A STUDY OF SERUM HOMOCYSTEINE LEVELS DURING NORMAL PREGNANCY AND PREECLAMPSIA
Dissertation submitted to
The Tamil Nadu Dr. M.G.R Medical University, Chennai In fulfilment of the requirements for the award of the degree of
M.S. OBSTETRICS & GYNAECOLOGY
Under the guidance of
Dr. REENA ABRAHAM MD, DGO.,
DEPARTMENT OF OBSTETRICS AND GYNAECOLOGY
PSG INSTITUTE OF MEDICAL SCIENCES & RESEARCH, COIMBATORE
THE TAMILNADU DR. M.G.R MEDICAL UNIVERSITY, CHENNAI, TAMILNADU
ENDORSEMENT BY THE HOD, DEAN / HEAD OF THE
INSTITUTION
This is to certify that the dissertation entitled, “A STUDY OF SERUM
HOMOCYSTEINE LEVELS DURING NORMAL PREGNANCY AND PREECLMPSIA” is the bonafide original research work of Dr. DIVYA S under
the guidance of Dr.REENA ABRAHAM Professor, Department of Obstetrics and
Gynecology, P.S.G IMSR, Coimbatore in partial fulfillment of the requirement for
the degree of Master of Surgery in Obstetrics and Gynecology.
Seal and Signature of the HOD Seal and Signature of the Dean
Dr. SEETHA PANICKER M.D, D.G.O, Dr. RAMALINGAM,
Professor & HOD, PSG IMSR, Coimbatore,
Department of Obstetrics and Gynecology
CERTIFICATE BY THE GUIDE
This is to certify that the dissertation entitled, “A STUDY OF SERUM
HOMOCYSTEINE LEVELS DURING NORMAL PREGNANCY AND PREECLAMPSIA” is a bonafide original work of Dr.DIVYA S, in partial fulfillment of the regulations of The Tamil Nadu Dr. M.G.R Medical University
for the award of MS degree in Obstetrics and Gynecology.
Signature of the guide
Dr. REENA ABRAHAM MD, DGO.,
Professor,
Department of Obstetrics and Gynecology,
DECLARATION BY THE CANDIDATE
I hereby declare that this dissertation entitled “A STUDY OF SERUM
HOMOCYSTEINE LEVELS IN NORMAL PREGNANCY AND
PREECLAMPSIA” is a bonafide and genuine research work carried out by me
under the guidance of Dr. REENA ABRAHAM MD, DGO., Professor,
Department of Obstetrics and Gynecology, P.S.G IMSR, Coimbatore.
This dissertation is submitted to The Tamil Nadu Dr. M.G.R Medical
University in fulfillment of the University regulations for the award of DM degree
in Cardiology. This dissertation has not been submitted for award of any other
degree or diploma.
Signature of the Candidate
COPYRIGHT DECLARATION BY THE CANDIDATE
I, Dr. DIVYA S hereby declare that The Tamil Nadu Dr. M.G.R Medical University, Chennai shall have the rights to preserve, use and disseminate this
dissertation in print or electronic format for academic / research purpose.
Signature of the Candidate
CERTIFICATE – II
This is to certify that this dissertation work titled A STUDY OF SERUM
HOMOCYSTEINE LEVELS IN NORMAL PREGNANCY AND
PREECLAMPSIA of the candidate Dr. DIVYA S with Registration
Number 221416451 for the award of MASTER IN SURGERY in the branch of
OBSTETRICS & GYNAECOLOGY. I personally verified the urkund.com website for the purpose of plagiarism Check. I found that the uploaded thesis file
contains from introduction to conclusion pages and result shows 0% of plagiarism
in the dissertation.
ACKNOWLEDGEMENT
It gives me immense pleasure to express my heartfelt and profound sense of
gratitude to my respected teacher and guide, Professor Dr. Reena Abraham for
her valuable suggestions, meticulous guidance, support and encouragement in
doing this study.
I am grateful to Professor Dr. Seetha Panicker, Professor Dr.Chitra for their
invaluable help in preparing this dissertation. I am thankful to Dr. Karthikeyan,
Assistant Professor, Department of Community Medicine for helping with the data
analysis and statistics.
As he is always, the wind beneath my wings, my husband Dr.Prasanna Kumar, a
big thank you for perfecting and correcting my sentences and language and for
your constant support and encouragement.
I would also like to extend my gratitude to the entire Department of Obstetrics &
CONTENTS
SL.NO. TOPIC
PAGE NO.
1. INTRODUCTION 1
2. AIMS AND OBJECTIVES 7
3. REVIEW OF LITERATURE 8
4. MATERIALS AND METHODS 50
5. RESULTS 53
6. DISCUSSION 78
7. LIMITATIONS 87
8. CONCLUSION 88
9. REFERENCES
1
INTRODUCTION
Hypertensive disorders complicate upto 10% of all pregnancies.
Preeclampsia is one of the major causes for morbidity and mortality (1). Despite a
fairly high incidence, the etiology underlying preeclampsia is still incomplete. The
three major theories of preeclampsia are endothelial dysfunction, inflammation and
angiogenesis. Majority of the adverse outcomes can be avoided with proper
identification of high risk patients and proper management during antenatal period.
Homocysteine is a sulphur containing amino acid which is formed from
methionine that is involved in methylation and sulphuration pathways such as lipid
peroxidation and oxidative stress. Blood concentration of homocysteine is
determined by folic acid and Vitamin B12.
Hyperhomocysteinemia is associated with vascular disease. In normal
pregnancy homocysteine concentration falls(2). Alteration in maternal and fetal
homocysteine causes fetal neural tube defects and placental vasculopathy which
leads to preeclampsia, abruption and recurrent pregnancy loss. Oxidative stress and
endothelial dysfunction caused by hyperhomocysteinemia leads to preeclampsia.
In bio humoural parameters homocysteine has recently been considered a possible
cause of vascular damage. Hyperhomocysteinemia in early pregnancy may
2
Prediction, prevention, early diagnosis and treatment and providing optimal
health care to all hypertensive patients are necessary to achieve the millennium
developmental goals. Therefore a case control study was designed to examine
serum homocysteine in normal pregnancy and preeclampsia and its association
with the severity of preeclampsia
HISTORY OF HOMOCYSTEINE
Carson and Neil in 1962, for the first time suggested the association between
elevated serum homocysteine and diseases. A new and rare inborn error of
methionine - homocysteine metabolism is associated with increased urinary
excretion of homocysteine. Homocysteinuria was found in mentally retarded
children in USA and also in Ireland simultaneously. Then subsequently
cystathionine beta synthase deficiency was identified in 1964 by Mudd et al (3)in
the liver biopsy of a patient with homocysteinuria. Gibson et al(4) 2 years later
noted that the homozygous defect of the cystathionine beta synthase was associated
with increased risk of death in young children. In the same year
hyperhomocystenemia causing vascular changes and thrombosis was also
3
McCully(5) hypothesized that elevated homocysteine causes vascular changes
which lead to thrombosis and he also identified the link between homocysteine and
cardiovascular disease. He also said that hyperhomocysteinemia causes
arteriosclerosis, since it is disorder of the sulphur containing amino acid which
leads to accumulation of homocysteine and causes vascular changes.
In 1976 Wilcken and Wilcken confirmed that patients with coronary artery
disease had increased levels of cysteine-homocysteine after a methionine loading
test in comparison with the controls.
An international survey of 629 patients with cystathionine beta synthase
deficiency found that they had a 50% risk of suffering from thromboembolic
events before the age of 30 years. Large scale studies have been done on
hyperhomocysteinemia and cardiovascular disease in 1990’s.
Atherothrombotic disease has drawn the attention of scientists from many
fields. Herrmann et all in 2001 found that hyperhomocysteinemia is a risk factor
for cardiovascular diseases, pregnancy complications, neuropsychological
5
The percentage of each form of homocysteine is as follows: Reduced form of Homocysteine
Homocysteine is about 1-2%
OXIDIZED FORM OF HOMOCYSTEINE Free forms:
Homocysteine (5-10%)
Homocysteine – cysteine (5-10%)
Protein bound:
Protein –homocysteine (80-90%)
6
The non protein amino acid homocysteine is a byproduct of degradation of
methionine into non essential thiol cysteine. Methionine is a protein forming
amino acid which is obtained by two ways, mainly by food intake or remethylation
7
AIMS AND OBJECTIVES
To find any correlation between serum Homocysteine level in preeclampsia
and normal pregnancy.
To know the severity of preeclampsia, whether it is mild or severe
depending upon the elevated Homocysteine levels.
This study was done mainly to look for the presence of elevated serum
homocysteine levels in preeclampsia in our population. High prevalence of
hyperhomocysteinemia is present in our population. About 54.5% of the
population has MTHFR enzyme polymorphism.
Elevated homocysteine levels have been recently studied in pregnant
patients as it is associated with several pregnancy related complications and
8
REVIEW OF LITERATURE
HOMOCYSTEINE METABOLISM:
Homocysteine is a naturally occurring Sulphur containing amino acid.
Homocysteine metabolism stands at the intersection of two pathways:
a) Remethylation
Remethylation of homocysteineto methionine which requires vitamin B 12
and folate.
b) Transsulfuration
Transulfuration of homocysteine to cystathionine which requires vitamin B 6
REMETHYLATION PATHWAY
In this homocysteine acquires a methyl group from N5–Methyl
tetrahydrofolate (MTHFR) or from betaine to methionine. This reaction occurs in
all tissues and it is Vitamin B12 dependent, whereas the reaction with betaine is
confined mainly to the liver and it is not dependent on Vitamin B12. A proportion
of methionine is activated by ATP adenosine triphosphate to S adenosylmethionine
(SAM). SAM serves as a methyl donor to a variety of acceptors like
guanidinoacetate, nucleic acids, neurotransmitters, phospholipids and hormones. S-
9
subsequently hydrolyzed and the regenerated homocysteine now becomes
available to start a new cycle of methyl transfer group. This hydrolysis reaction is
reversible and favours the synthesis of SAH.
TRANSSULFURATION PATHWAY:
In transsulfuration pathway homocysteine condenses with serine to form
cystathionine. It is an irreversible reaction which is catalyzed by a Vitamin B6
containing enzyme, cystathionine synthase. Cystathionine is hydrolyzed by a
second vitamin B6 containing enzyme - cystathionase to form cysteine and alpha
ketobutyrate. Excess cysteine is oxidized to inorganic sulphates or taurine and is
excreted in the urine. In addition to the synthesis of cysteine, the transsulfuration
pathway effectively catabolizes the excess homocysteine which is not needed in
methyl transfer.
Homocysteine is not a dietary constituent. The sole source of homocysteine
is methionine. The primary source of methionine is from dietary protein, mainly
derived from animal proteins, however methionine is also supplied from proteins
within the body, mainly due to muscle mass turnover. Plasma normally contains a
small amount of homocysteine averaging about 5 to 15 micro mol/L, which is
10
This mechanism complements the catabolism of homocysteine through
transsulfuration pathway and altogether these mechanisms help to maintain a low
11
Hyperhomocysteinemia indicates that the homocysteine metabolism is in
some way disrupted and the export mechanism is producing excessive
homocysteine into the blood. The export mechanism to some extent limits the
intracellular toxicity, but the vascular tissue is exposed to excess homocysteine
which leads to deleterious effects.
In the first step of the Meth adenosyltransferase catalysis the transfer of
adenosine from ATP to Methionine yields a high energy compound that is S
adenosylmethionine. This methyl group can be transferred to a wide range of
12
to several metabolic processes, in particularly the methylation of DNA which is
important in the regulation of gene expression.
About 100 methyl transferases exist of which most of them are subjected to
potent feedback inhibition by the product of the methyl transfer group, S-
adenosylhomocysteine. S–adenosylhomocysteine is quickly removed by the
process of hydrolysis which forms homocysteine and adenosine. Under
physiological conditions the enzyme which is responsible is
S-adenosylhomocysteine which actually favours reverse condensation reaction. In
vivo, the reaction is driven forward to produce homocysteine by effectively
removing adenosine by adenosine kinase and adenosine deaminase.
Vitamins are important cofactors for the subsequent reactions in
homocysteine metabolism. Therefore nutritional deficiencies because of poor diet
or malabsorption can cause disturbances within the pathway. The fate of
homocysteine is governed by the availability of the methionine from the diet. If
methionine is deficient then the transfer of a methyl group from a donor compound
brings about the remethylation of homocysteine. In most of the tissues the reaction
involves 5 methyltetrahydrofolate as a donor compound and vitamin B12 acts as a
13
Methionine synthase metabolizes the methyl group from
5- methyltetrahydrofolate which channels one carbon unit derived from formate
and aminoacids such as serine, glycine and histidine into the methylation cycle,
which provides the methyl group for the synthesis of S – adenosinemethionine.
Serine and glycine hydroxymethyltransferase, methylenetetrahydrofolate
dehydrogenase and methylenetetrahydrofolate reductase serve to regenerate 5
methylenetetrahydrofolate from tetrahydrofolate.
Most of the body’s cells contains methionine synthase, however some
tissues like liver and kidneys exhibits alternate remethylation pathways which
utilizes betaine that is trimethylglycine as a methyl donor instead of 5-
methyltetrahydrofolate.
Sometime when methionine is present abundantly, homocysteine enters the
transulfuration pathway, where it undergoes a condensation reaction with serine to
form cystathionine. Vitamin B6 acts as a cofactor for the enzyme cystathionine β
synthase.
Cysteine is formed by the hydrolysis of cystathionine by γ- cystathinase, the
byproduct of which is - ketobutyrate. Cysteine is used for the synthesis of
14
Sulphur is excreted 70 % via urine by oxidation of the Sulphur atom of cysteine as
an inorganic sulphate.
S – adenosylmethionine acts upon the key enzymes . The abundance of
S-adenosylmethionine promotes the transulfuration pathway by its negative allosteric
effect on methytetrahydrofolate by blocking remethylation and a positive allosteric
effect on cystathionine β synthase, which allows the removal of excessive
methionine and homocysteine. If the S adenosylmethionine levels are low then the
allosteric regulation is removed, enters the remethylation pathway and methionine
is regenerated.
The oxidized ferric state of hemoglobin is more favourable to homocysteine
binding, possibly conformational changes in the proteins. Under oxidative stress
homocysteine is channeled into the regeneration of depleted glutathione through
transulfuration pathway.
Homocysteine metabolism is very complex which involves enzymes, B
vitamins and folate. Deficiency in the supply or functioning of any of these
15
The cellular folate cycle is shifted towards the formation of methyl
tetrahydrofolate. Utilization of the Methionine for purine and pyrimidine
biosynthesis is reduced. Methionine rich diet increases SAM levels within the
cells. SAM acts as an allosteric inhibitor for MTHFR betaine: homocysteine
methyltransferase causing aberration in the remethylation pathway.
SAM level causes an increased cellular SAH concentration which is a
strong inhibitor of the adenosylmethionine dependent methyl transferase.
Homocysteine is recycled to Meth several times and it becomes irreversibly
16 FOLATE
Folate is a water soluble vitamin B which acts as a coenzyme to accept or
donate one carbon unit needed in several metabolic pathways. The name folic
acid is derived from Latin word folium. It was first identified from spinach leaves
in 1941 and was synthesized in 1946.
The chemical name of folic acid is pteroylmonoglutamic acid. Naturally
occurring dietary folate contains pteridine ring and polyglytamate polypeptide
which is hydrolyzed in the intestinal lumen into monoglutamate. Oxidized form of
folic acid is only present in tablet form, fortified food and in multivitamin tablets.
17
The first step in the folate cycle is the conversion of tetrahydrofolate to
5,10-methylene-THF using serine as a source of carbon units and Vit B6 dependent
serine hydroxy methyl transferase enzymes. The cell proliferation and DNA
synthesis requires transfer of one carbon atom, this is the main fundamental
function of folic acid. Carbon atoms are passed on for the synthesis of purines and
pyrimidines. Hence, folate deficiency leads to megaloblastic anemia,
reticulocytopenia, thrombocytopenia, and leukopenia, all of which are reversible
after folic acid supplementation and reintroduction into diet. Biochemical status to
assess the folate status are serum and RBC folate levels.
Serum folate is a circulating folate, which can change quickly and is
influenced markedly by diet. RBC folic acid status is intracellular, which gets
accumulated during erythropoiesis and it is retained throughout RBC life span.
RBC folate is the most representative of body stores of folate, in the absence of
pernicious anemia.
The main regulator of homocysteine degradation is folate cycle.
Methyltetrahydrofolate is the only circulating form of folate which is used for the
premethylation of homocysteine to Methionine. Folate, Vit B12 and Methionine
synthase work together within the cell. Fasting plasma homocysteine is increased
18
Homocysteine level is usually normal in Vit B6 deficient subjects. The
genetic defect of Methionine synthase and Vit B12 deficiency leads to increased
level of Methyltetrahydrofolate. The consequences are abnormal intracellular
folate level in the presence of normal or increased circulating folate levels. The
blot THF regeneration leads to reduced thymidylate synthesis causing
megaloblastic anemia.
HYPERHOMOCYSTEINEMIA
Abnormal elevation of homocysteine levels, above 90thor 95th percentile of
the corresponding reference group is known as hyperhomocysteinemia. Normal
range of homocysteine is 5-12micro mol/l. In antenatal patients generally
homocysteine levels fall due to hemodilution and also folic acid intake by patients.
In pregnancy 5-7 micro mol/l is considered as normal. Anything above this level is
considered as hyperhomocysteinemia in pregnancy.
In non – pregnant population:
Moderate hyperhomocysteinemia is defined as homocysteine concentration
levels between 12- 30 micro mol/l. Seen in mild folate and Vit B12
19
Intermediate hyperhomocysteinemia is defined as serum homocysteine
concentration between 30-100 micro mol/l. Seen in moderate to severe folate
and Vit B12 deficiency and also with renal failure.
Severe hyperhomocysteinemia is defined as serum homocysteine
concentration more than 100 micro mol/l. Seen in severe Vit B12 deficiency.
In normal pregnancy homocysteine levels are lower when compared to the
non pregnant state. Thus these values cannot be applied to pregnant women which
becomes misleading. Till date no value is available as a cut-off value of
homocysteine in pregnant women. Homocysteine value less than 5-7 micro mol/l
in normotensive pregnant women who had adequate folate and Vit B12 was
20
HYPERHOMOCYSTENEMIA CAUSED BY FOLIC ACID DEFICENCY
Folic acid deficiency causes decreased synthesis of methionine, which leads
to the metabolism of homocysteine to transulfuration pathway. The decreased
synthesis of methionine causes decreased synthesis of SAM concentration. N 5
tetrahydofolate deficiency causes N – Methyltranferase to be fully active, which
further decreases the SAM level and increases the synthesis of serum
homocysteine which is a byproduct of glycine methylation.
So the transulfuration pathway becomes ineffective due to increased
homocysteine concentration in addition to decreased level of SAM, which is too
low to activate cystathionine synthesis. As a result homocysteine accumulates in
the blood which leads to hyperhomocystenemia.
FACTORS INFLUENCING HYPERHOMOCYSTENIEMIA
The regulation of Methionine cycle and Homocysteine pathway is associated
with the availability of folate, Vit B6 and Vit B12. Folate donates its methyl for
remethylation of homocysteine.
There are many other factors influencing Homocysteine which is classified as:
Physiological determinants: sex, race, age and pregnancy.
21
Genetic factors: methionine synthase reductase enzyme, CBS enzyme,
MTHR enzyme.
Drugs : hormones, antiepileptic drugs, lipid lowering drugs
AGE AND GENDER:
Advanced age and male gender are found to be associated with
hyperhomocysteinemia. This may be due to difference in the status of vitamins
between different age groups and gender. Homocysteine levels may also be altered
and may be related to muscle mass, hormonal factors and renal function.
Homocysteine levels were found to be high in women who are post menopausal
when compared to perimenopausal women. In pregnant women homocysteine was
found to be lower than the non pregnant women.
DIETARY FACTORS
Several studies have provided evidence of the importance of vitamin B in
homocysteine metabolism. A metaanalysis of 12 RCT’s showed that folic acid
supplementation had decreased the homocysteine levels in serum by 25%, with the
dosage of 0.5 to 5 mg. However higher pretreatment of homocysteine showed
reduced level of homocysteine in response to folic acid treatment. Vitamin B6 and
B12 are not utilized in homocysteine metabolism, but they function as a cofactors
22 LIFESTYLE FACTORS
In a cohort study conducted by Caerphilly et al showed that smokers had
significantly elevated homocysteine level than non smokers. Following cessation
of smoking, homocysteine value returns to normal.
Smoking itself can affect the metabolism of Vitamin B, which leads to
abnormal homocysteine metabolism. Generally smokers consumes more of an
unhealthy diet than non smokers.
Chronic alcoholism causes hyperhomocysteinemia due to low levels of
vitamin B and it is also influenced by the irregularity of the feeding patterns. It is
believed that acetaldehyde which is a metabolite of ethanol forms a covalent bond
with the proteins that inhibit MS activity.
Folate deficiency also causes hyperhomocystenemia in alcoholics. Mild
alcohol consumption reduces the serum homocysteine levels as observed by
Hordaland cohort.
Caffeine intake causes hyperhomocysteinemia. Caffeine interferes with
23 PREGNANCY
Homocysteine level decreases during pregnancy which is independent of
folate status, and it becomes normal within 3 to 4 days of the postnatal period.
Reason for this low level of homocysteine is not clear. Studies say that it may be
an adaptation to maintain the placental circulation. Fetus is also is involved in the
uptake of maternal homocysteine.
DISEASES
The basis of hyperhomocysteinemia in renal failure is not clearly
understood, although several processes explain the close correlation between
hyperhomocysteinemia and renal function.
The most common cause of hyperhomocysteinemia other than nutritional
deficiency of vitamin B12 and folate is renal failure. Cancer is associated with
higher levels of homocysteine. Type II DM who are on metformin, can have mild
hyperhomocysteinemia. Gastro intestinal disorders like ulcerative colitis, Crohns
disease and IBS leads to malabsorption of vitamin B12 and folate, which in turn
24 MEDICATIONS:
Sex steroid hormones have influence on homocysteine levels. It was found
that tamoxifen which is an anti oestrogen was found to decrease the serum
homocysteine concentration.
Drugs like methotrexate which inhibits the conversion of dihydrofolate to
tetrahydrofolate decreases the synthesis of DNA, RNA nucleotides. Since
methotrexate inhibits cell proliferation, it is used in the treatment of cancer,
psoriasis and rheumatoid arthritis.
Phenytoin causes folic acid deficiency. Drugs like carbamazepine,
pyrimidone and sodium valproate also interfere with remethylation of
homocysteine.
Metformin and theophylline decreases the B vitamin concentration which
increases the serum homocysteine concentration. The drugs which acts as B6
antagonist are isoniazid, cyloserine, procarbazine, hydralazine and phenelzine also
causes increase in serum homocysteine levels.
Drugs which have a free thiol group like N – acetylcysteine and D
penicillaminecan be used to decrease serum homocysteine concentration in patients
25 GENETIC FACTORS:
Cystathionine beta synthase:
Mutation in this enzyme leads to homocysteinuria which was published by
Gaustadnes et al in 1998. It is an autosomal recessive disease, in which it was
found that there was elevation of serum homocysteine and also excretion of
homocysteine in urine. There is luxation of optic lenses due to disruption of the
zonularfibres, seen in children who have homocystinuria and who are aged
between 2 to 10 years. It may also be associated with mental retardation,
psychiatric illness, skeletal abnormalities and osteoporosis.
Methyltetrahydrofolatereductase (MTHFR):
These are rare inborn errors of metabolism. Mutation of
methyltetrahydrofolate causes weak effect on the enzymes. The prevalence of
these mutations are high in general population. MTHFR catalyzes the formation of
5-methyl THF from 5,10methylene tetrahydrofolate. The folate from the above
reaction is necessary for the remethylation of homocysteine to methionine. Higher
homocysteine levels are found in subjects with marginal folate status. MTHR itself
is not the reason for venous thrombosis but it is reported that it decreases the bone
26
PATHOGENESIS IN HOMOCYSTEINE INDUCED THROMBOSIS
Hyperhomocysteinemia causes both arterial and venous thrombosis, which
causes stroke, myocardial infarction, pulmonary embolism and retinal vein
thrombosis. Hyperhomocysteinemia also causes other diseases like preeclampsia,
recurrent pregnancy loss, IUGR, preterm delivery, abruptio placenta and it also
associated with neural tube defects.
Russel Rose proposed a hypothesis on hyperhomocysteinemia causing
endothelial dysfunction, which alters the normal homeostatic response of the
endothelium. Hence endothelial injury causes adhesion of leukocytes and platelets
and also an increase in the vascular permeability. This causes the endothelium to
have procoagulant property.
Due to inflammation cytokines and other inflammatory factors there is
further damage and focal necrosis. All these changes leads to proliferation of
smooth muscle and formation of fibrous tissue which prevents further dilatation of
27
The influence of hyperhomocysteinemia on endothelial function:
The effect of homocysteine is the reduction in the endothelial function.
Endothelial cells secrete several agents involved in vasoconstriction and
vasodilatation. Endothelium derived vasoconstrictors are thromboxane A2,
prostaglandins H2 and endothelin 1. The endothelium derived vasodilators are
nitric oxide and prostacyclin. Homocysteine mediates the endothelial dysfunction.
Homocysteine reduces the bioactivity of nitric oxide; the reaction of nitric
oxide with superoxide produces peroxynitrite which is a potent oxidant which
causes activation of polymerase, which is a mediator of vascular dysfunction in
disease. Superoxide can oxidize tetrahydrobiopterin which is a co factor for Nitric
Oxide synthase leading to reduced activity of eNOS where the electrons are
transported to molecular oxygen forming O2 rather than to L arginine forming
Nitric Oxide. Homocysteine inhibits the activity of eNOS by increasing the levels
of asymmetric dimethylarginine, which is an inhibitor of NO synthase which leads
to reduce the bioavailability of NO.
Homocysteine increases the oxidative stress and increase the levels of
reactive oxygen species. Elevated Homocysteine levels inhibit the expression and
function of antioxidant enzymes such as extracellular superoxide dismutase by
28
Homocysteine increases the vascular sources of O2 including xanthine oxidase,
cyclooxygenase, nitric oxide synthase.
Homocysteine can upregulate the components of the inflammatory cascade.
Homocysteine activates nuclear factor B, which causes overexpression of
cytokines leading to inhibition of vasoconstriction, thereby leading to impairment
of endothelial function. TNF alpha increases the activity of NADPH oxidase
causing consequently increased superoxidase levels in Hyperhomocysteinemia.
The influence of homocysteine on smooth muscle and extracellular matrix:
Homocysteine causes proliferation of smooth muscles, which is inhibited by
folic acid. Homocysteine causes accumulation of collagen. When vascular smooth
muscle is exposed to homocysteine, cytokine stimulation occurs which leads to
production of NO and leads to early atherosclerosis. Hyperhomocysteinemia
causes thrombosis which explains homocysteine associated illness which was
already mentioned.
The influence of homocysteine on the coagulation system:
Homocysteine induces the activity of TF in the endothelial cells and also in
the monocytes and peritoneal macrophages, which triggers the coagulation system.
Homocysteine increases the platelet activation and aggregation. Stimulation of
29
homocysteine indirectly inhibits the Nitric oxide production. Nitric oxide
inhibition can also be due to endogeneous inhibition of nitric oxide synthase and
asymmetric dimethylarginine.
Undas et al suggested the impaired inactivation of factor Va by activated
protein C due to homocysteinylation of the cofactor by modification of free
cysteine. Factor VII a and thrombin activities are increased in patients with
coronary artery disease, but Bos et al suggested that increased risk of venous
thrombosis in hyperhomocysteineima is not reflected by an increased endogenous
thrombin potential.
Impaired fibrinolysis is an important mechanism for thrombosis
predisposition in hyperhomocysteinemia. Homocysteine possesses anti fibrinolytic
properties, which enhances the binding of lipoprotein to fibrin and causes
inhibition of tissue plasminogen activator to the endothelial cell through annexin
II. Hyperhomocysteinemia favours thrombogenesis. To prove this clinical studies
have been performed in hyperhomocysteinemic patients. Some studies provide
30
HOMOCYSTEINE IN NORMAL PREGNANCY AND PREGNANCY COMPLICATED WITH PREECLAMPSIA:
Preeclampsia is the most common cause of maternal morbidity and mortality
in developing countries.
Preeclampsia is a two stage disease:
STAGE 1: It is characterized by the reduction in the placental perfusion.
STAGE 2: It denotes the maternal syndrome in which hypertension is accompanied with proteinuria. Oxidative stress is the most important factor for the
progression of the disease. Along with other maternal factors like age, twin
pregnancies, nulliparity, oxidative stress and endothelial dysfunction is the main
pathogenesis of preeclampsia. The etiology of preeclampsia is still not clearly
understood.
The genetic makeup and acquired factors are reasons for the disease
occurrence and progression. The nutritional demands are high in pregnancy, hence
it is associated with higher vitamin B requirements for both the mother and the
growing fetus.
Vitamin B includes folate, Vitamin B12 and Vitamin B6 which acts as a
31
division. Serum concentration of these vitamins decreases during pregnancy. The
low concentration of these vitamins is due to higher metabolic rate and increased
active transport of the vitamins to the fetus through the placenta.
Maternal concentration of these vitamins even from the preconceptional
period affects the serum concentration of these infants at birth. A study conducted
by Casterline and et al, measured the vitamin levels from the breast milk in women
who are lactating and found that 31% of mothers had low vitamin B12 and 61 % of
32
Maternal nutritional status influences the outcome of pregnancy. Decreased
folate levels in the mother is associated with preterm delivery, low birth weight
and neural tube defects. Vitamin B12 deficiency is associated with recurrent
abortion, neural tube defects and megaloblastic anemia.
Therefore folic acid supplementation pre conceptionally reduces the chances
of neural tube defects by 30 %. In addition to this, folic acid supplementation also
reduces the incidence of preeclampsia and gestational hypertension.
Elevated serum homocysteine levels indirectly denotes the deficiency of B
vitamins. Maternal hyperhomocysteinemia affects both the mother and the fetus
(Vollestet all 2000).
A study was conducted by Murphy et al(8), where they included 93 women
and their off springs, found that fetal Homocysteine concentration and birth weight
significantly correlated to maternal Homocysteine from preconceptional period to
pregnancy.
A mother with high serum Homocysteine levels at 8 weeks had 3 fold risk of
delivering low birth weight babies. Several other studies were done concerning the
association between maternal hyperhomocysteinemia with adverse maternal and
33
Serum homocysteine levels fall during pregnancy at around 8-10 weeks.
Homocysteine levels remains lower in the third trimester, less than that of the
preconceptional period. Lowest value is found in the second trimester (Andersson
et al., Kang et al).
Several mechanism have been proposed the reason of decreased homocysteine level in pregnancy,
Increased glomerular filtration rate
Increased plasma volume in pregnancy
Hemodilution
Increased transfer of B vitamins to the fetus
Hormonal effect on homocysteine during pregnancy
The exact mechanism is still not clear. Lower levels of Homocysteine acts as
a protection to the mother and fetus from the hyperhomocysteinemia associated
with pregnancy complications. Several studies were done which initiated and
addressed homocysteine as a biomarker which has predictive value in early
trimester at around 8- 10 weeks for identifying women who are at risk of
subsequently develop preeclampsia in the later trimester (Anna et al., Heitala et al
34
Hyperhomocysteinemia causes endothelial dysfunction which is one of the
major complications that is closely related to preeclampsia (Geseil et al., Herrmann
and Knapp et al., Stanger et al., 2001)(10)
In preeclampsia the homocysteine concentration is elevated throughout
pregnancy during all 3 trimesters and postpartum period. Women with previous h/o
preeclampsia also have elevated levels of serum homocysteine. The reason is still
unclear.
The other mechanisms proposed are:
Renal insufficiency(11)( a study done by Brattstrom L et all in 2003)
Decreased reformation of methionine from Homocysteine (Malinow et al in
1998)
Decrease in remethylation (Powers et al in 2004)
Disturbance in liver metabolism of Homocysteine
Reduced Vitamin B during preeclampsia
Folate was measured in only some studies. A study was conducted by
Powers et all, found that low levels of folate were found to have increased risk of
preeclampsia. Two ongoing studies on low Vitamin B12 and preeclampsia found
35
study conducted in Australian women by Kaiser et al demonstrated that MTHFR
mutation is not a risk factor for preeclampsia, if prenatal folate is substituted.
THE PATHOGENESIS OF PREECLAMPSIA:
It important to know the pathogenesis of preeclampsia which is similar to
that of elevated levels of homocysteine causing endothelial dysfunction.
In normal pregnancies, a subset of cytotrophoblasts called invasive
cytotrophoblasts migrate and invade the decidua tunica media of maternal spiral
arterioles and replace its endothelium with the subsequent destruction of the
medial, elastic, muscular and neural tissue. This process is known as pseudo
vascularization. By the end of the second trimester of pregnancy, the uterine spiral
arteries are lined exclusively by cytotrophoblast, and endothelial cells are no
longer present in the myometrial regions.
As a result of these changes, the maternal spiral arterioles undergo
transformation from small, muscular arterioles to large capacitance, low resistance
vessels. This allows increased blood flow to the maternal and fetal interface.
Remodelling of these arterioles begins in the first trimester and ends by 18-20
36
The early stage of pregnancy: Epithelial – endothelial transformation
During normal differentiation, invading cytotrophoblasts alter their adhesion
molecule expression from epithelial cells like integrins alpha 6/beta 1, alpha V/beta
5, & E-cadherin to those of endothelial cells like integrins alpha 1/beta 1, alpha V/
beta 3 and VE- cadherin.
FAILURE OF PSEUDOVASCULARISATION IN PREECLAMPSIA
Invasion of the decidual arterioles by cytotrophoblasts is incomplete and
limited to the proximal decidua. Shallow placentation is noted which leads to
reduction in uteroplacental perfusion.
This is due to a failure in the alteration in molecular expression necessary for
the differentiation of cytotrophoblasts required for pseudovascularization.
The up regulation of matrix metalloproteinase – 9 (MMP-9) and HLA-G
does not occur. Causes for the failure may be immunological and genetic factors.
Early hypoxic insult to the differentiating cytotrophoblast has also been
37 ABNORMAL PLACENTATION:
On the basis of the observation, that the only definitive cure for
preeclampsia is delivery of the placenta which plays a central role in the
pathogenesis, is supported by epidemiologic and experimental data.
Abnormal development of the placenta
Abnormal remodelling of spiral arteries
Defective trophoblastic differentiation
38 IMMUNOLOGICAL FACTORS
Dysregulation of maternal tolerance to paternally derived placental and fetal
antigens has been considered. This maternal-fetal immunomaladaptations is
characterized by defective co-operation between uterine natural killer (NK)cells
and fetal human leukocyte antigen (HLA) – C and the result is changes similar to
those seen in acute graft rejection.
Excessive production of immune cells causes secretion of tumour necrosis
factor alpha which causes apoptosis of the extravillous cytotrophoblasts. Women
with preeclampsia show decreased levels of HLA – G and HLA – E. The
endothelial cell dysfunction is due to an extreme activation of leukocytes in the
maternal circulation.
SYSTEMIC ENDOTHELIAL DYSFUNCTION
The circulating proangiogenic factors secreted by the placenta include
vascular endothelial growth factor (VEGF) and placental growth factor (PGF). The
antiangiogenic factors include soluble fms- like tyrosine kinase I receptor (sflt-1)
(otherwise known as soluble VEGF receptor type I).
Data show that an imbalance of proangiogenic and antiangiogenic factors
produced by the placenta may play a major role in mediating endothelial
39
Maynard et al observed that the serum levels of VEGF & PIGF were
decreased in women with preeclampsia. The receptor sFlt-1 is a receptor for
binding circulating VEGF and PIGF through its ligand binding region. Thus SFlt-1
has an antiangiogenic effect.
Excess production of s Flt – 1 is associated with an increased risk of
preeclampsia and early onset preeclampsia. Endoglin binds to circulating TGF-beta
and decreases the circulating levels. TGF-beta is a pro angiogenic molecule.
Risk factors include personal or family history of preeclampsia, primiparity,
new paternity, maternal age more than 40, higher BMI, Multiple pregnancy,
hyper-placentosis, interval between pregnancies either <2 years (or) >10 years, pre-
existing medical disorders like chronic kidney disease, chronic hypertension, and
homozygosity / heterozygosity.
Also, Kidney donors and women with sub clinical hypothyroidism and
antithyroid antibodies are at a higher risk of developing preeclampsia. It does not
40
CLASSIFICATION OF HYPERTENSION IN PREGNANCY
Chronic Hypertension: Hypertension present before pregnancy or before 20 weeks of gestation or which is diagnosed for the first time during
pregnancy and also does not resolve by 12 weeks postpartum.
Gestational Hypertension: Hypertension after 20 weeks of gestation.
Transient Hypertension: hypertension diagnosed after 20 weeks of pregnancy and resolves by 12 weeks postpartum.
Preeclampsia superimposed upon chronic hypertension occurs in a woman with
pre-existing hypertension.
PREECLAMPSIA CLASSIFICATION:
Mild preeclampsia is defined as the presence of hypertension (Systolic BP ≥ 140-159 and Diastolic BP ≥ 90 - 109 mm Hg) on 2 occasions, at least 6 hours apart, but
without any evidence of end-organ damage in a woman who was normotensive
before 20 weeks of gestation.
In a patient with pre existing essential hypertension, preeclampsia is
diagnosed if Systolic BP has increased by 30 mmHg or if Diastolic BP has
41
Severe preeclampsia is defined as if the BP is more than 160/110 on 2 occasions at least 6 hours apart in a pregnant woman who is on bed rest
Non severe SeverePE
Blood pressure >140/90 mmHg > 160/110 mmHg
Proteinuria >0.3 gm/24 hrs
Or dipstick > 1+
>5gms/ 24 hrs
Or dipstick > 3+
Serum creatinine normal Elevated
Pulmonary edema - +
Oliguria - +
IUGR - +
Headache - +
Visual disturbance - +
Epigastric pain - +
HELLP syndrome - +
Revisiting the role of First trimester as an Index of Maternal and Fetal
outcome was a study conducted by Mariano Mascarenhas et al. It was a cohort
study conducted between 18 to 12 weeks out comprising 100 antenatal women. In
this study serum homocysteine was significantly elevated in patients who had
previous history of PIH, IUGR, oligohydramnios and pregnancy loss. It was
42
A case control study was conducted in department of Obstetrics and
Gynaecology in in Ghaem hospitals, Iran in 2006 about Elevated plasma total
homocysteine in preeclampsia by Malihe Hasanzadehet al(15). The study group was
included from 28 to 40 weeks of gestation. It consists of 40 women in control
group, 37 women with mild pre eclampsia and 38 in severe preeclampsia.
Homocysteine was done in all patients. There was no difference between serum
homocysteine level in mild pre eclampsia and normal antenatal women but it was
significantly higher in severe preeclampsia patients.
A study conducted in Uttarakhand on “Hyperhomocysteinemia in pre
eclampsia: is routine screening rational?” By Sonia Miglaniet all from the
Department of Obstetrics and Gynaecology, Himalayan Institute Of Medical
Sciences in November 2015. It was also a case control study including 30
preeclamptic women and 30 normal women. p value was statistically significant
but mean value of serum homocysteine did not correlate with the severity of
preeclampsia. They concluded that maternal serum homocysteine level has a casual
role in pathogenesis of preeclampsia but to recommend it as a routine test, larger
studies were required.
A study on Hyperhomocysteinemiain preeclampsia was found to be
associated to higher risk pressure profiles by R.NOTO et all conducted in Italy at
43
weeks of pregnancy. Homocysteine levels in normal pregnancy was very low 5+/-
1.7 and significantly higher level was present in the PIH group which was
statistically significant. Serum homocysteine level was decreased in pregnant
women when compared to non pregnant women of the same gestational age. It is
proved that in the complex biochemical mechanism, high homocysteine levels is
due to lack of precursors like folate and B12. Genetic alteration is found in N5 N10
MTHFR enzyme associated with high levels of homocysteine as early as 15 weeks.
The results of the study shows that pregnant women developing preeclampsia
showed hyperhomocysteinemia already at 20- 24 weeks. Probably high levels of
homocysteine may be considered a marker for vascular damage and may lead to
Preeclampsia.
Homocysteine in pregnancies complicated by pre eclampsia with and
without IUGR; A comparison with normotensive pregnant women with isolated
IUGR and healthy pregnant women’. This study was conducted by Marzena
Laskowsha and et all. Chair and department of Obstetrics and Perintology,
Medical University of Lubin, Poland.
One of the common maternal conditions associated with IUGR is
maternal hypertension and especially the highest level of homocysteine is seen in
both the groups complicated by preeclampsia with or without IUGR.
44
causes alteration in the coagulation system and enhances platelet activation.
Hyperhomocysteinemia is also present in normotensive pregnant patient with
IUGR.
Vitamin B6, B12, and folate play central role in metabolism of
homocysteine. If there is no B vitamins in adequate amount for the metablolic
changes there is natural decrease of homocysteine may not occur and causes
hyperhomocystenemia.
Hyperhomocystenemia due to MTHFR mutation can cause
hyperhomocystenemia which can be corrected by administration of folate. A study
conducted by Markedos et al, showed that maternal levels of vitamin B12 and folic
acid are not significantly different in preeclampsia patients who have
hyperhomocystenemia when compared to normal antenatal patients . So
hyperhomocystenemia in pregnancy might not always be due to B vitamin or folic
acid deficiency. In this study, all women were supplemented with B vitamins and
folic acid.
On contrary several studies says that homocysteine levels were significantly
elevated in severe preeclampsia when compared to mild preeclampsia.
Mao et al observed elevated serum homocysteine levels in patients with
45
levels and asymmetric dimetylarginine (ADMA). They concluded that the
homocysteine - ADMA-NO pathway may be reason for the pathogenesis in
preeclampsia and may be used as a marker to assess the severity of the disease.
Lopez – Quesada et al, also concluded that the homocysteine level was
significantly higher in preeclampsia. Similar study was conducted by Wang et all
and concluded the same.
A study was conducted by Shilpa A.V et al about changes in the
homocysteine levels in normal pregnancy and preeclampsia and its relation to
oxidative stress. They studied hyperhomocysteinemia causing oxidative damage by
release of Malondialdehyde (MDA), which is a metabolite in lipid peroxidation
found to be elevated in hyperhomocyteinemia. The mean value of MDA
352.13±52.54 which was statistically significant P value was <0.001 in
preeclampsia when compared to normal pregnancy where mean value of MDA
208.67±43.54. This may be due to reaction between the maternal dyslipidemia and
diseased placenta causing oxidative damage in the pathogenesis of preeclampsia.
The mean homocysteine value also was significant in preeclampsia group, P
value < 0.001. They concluded that B vitamin supplementations, folic acid, nitric
46
stress and restores the endothelial damage. Similar study was also reported by
Uzen et al and concluded the same.
In this study homocysteine was elevated in the preclampsia group but
statistically was not significant to support the fact to use it as a predictor test which
was supported by Laxmi Maru et al., who studied that homocysteine can be used as
a predictive marker for PIH and it was compared with the normotensive patient
along with the complications. A higher level of homocysteine was associated with
many maternal complications like abruptio placenta, retinopathy, eclampsia,
MODS. Homocysteine to be used as a predictor is under study. More large scale
studies are need.
TREATMENT OF HYPERHOMOCYSTENEMIA:
Elevated homocysteine levels is associated with number diseases which was
already explained. In case of homocystienuria methionine restriction diet is given,
which produces cysteine from methionine through homocysteine, after which
cysteine becomes the essential amino acid.
In patients with Cystathionine beta synthase (CBS) deficiency, which is
heterogeneous disease, vitamin B 6 is supplemented, which improves the function
47
patients this treatment may not respond, in that case folic acid and vitamin B12 is
supplemented to induce MS activity which reduces the serum homocysteine level.
In patients who don’t respond to vitamin B6 have higher mortality rate in
their early years. A study of homocystinuria was conducted, which showed that
patients before 30 years, 23% did not respond to vitamin B6 and only 4 %
responded to vitamin B6, 50% of the untreated cases were expected to have
thromboembolic events. Non responsive cases showed higher rate of mental
retardation and low IQ when compared with the responsive group when treated
early.
Mammals cannot produce folate, therefore folate is an essential nutrient,
which is available in fruits and vegetables, especially green leafy vegetables.
Improved diet and vitamin supplementation can reduce the homocysteine levels by
activating the remethylation pathway.
Additional supplementation of 1 mg of vitamin B12 reduces the unopposed
folic acid administration in patients with vitamin B12 deficiency and also in
patients with intrinsic factor deficiency and malabsorption status.
A study was conducted by Esraa et al, about treatment of
hyperhomocysteinemia and pregnancy outcome. In this study the patients with
48
patients with recurrent pregnancy loss in around 18-20 weeks of gestation, these
patients were treated with folic acid 5mg and vitamin B12 40 mg daily and was
followed throughout pregnancy. There was significant lowering of homocysteine
levels after treatment P value was < 0.0001 and better improvement in outcomes.
MTHFR gene mutation or folate deficiency is associated with major 3rd
trimester complications like preeclampsia, recurrent pregnancy loss, abruptio
placenta and IUGR. Due involvement of hyperhomocysteinemia in the production
of thrombotic events anticoagulant treatment is also recommend throughout
pregnancy. In case of unfractionated heparin patients PT INR/APTT levels are
monitored.
CURRENT POSITION:
Although it is proved that folate, Vitamin B supplementation decreases the
homocysteine levels, the true effect on reducing the disease remains to be
determined.
From the above studies it is understood that treatment will at least stop the
progression of the disease and to some extent it is possible to reverse the damage
of the disease that has been already done by regeneration of the tissues and
49
Lowering homocysteine levels improves cardiovascular function and
restenosis following vessel stenosis.
Folic acid supplementation during pregnancy found that there is a decrease
in the neural tube defects but prevention of preeclampsia is not clearly explained,
need more further large group of studies.
Hyperhomocysteinemia is a risk factor for several diseases. Homocysteine
level is influenced by several factors either acquired and also lifestyle factors.
Treatment in reducing homocysteine levels is cheap and safe. Therefore it is
important to continue studies this field so that we may be able to establish the
mechanisms by which hyperhomocystenemia causes disease and the potential
50
MATERIALS AND METHODS
The study was conducted in the department of Obstetrics and Gynaecology
at PSG Institute Medical Sciences and Research from June 2016 to June 2017.
The study period was 12 months.
STUDY DESIGN
Prospective observational study
STUDY POPULATION
Patients were selected between 28- 40 weeks. Total study group included 60
subjects out of which 30 patients were admitted in labour ward with preeclampsia
was chosen as cases and 30 patients was chosen as controls who was admitted for
safe confinement towards term in labour ward. Sample was collected
simultaneously in both cases and controls.
INCLUSION CRITERIA
Antenatal women, either primigravida or multigravida with 28 - 40 weeks of
gestational age.
CASES: 30 women with preeclampsia
51
Cases were selected as and when they presented. Preeclampsia was
diagnosed if BP > 140/90 mmHg on 2 occasions measuring 6 hours apart in sitting
or semi recumbent position associated with proteinuria 300 mg or more in 24 hours
urine sample or more than 1+ in dipstick in random urine samples.
With detailed history taking and routine antenatal investigations along with
PIH investigations like BT, CT, APTT, PT, INR, fibrinogen, urine albumin, urine
PCR and uric acid. Severity of preeclampsia was diagnosed depending upon the
BP reading > 160/110 mmHg and the presence of proteinuria more than 5 gms
with imminent symptoms. Serum homocysteine was done in all patients.
EXCLUSION CRITERIA:
Chronic hypertension
Diabetes mellitus
APLA syndrome
Preterm labour
Twins
Smokers
52 METHODOLOGY
5 ml of venous blood is drawn from the antecubital vein , in cases along with
other PIH investigations and in controls , when the blood was drawn for
other routine antenatal investigations.
Collected in the EDTA tube
Send to biochemistry Lab within 30 minutes
53
[image:62.612.67.537.201.419.2]RESULTS AND ANALYSIS
TABLE-1:
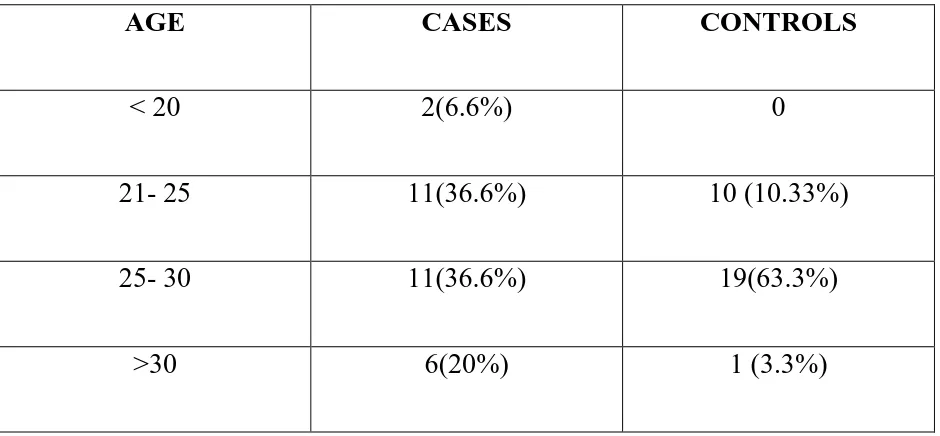
AGE DISTRIBUTION
AGE CASES CONTROLS
< 20 2(6.6%) 0
21- 25 11(36.6%) 10 (10.33%)
25- 30 11(36.6%) 19(63.3%)
>30 6(20%) 1 (3.3%)
In my study, total number patients were 60 out of which 30 patients were
controls and 30 patients were cases.
In cases, 2 patients were less than 20 years, 11 patients between 21 to 25
years, 11 patients between 25-30 years and 6 patients above 30 years.
In controls, 10 patients between 21-25 years, 19 patients between 25-30
54 TABLE-2:
THE MEAN AGE DISTRIBUTION OF THE STUDY
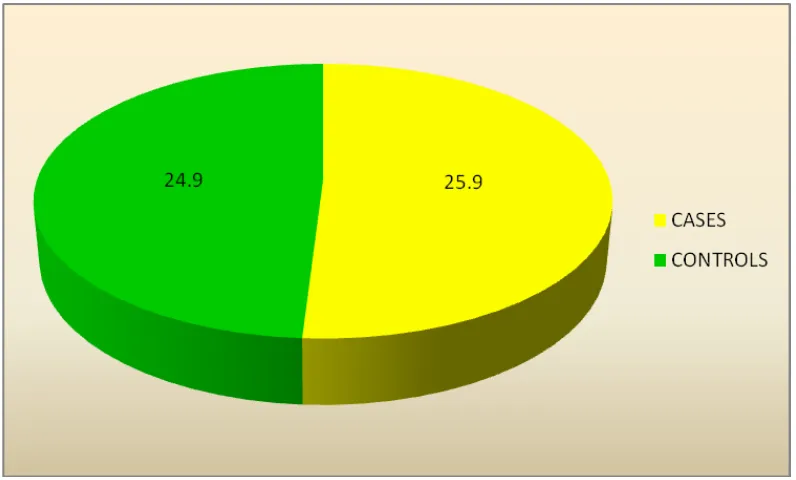
S NO N Mean SD
1 CASES 30 25.90 5.274
2 CONTROLS 30 24.90 2.480
The mean Age distribution in cases is 25.90±5.274, which is similar to that
55 TABLE-3:
OBSTETRIC SCORE:
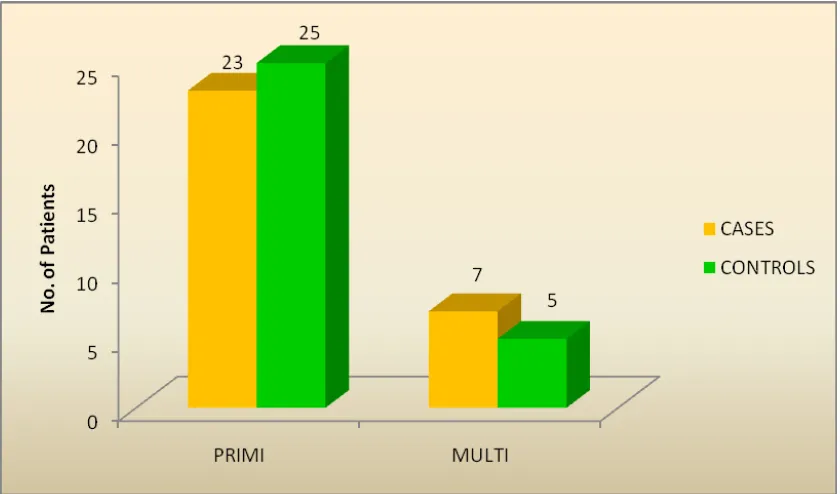
SNO PRIMI MULTI
1 CASES 23 7
76.7% 23.3%
2 CONTROLS 25 5
83.3% 16.7%
In cases 23(76.7%) are primi and 7(23.3%) multi. In controls 25(83.3%) are
56 TABLE-4:
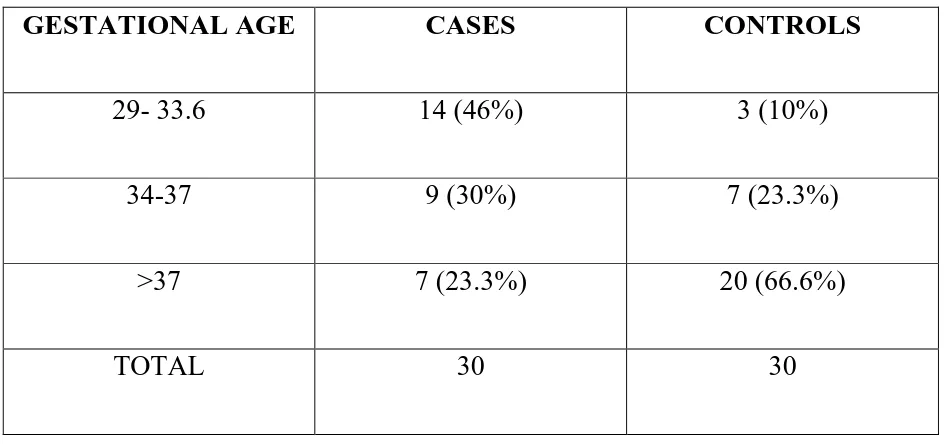
GESTATIONAL AGE:
GESTATIONAL AGE CASES CONTROLS
29- 33.6 14 (46%) 3 (10%)
34-37 9 (30%) 7 (23.3%)
>37 7 (23.3%) 20 (66.6%)
TOTAL 30 30
In 30 cases, 14 patients were between 29-34 weeks, 9 patients between
34-37 weeks, 7 patients were more 34-37 weeks. In controls, 3 patients between 29-34
57 TABLE-5:
THE MEAN GESTATIONAL AGE:
SNO N Mean SD
1 CASES 30 34.80 2.398
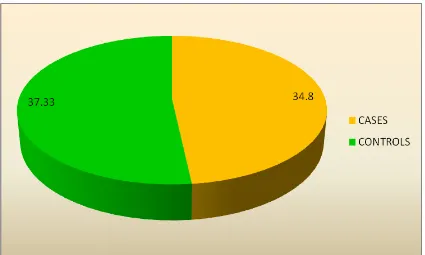
2 CONTROLS 30 37.33 1.493
The Mean Gestational Age in cases are 34±2.398 and the mean gestational
Age of the controls are 37.33±1.493.The mean gestational was more in controls
when compared to cases, this is because all the preeclamptic patients were
58 TABLE-6:
BMI
BMI CASES CONTROLS
18-24.9 0 5( 16.6%)
25-29.9 9( 30%) 10( 33.3%)
>30 21(70%) 15( 50%)
>35 0% 0%
TOTAL 30 30
From the above table out of 30 cases, 21 patients ( 70%) comes under class I
obesity when compared to controls it was only 50%. No patients were found to
59 TABLE-7:
THE MEAN BMI
N Mean SD P value
CASES 30 30.333 2.2489
.002
CONTROLS 30 28.517 2.0615
BMI of both the cases and controls were noted and mean was calculated .
The mean of BMI in cases was 30.333±2.2489 when compared to controls the
mean value was 28.517±2.06 which was statistically significant . P value was <
60 TABLE-8:
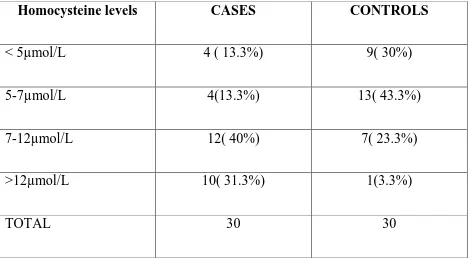
HOMOCYSTEINE LEVELS
Homocysteine levels CASES CONTROLS
< 5µmol/L 4 ( 13.3%) 9( 30%)
5-7µmol/L 4(13.3%) 13( 43.3%)
7-12µmol/L 12( 40%) 7( 23.3%)
>12µmol/L 10( 31.3%) 1(3.3%)
TOTAL 30 30
The normal value of homocysteine is 5-7µmol/L, 4( 13.3%) patients in
cases and 9( 30%) patients in controls had low values that is below 5µmol/L. 4
(13.3%)patients in cases and 13 ( 43.3%)patients in controls had normal value of
homocysteine. 12( 40%) patients in cases and 7( 23.3%)patients in controls had
mild elevation of homocysteine. In my study 10( 31.3%) patients in cases and
61
62 TABLE-9:
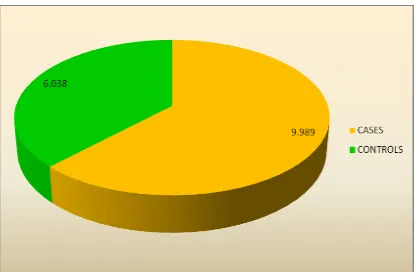
MEAN HOMOCYSTEINE LEVELS IN CASES AND CONTROLS
SNO N Mean SD P value
1 CASES 30 9.989 4.1624
0.000
2 CONTROLS 30 6.038 2.0390
This table shows the association of serum homocysteine levels in cases and
controls . The mean homocysteine value in case is 9.989±4.1 and the mean
homocysteine level in control is 6.038±2.03 , which found to be statistically
63 TABLE-10:
ASSOCIATION OF HOMOCYSTEINE IN NORMOTENSIVES AND IN WOMEN WITH PREECLAMPSIA:
Normal Increased
homocysteine level
Less than normal homocysteine
CONTROLS 19(63.33%) 4(13.333%) 7(23.33%)
CASES 6(20%) 24(80%) 0
The above table shows that homocysteine value is unchanged in 63.3% of
the normotensive patients and is decreased less than normal range in 23.33% when
compared to homocysteine levels in preeclampsia patients were 80% of patients
64 TABLE-11:
ASSOCIATION OF HOMOCYSTEINE LEVELS WITH MILD PREECLAMPSIA AND SEVERE PREECLAMPSIA
Normal homocysteine
Increase homocysteine
Total
Non severe preeclampsia 8(61%) 5(38%) 13
Severe preeclampsia 5(29.4%) 12( 70.5%) 17
From the above table it is found that increased homocysteine levels was
found in severe preeclampsia 12(70% )patients when compared to mild
preeclampsia which was only 5(38%) patients. More number of patients are
65
TABLE-12:
HEMOGLOBIN
HEMOGLOBIN CASES CONTROLS
<8G/DL 3 (10%) 0%
8.1-9.5 G/DL 5(16.6%) 0%
9.6-10.5 G/DL 6(20%) 16(53.33%)
>10.5 G/DL 16(53.3%) 14(46.6%)
TOTAL 30 30
In cases, 3 patients had severe anemia, 5 patients had moderate anemia, 6
patients with mild anemia, more than 16 patients had normal HB levels. In
66
TABLE-13:
THE MEAN HB VALUE FOR CASES AND CONTROLS
N MEAN SD P VALUE
CONTROLS 30 10.800 2.1148
.924
CASES 30 10.840 0.8830
The mean value of HB was done in both cases and controls. It was found
that the mean HB value of cases 10.800±2.11 and the mean HB value of controls
67 TABLE-14:
ASSOCIATION OF HOMOCYSTEINE LEVELS WITH IUGR
Homocysteine With IUGR Without IUGR P value
Normal 2 (25%) 6 (75%)
0.614
Mild elevation 2(15.4%) 11(84.6%)
Moderate elevation 6(66.7%) 3( 33.3%)
Raised homocysteine value showed higher incidence of IUGR 6 ( 66.6%)
when compared to low level of homocysteine 2 (15.4%) but it is not statistically