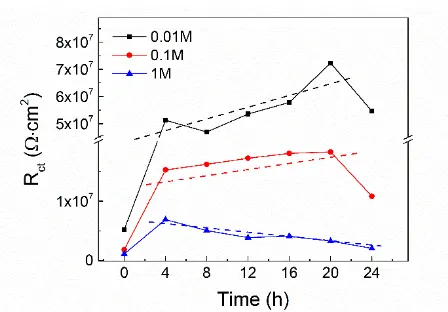
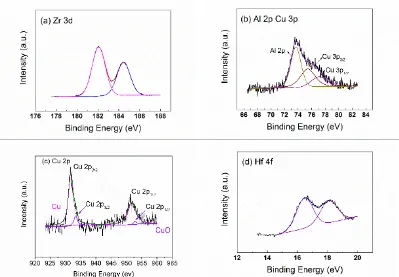
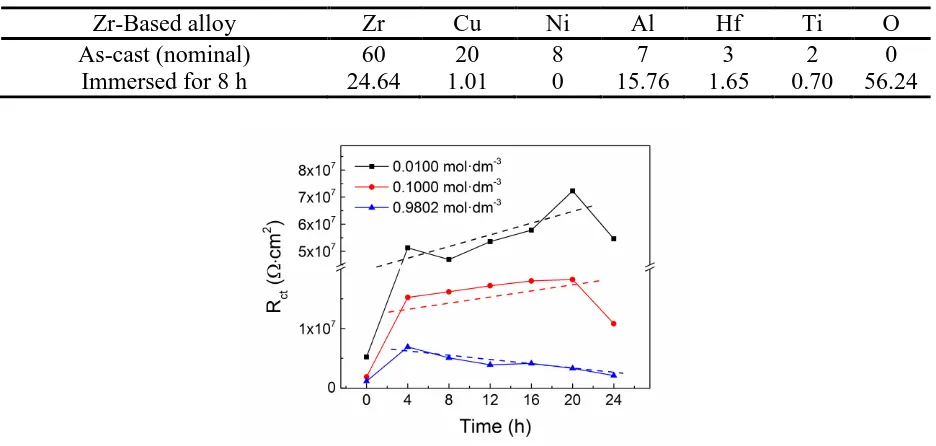
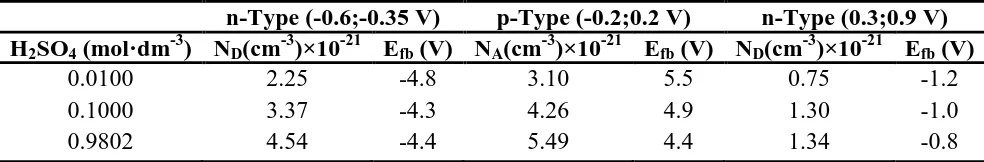
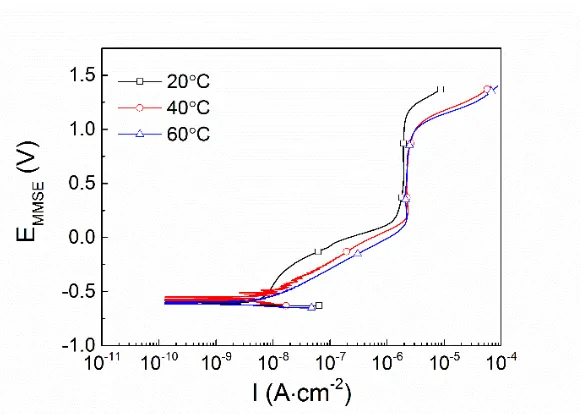
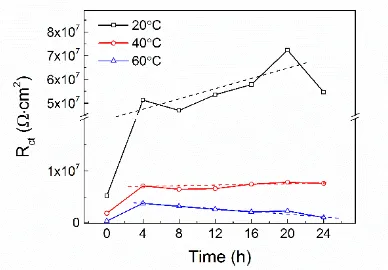
Passivation Behavior of a Zr60Cu20Ni8Al7Hf3Ti2 Bulk Metallic Glass in Sulfuric Acid Solutions
Full text
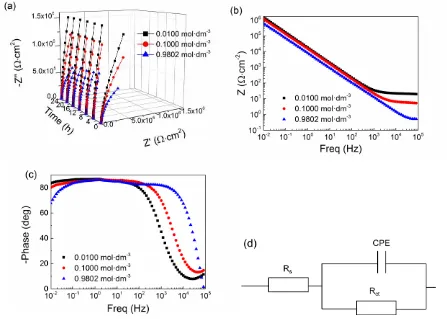
Figure
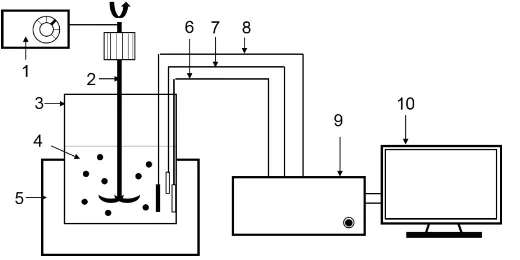
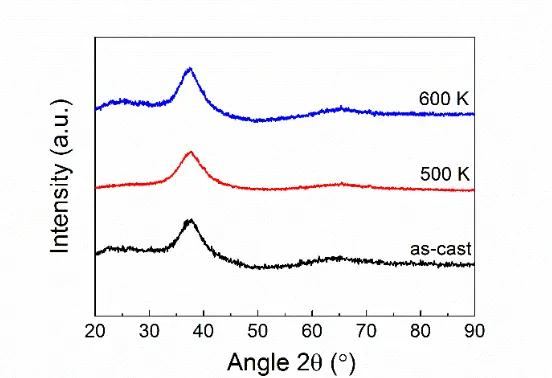
Related documents
(HiCel TM SMCC) Silicified Microcrystalline Cellulose is made by co-processed technology.HiCel TM SMCC90M grade of Silicified microcrystalline cellulose is used to manufacture the
Yin and Gan Advances in Difference Equations (2015) 2015 113 DOI 10 1186/s13662 015 0447 1 R E S E A R C H Open Access Chebyshev spectral collocation method for stochastic delay
The paper investigates the pressure variation inside a diesel engine during a complete cycle with respect to the crank angle at a constant speed, without considering
MAP: Milk Allergy in Primary Care; CMA: cow’s milk allergy; UK: United Kingdom; US: United States; EAACI: European Academy of Allergy, Asthma and Clinical Immunology; NICE:
According to Ballentine (2003) rewarding employees is to recognize excellent job performance, provide feedback, make it easier to get work done, encourage
Phosphorylated CheY (CheY-P) binds to the motor switch to induce tumbling of the cells. CheR and CheB confer adaptation to persisting stimuli by adding and removing methyl groups
the Siberian stonechats, whereas the growth rate towards adult wing length is higher in the European stonechats (note, however, that in the European birds the wing
21 Department of Neurosurgery, Tangdu Hospital, The Second Affiliated hospital of the Fourth Military Medical University, 1 Xinsi Road, Xian, Shanxi Province 710038, People ’ s