
ofadenovirus-infected cells. J. Gen. Virol.15:45-47. 23. Saborio, J.,B.oberg,and L.Philipson. 1975.In vitro
synthesis of adenovirus type 2 early proteins, p.
325-330. In A.L. Haenniand G. Beaud(ed.), Invitro
transcription and translation of viral genomes.
IN-SERM, Paris.
24. Shanmugam, G., S. Bhaduri, M. Arens, and M.
Green. 1975.DNAbindingproteinsinthe cytoplasm
and in a nuclear membrane complex isolated from
uninfectedandadenovirus2infected cells. Biochemis-try 14:332-337.
25. Sharp,P.A.,P. H. Gallimore,and S. J. Flint. 1974. Titration of viral RNA sequences in adenovirus 2
on November 10, 2019 by guest
http://jvi.asm.org/
Ad2 EARLYPROTEIN SYNTHESIS 875 lytically infected cells and transformed cell lines.Cold
Spring Harbor Symp. Quant. Biol. 39:457-474.
26. Shiroki, K., and H. Shimojo.1974.Analysisof
adenovi-rus12temperature-sensitivemutantsdefective in viral DNAreplication. Virology61:474-485.
27. Tibbetts, C., U. Pettersson, K. Johansson, and L. Philipson. 1974.Relationshipof mRNA from produc-tively infected cellstothe complementary strandsof
adenovirustype2DNA.J.Virol. 13:370-377. 28. Van derVliet, P. C., and A. J. Levine. 1973.
DNA-binding proteinsspecific forcellsinfectedby
adenovi-rus.Nature (London)New Biol. 246:170-174.
29. Van derVliet,P. C., A. J. Levine, M. J. Ensinger, and
H. Ginsberg. 1975. Thermolabile DNA binding
pro-teinsfrom cellsinfected with atemperature-sensitive
mutantofadenovirusdefective in viral DNAsynthesis. J. Virol. 15:348-354.
30. Walter, G.,andJ.V. Maizel,Jr. 1974. The polypep-tides of adenovirus. IV. Detection of early and late
virus-induced polypeptides and their distribution in
subcellular fractions.Virology57:402-408.
31. Wilkie,N.M., S.Ustacelebi,andJ.F.Williams.1973.
Characterization oftemperature-sensitive mutantsof
adenovirus type 5. Nucleic acid synthesis. Virology
51:499-503. VOL. 17, 1976
on November 10, 2019 by guest
http://jvi.asm.org/