Copyright © 2003, American Society for Microbiology. All Rights Reserved.
Peripheral Blood Cytotoxic
␥␦
T Lymphocytes from Patients with
Human Immunodeficiency Virus Type 1 Infection and AIDS
Lyse Uninfected CD4
⫹
T Cells, and Their Cytocidal
Potential Correlates with Viral Load
Sardar T. A. K. Sindhu, Rasheed Ahmad, Richard Morisset, Ali Ahmad,
and Jose´ Menezes*
Laboratory of Immunovirology, Department of Microbiology and Immunology, Ste. Justine Hospital Research Center and Hotel-Dieu Hospital, University of Montreal, Montreal, Quebec, Canada
Received 8 August 2002/Accepted 6 November 2002
Progression of human immunodeficiency virus type 1 (HIV-1) infection in humans is marked by declining
CD4ⴙ-T-cell counts and increasing virus load (VL). Cytotoxic T lymphocytes (CTL) play an important role in
the lysis of HIV-infected cells, especially during the early phase of asymptomatic infection. CTL responses in
the later phase of disease progression may not be as effective since progressors with lower CD4ⴙ-T-cell counts
have consistently higher VL despite having elevated CTL counts. We hypothesized that, apart from antiviral
effects, some CTL might also contribute to AIDS pathogenesis by depleting CD4ⴙT cells and that this CTL
activity may correlate with the VL in AIDS patients. Therefore, a cross-sectional study of 31 HIV-1-infected patients at various clinical stages was carried out. Purified CTL from these donors as well as HIV-seronegative
controls were used as effectors against different human cell targets by using standard51Cr release cytolytic
assays. A direct correlation between VL and CTL-mediated, major histocompatibility complex
(MHC)-unre-stricted lysis of primary CD4ⴙ-T-cell, CEM.NKR, and K562 targets was observed. CD4ⴙ-T-cell counts and
duration of infection also correlated with MHC-unrestricted cytolytic activity. Our data clearly show that␥␦
CTL are abnormally expanded in the peripheral blood of HIV-infected patients and that the V␦1 subset of␥␦
T cells is the main effector population responsible for this type of cytolysis. The present data suggest that␥␦
CTL can contribute to the depletion of bystander CD4ⴙ T cells in HIV-infected patients as a parallel
mechanism to HIV-associated immunopathogenesis and hence expedite AIDS progression.
Human immunodeficiency virus type 1 (HIV-1) infection in humans is marked by an initial phase of viremia and febrile reaction. Chronic immune activation induced by HIV-1 leads to increased levels of activated cytotoxic T lymphocytes (CTL) in the peripheral circulation (40) which are sustained for years following seroconversion (23). During the asymptomatic pe-riod, despite low viremia and the infection of only a fraction of the CD4⫹T cells, the number of blood CD4⫹T cells generally
declines over time (20, 36). It may therefore be speculated that there is some parallel mechanism involved in the depletion of CD4⫹T lymphocytes besides the direct cytolytic effects of HIV
replication (2). The mechanisms leading to depletion of CD4⫹
T lymphocytes in vivo (reviewed in reference 14) appear to be pivotal to AIDS immunopathogenesis, and the various phe-nomena suggested include immunopathology (56; M. B. Fein-berg, J. M. McCune, F. Miedema, J. P. Moore, and H. Schuite-maker, Letter, Nat. Med. 8:537, 2002), autoimmunity (27), spontaneous (Fas-mediated) apoptosis (3, 4, 16), superanti-gen-mediated deletion (32), and complement-dependent lysis (21).
The majority of circulating CTL in humans expresses CD8 antigen in association with the␣T-cell receptor (TCR) phe-notype. These classical CTL engage and eliminate
virus-in-fected cells and tumor cells via recognition of MHC class I peptide complexes on the target cells (50) and may also sup-press virus replication via a non-contact-mediated mechanism involving cytokines (35, 51, 52). However, a minor population (5 to 10%) expresses an alternative heterodimer consisting of
␥and␦chains, and in contrast to␣T cells, functional␥␦CTL identify and lyse targets in an MHC-unrestricted manner. The majority of circulating ␥␦T cells belongs to the V␦2 subset, whereas a smaller number belong to the V␦1 subset (8). Very little is known about the function of, and the antigen(s) rec-ognized by, V␦1␥␦CTL, but their selective expansion during certain disease conditions has been reported, e.g., in lungs of pulmonary sarcoidosis patients (19), in synovial fluid from pa-tients with rheumatoid arthritis (11), in leprotic lesions (49), in the intestinal lesions of patients with celiac disease (43), in cerebrospinal fluid of multiple sclerosis patients (41), and in the peripheral blood of HIV-infected patients (8).
After polyclonal activation in vitro, some CTL subsets from AIDS patients can eliminate CD4⫹ T lymphocytes without
MHC-restricted target recognition (7). Such CTL subsets in HIV-1-infected individuals seem to cause immunopathology by destroying the bystander (uninfected) CD4⫹T lymphocytes in
blood (23, 55). We hypothesized that if such CTL subsets were present in blood of AIDS patients and caused CD4⫹-T-cell
depletion, then their anti-CD4⫹cytocidal activity would
cor-relate with the viral load (VL) as well as with the course of HIV disease progression. Therefore, whereas previous studies * Corresponding author. Mailing address: Laboratory of
Immuno-virology, Ste. Justine Hospital, 3175 Coˆte Ste.-Catherine, Montreal, Quebec H3T 1C5, Canada. Phone: (514) 345-4931/6134. Fax: (514) 345-4801. E-mail: jmenezes@justine.umontreal.ca.
1848
on November 8, 2019 by guest
http://jvi.asm.org/
(23–26) have focused mainly on the phenomenon of CD4⫹
-T-cell depletion, we investigated the pathological role of various CTL in AIDS infection by analyzing cytolytic responses in relation to CD4⫹-T-lymphocyte counts, VL, and the duration
of clinical infection. Here, we present evidence showing the following: (i)␥␦CTL can lyse cells other than CD4⫹T
lym-phocytes, which implies that the pathogenic ramifications at-tributable to CTL-linked cytolysis may be even more diverse than previously thought; (ii) CTL-mediated lysis of target cells is directly correlated with the VL of patients; and (iii)␥␦CTL are abnormally expanded in the blood of patients (interesting-ly, the effector cells responsible for the lysis we observed were found to be mainly of the V␦1 phenotype).
(This work was presented in part at the 19th Annual Meet-ing of the American Society for Virology, Fort Collins, Colo., 8 to 12 July 2000 [abstract no. W9-6].)
MATERIALS AND METHODS
Subjects.Thirty-one HIV-1-infected patients classified as being asymptomatic or symptomatic or as having AIDS according to the guidelines of the Centers for Disease Control and Prevention (15) were recruited through the University of Montreal Medical Center’s AIDS clinic (at Hotel-Dieu Hospital, Montreal, Canada) for this cross-sectional study. The subjects were found to be seropositive for HIV-1 infection by enzyme-linked immunosorbent assay, with confirmation by Western blotting. Their infections were at different stages of clinical progres-sion, and their CD4⫹-T-lymphocyte counts ranged from 513 perl to as low as
16 perl. The VL of these patients varied from log105.38 copies of HIV-1
RNA/ml to less than log102.70 copies of HIV-1 RNA/ml. The duration of known
HIV seropositivity (hereafter referred to as duration of infection) of the patients studied varied from 1 to 10 years. The associated infections and malignancies in the AIDS patients included shingles, Epstein-Barr virus, hepatitis C, tuberculo-sis, pneumococcal and pneumocystis pneumonias, candidiatuberculo-sis, fungal pharyngitis, Kaposi’s sarcoma, and lymphopathies (polyadenopathy, Hodgkin lymphomas, and undifferentiated lymphomas). The patients were on similar regimens of highly active antiretroviral therapy (HAART). The controls in this study in-cluded 14 HIV-1-seronegative, healthy, and age-matched donors. All blood sam-ples were collected after informed, written consent of the individuals and ap-proval of the institutional ethics committee had been obtained.
Separation of PBMC.Peripheral blood mononuclear cells (PBMC) were sep-arated from blood as described previously (23). Briefly, heparinized blood sam-ples were diluted (1:2) with RPMI 1640 medium (Gibco, Grand Island, N.Y.), layered over Ficoll-Hypaque gradient (Pharmacia Chemicals, Montreal, Cana-da), and spun at 400⫻g(GPR centrifuge; Beckman, Palo Alto, Calif.) for 25 min. The interface cells were washed four times in RPMI 1640 medium supple-mented with 2% decomplesupple-mented fetal bovine serum (FBS; Gibco Life Tech-nologies, Grand Island, N.Y.) and resuspended at a concentration of 106cells/ml
in RPMI 1640 culture medium supplemented with 10 mM HEPES, 2 mM
L-glutamine, 100 U of penicillin/ml, 100g of streptomycin/ml, and 10% FBS. Stimulation of PBMC.For activation and expansion of CTL (used later as effectors), PBMC from HIV-infected patients and HIV-seronegative donors were incubated with concanavalin A (20g/ml; ICN Biomedicals, Montreal, Canada) for 3 days (at 37°C, an atmosphere of 5% CO2, and 84% relative
humidity). Interleukin 2 (IL-2) (Proleukin; Cetus, Emeryville, Calif.) was then added at a concentration of 100 U/ml, and the cells were incubated for 4 more days.
For expansion of CD4⫹T lymphocytes (used as targets), PBMC from healthy
donors were cultured in phytohemagglutinin (PHA; 10g/ml; ICN Biomedicals) for 3 days and subsequently with IL-2 (100 U/ml) for 24 h.
Antibodies and TCR blocking.The different monoclonal antibodies (MAbs) used for labeling and/or immunomagnetic purifications of CTL and CTL subsets were as follows: anti-human␣TCR (clone, T10B9.1A-31; isotype, mouse im-munoglobulin M [IgM]; BD Pharmingen, San Diego, Calif.),␣pan-TCR (clone, BMA031; isotype, IgG2b; Endogen,),␥␦TCR (clone, B1; isotype, mouse IgG1; BD Pharmingen),␥␦pan-TCR (clone, 5A6.E91; isotype, IgG1;
Endo-gen), V␦1 TCR (clone, R9.12; isotype, IgG1; Biodesign), and V␦2 TCR (clone, Immu389; isotype, IgG1; Biodesign) and fluorescein isothiocyanate- and phyco-erythrin-labeled goat anti-mouse IgG isotype control antibodies (BD Pharmin-gen, Mississauga, Ontario, Canada). All antibodies were used according to the
manufacturers’ instructions. Briefly, the cells were washed twice with sterile phosphate-buffered saline and once with RPMI 1640 medium containing 2% FBS. Separate lots were incubated with control antibodies and MAbs (1g/2⫻ 105cells) on ice for 30 min, washed twice, and used as labeled effectors in
cytolytic assays.
Cell purifications.CTL (CD3⫹, CD8⫹, and CD8⫺) and CD4⫹T lymphocytes
were separated by using a negative selection method (immunomagnetic column separation technique; Stem Cell Technologies, Vancouver, Canada) according to the manufacturer’s instructions. Briefly, in vitro-activated PBMC were washed thrice and the pellet was resuspended in 1 ml of RPMI 1640 medium containing 2% FBS. Respective antibody cocktails for purification of CTL (i.e., CTL en-richment cocktail containing MAbs to CD4, CD16, CD19, CD36, and CD56 cell surface antigens) and of CD4⫹T lymphocytes (CD4⫹-T-cell enrichment cocktail
containing MAbs to CD8, CD16, CD19, CD36, and CD56 cell surface antigens) were added separately (100-l/ml concentration each) to the cell suspension, followed by mixing and then incubation on wet ice for 30 min. Magnetic colloid (microbeads precoated with anti-mouse IgG) was then added (60l/ml), and the cells were reincubated on ice for 30 min with intermittent shaking every 10 min. The cell suspension was then passed through a magnetic column, and the purified cells (unlabeled fraction) were eluted. The purities of separated cell populations for the selection markers (89%⫾5% [mean⫾standard deviation] for CD8⫹
cells and 91%⫾4% for CD4⫹T cells) were determined by flow cytometry (data
not shown).
To isolate␣and␥␦CTL (␦1 and␦2) populations, two-step and three-step immunomagnetic column purifications, respectively, were carried out using PBMC according to the manufacturer’s instructions. Briefly, purified CTL (i.e., following removal of CD4-, CD16-, CD19-, CD36-, and CD56-positive cells) were treated with anti-human␥␦pan-TCR MAbs to elute␣CTL through the column. The other aliquot was labeled with␣pan-TCR MAbs to obtain the␥␦ CTL fraction, which, at the third step of purification, yielded V␦1 and V␦2 subsets when anti-V␦2 and anti-V␦1 TCR-specific MAbs, respectively, were used as two separate treatments. The purities of the CTL preparations thus obtained were as follows: 99.08%⫾1.09%, 97.10%⫾1.42%, and 96.46%⫾0.81% for␣, ␦1, and␦2 CTL, respectively.
CD4ⴙ-T-lymphocyte counts and plasma VL.The peripheral blood CD4⫹
-T-lymphocyte counts of the patients were determined by flow cytometry by using the whole-blood lysis method and MAbs from Becton Dickinson (San Jose, Calif.). The plasma VL (log10HIV RNA copies per milliliter) was quantified by
an Amplicor HIV monitor test (Roche Diagnostic System Inc., Somerville, N.J.) following the manufacturer’s instructions. Briefly, a 142-bp region in thegaggene of HIV-1 was amplified by reverse transcription-PCR, and enzyme-linked im-munosorbent assay was used to detect biotinylated HIV-1 and standard ampli-cons. Finally, the RNA copy number was determined by following a standard protocol.
Cytotoxicity (51Cr release) assay.The standard procedure for the51Cr release
assay (1, 7) was followed with minor modifications. Briefly, effector cells were prepared by washing purified CTL thrice (with RPMI 1640 medium with 2% FBS) and resuspending them in RPMI 1640 medium with 10% FBS at a con-centration of 4⫻106cells/ml. CD4⫹-T-lymphocyte targets were also washed in
the same way and were chromium labeled by incubation with Na251CrO4(New
England Nuclear, Boston, Mass.) at a dose rate of 100Ci/106cells for 90 min.
The cells were later washed four times and resuspended at a concentration of 2⫻ 105cells/ml. Other cell targets, i.e., CEM.NKRcells (a CD4⫹-T-cell line resistant
to NK cell-mediated lysis and lacking expression of HLA class II antigens) and K562 cells (an erythroleukemic cell line lacking CD4 molecules and MHC class I antigens), were similarly labeled and prepared. First, each target cell suspen-sion was added (50l/well) to triplicate wells of 96-well, V-bottom microtiter plates (Nunclon, Roskilde, Denmark). Effector cell suspension was then added to triplicate wells in amounts of 50 and 100l/well to achieve E:T ratios of 20:1 and 40:1, respectively. No effectors were added to the negative (i.e., spontane-ous) control wells. The volume was increased to 200l/well by the addition of medium to the experimental and control wells. HCl (0.1 N) was added to the maximum control wells. The plates were subsequently incubated at 37°C in a humidified, 5% CO2incubator for 8 h. Supernatants from triplicate wells were
drawn (100l each) for gamma counting, and the percentage of specific lysis was calculated using the following standard formula: percentage of specific lysis⫽ [(experimental 51Cr release ⫺spontaneous release) ⫻100]/(maximum 51Cr
release⫺spontaneous release).
Statistical analysis.Determination of the correlation (rs) between the level of specific cytolysis and each of the different clinical parameters was carried out with Spearman’s rank correlation test by using the Microsoft Excel program. The Shapiro-Wilk’s test was used to check the normality of distributions. The differ-ences between mean values (Mann-Whitney nonparametricttest) were
on November 8, 2019 by guest
http://jvi.asm.org/
ered significant atPvalues ofⱕ0.05. The Prism program (GraphPad Software, Inc., San Diego, Calif.) was used to create graphic presentations of the data.
RESULTS
Lysis of autologous and allogeneic CD4ⴙT, CEM.NKR, and
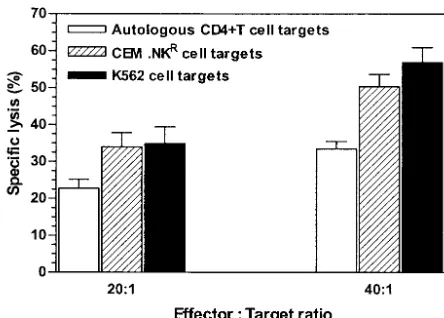
K562 cells by purified CTL from AIDS patients.Experiments
were first carried out by using CTL effectors from four AIDS patients against autologous primary CD4⫹ T cells and two
target cell lines at E:T ratios of 20:1 and 40:1 (Fig. 1). Given that autologous CD4⫹T cells were lysed by CTL, the study was
then extended by including allogeneic CD4⫹T cells from
HIV-1-seronegative healthy donors. Target cytolysis was analyzed in relation to three clinical parameters of CTL donors, i.e., pe-ripheral CD4⫹-T-cell counts, VL, and duration of infection.
For each of the three clinical parameters, CTL donors were arbitrarily subdivided as follows. One subgroup included indi-viduals with CD4⫹-T-cell counts ofⱖ300/l, while the
coun-terpart group consisted of subjects with counts of ⬍300/l. With respect to the VL of the patients at the time of blood sampling, those regarded as having low copy numbers of plasma viral RNA (ⱕlog10 2.70/ml) formed one group and
those regarded as having high copy numbers of plasma viral RNA (⬎log102.70/ml) formed another group. Similarly,
sub-jects having a duration of infection ofⱕ3 years were consid-ered as one subgroup and the rest were considconsid-ered as the counterpart group. Experimental controls were run
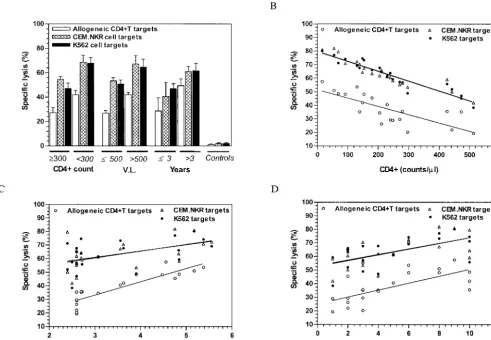
simulta-neously by using purified in vitro expanded CTL from age-matched HIV-1-seronegative individuals. The obtained data on CTL-mediated lysis of allogeneic CD4⫹-T-cell, CEM.NKR,
and K562 targets with respect to the three clinical parameters are shown in Fig. 2A. The mean differences between the data for each target and that for the respective controls were sta-tistically significant (P ⬍ 0.05). The data on the correlation between the specific lysis of each target and CD4⫹-T-cell
counts, VL, and duration of infection are summarized in Fig. 2B, C, and D, respectively. For CD4⫹-T-cell targets, the
per-centage of cytolytic activity correlated negatively with CD4⫹
-T-cell counts (rs⫽ ⫺0.79) and positively with both VL and the
duration of infection (rs⫽0.92 andrs⫽0.76, respectively). As
for CEM.NKRtargets, the correlations (r
svalues) with CD4⫹
-T-cell counts, VL, and duration of infection were⫺0.97, 0.50, and 0.59, respectively. The specific lysis of K562 cells also correlated with the CD4⫹-T-cell counts (r
s⫽ ⫺0.96), VL (rs⫽
0.47), and duration of infection (rs⫽0.59) of the CTL donors
(Pvalue of⬍0.05 in each case).
Hence, both the CEM.NKR and K562 cell lines were as
susceptible to CTL from AIDS patients as were the autologous and allogeneic CD4⫹ T lymphocytes. Moreover, unlike
pri-mary CD4⫹-T-cell targets, both the CEM.NKRand K562 cell
lines did not require activation for CTL-mediated cytolysis in vitro (data not shown). We thus found that CTL-mediated lysis of these targets was related neither to the presence or absence of MHC class I and II surface antigens nor to the CD4⫹
receptors on them.
CTL immunophenotypes and subtypes in HIV-infected
pa-tients. Since CTL from AIDS patients showed
MHC-unre-stricted killing of various cell types, we further determined the TCR phenotypes in the peripheral blood of these donors. The data were statistically analyzed by grouping donors as low virus load (LVL; i.e., VL of ⬍log102.70 copies/ml), medium virus
load (MVL; i.e., VL between log102.70 and log103.70 copies/
ml), and high virus load (HVL; i.e., VL of⬎log103.70 copies/
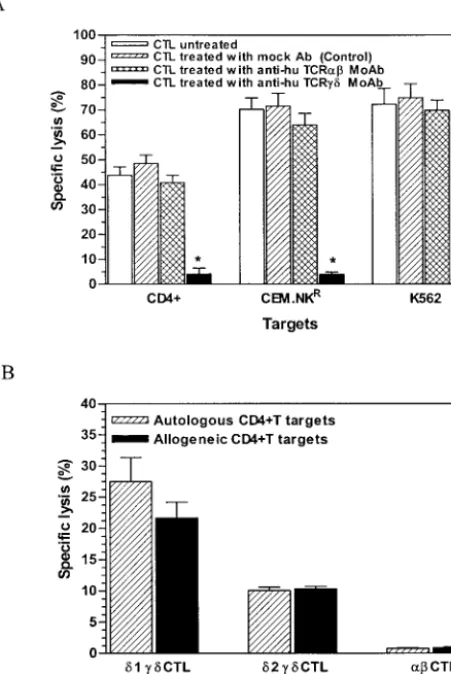
ml) patients. As shown in Fig. 3A, the mean percentage of expression of␣TCR⫹CTL decreased and that of␥␦TCR⫹
CTL increased with increasing VL in HIV-infected patients. Compared with the results for the control individuals, however, only the HVL group showed overall significant alterations (for
␣ CTL, P ⬍ 0.0092; for ␥␦ CTL, P ⬍ 0.0005). The CTL effector populations from 10 HVL patients were also used to determine the ␦1 and ␦2 ␥␦ CTL subsets by fluorescence-activated cell sorter analysis. Figure 3B shows that the V␦1␥␦
CTL subset was predominant in HIV-infected patients while the V␦2 subset was predominant in the HIV-seronegative con-trols (P⬍0.05).
Lysis is precluded following TCR blockage by specific MAbs
and elimination of the V␦1 subset from ␥␦ CTL effectors.
[image:3.603.52.274.68.227.2]Because the above-mentioned data showed that peripheral blood␥␦CTL are expanded in HIV progressors with HVL and that the CTL from these individuals displayed relatively higher killing activity, we sought to determine which subset of␥␦CTL was the principal effector of this cytolytic activity. Therefore, anti-human␣and␥␦pan-TCR MAbs were used to pretreat CTL before they were mixed with the targets. The data ob-tained indicate that anti-␥␦pan-TCR MAb suppressed target cytolysis (P⬍0.0001) (Fig. 4A). A similar suppression of lysis was observed after either CTL pretreatment with anti-human FIG. 1. CTL-mediated killing of autologous CD4⫹-T-cell,
CEM.NKR, and K562 targets. CTL were prepared by first treating PBMC from four HIV-infected patients with concanavalin A (20g/ml for 3 days) and IL-2 (100 U/ml for 4 days) and then purifying the effectors immunomagnetically (i.e., by negative selection, following elimination of CD4-, CD16-, CD19-, CD36-, and CD56-positive cells). CD4⫹-T-cell targets were prepared by treating PBMC from the same donors with PHA (10g/ml for 3 days) and IL-2 (100 U/ml for the following 24 h) and then purifying them by using an immunomagnetic separation method (i.e., by removing CD8-, CD16-, CD19-, CD36-, and CD56-positive cells). Effectors were mixed with51Cr-labeled tar-gets (CD4⫹T, CEM.NKR, and K562 cells were mixed with Na251CrO4 at a concentration of 100Ci/106cells for 90 min, and the cells were then washed four times) at the indicated E:T ratios in triplicate wells of 96-well microculture plates and incubated at 37°C for 8 h. The percentage of specific lysis (cytotoxicity) was determined from the radioactivity released into the supernatants. Each bar in the figure represents mean cytotoxicity plus standard error of mean (SEM) de-termined from triplicate experiments. All lysis values were normalized
with respect to the relevant spontaneous and maximum controls.
on November 8, 2019 by guest
http://jvi.asm.org/
V␦1␥␦ TCR MAb or removal of this particular subset from the effector population (Fig. 4B, autologous CD4⫹-T-cell
tar-gets). Interestingly, the decrease in cytolysis observed when anti-␣TCR MAb was used was insignificant in most cases and CTL labeling with anti-human V␦2 ␥␦ TCR MAb also pro-duced less cytolysis (data not shown). As expected, mock-treated and unmock-treated CTL effectors showed comparable cyto-lytic activities for the targets used.
DISCUSSION
The depletion of CD4⫹ T lymphocytes is a hallmark of
HIV-related pathogenesis in humans, and different scenarios have been proposed to explain this phenomenon. Since the massive replication of HIV during progression has been as-cribed as being only one of the contributive mechanisms of CD4⫹-T-cell depletion, more information is required to
vali-date any of the other suggested hypotheses. It is well known that chronic immune activation associated with HIV-1 infec-tion is the major reason for the inducinfec-tion of various CTL subsets and that HIV-1-specific CTL are primarily involved in suppressing virus replication (9, 35, 48). Nonetheless, a posi-tive correlation between CTL expansion and rapid progression to AIDS (24) has also been reported, and previous studies have shown that CTL effectors from AIDS patients lyse CD4⫹T
lymphocytes (22, 24, 55, 56) and are of the␣TCR⫹
pheno-type (23). However, no study has evaluated simultaneously the roles of␣and␥␦CTL effectors in the depletion of CD4⫹T
cells in HIV-positive patients. Therefore, the present study adds a new element to these scenarios by showing that␥␦CTL from HIV-infected patients can lyse not only autologous CD4⫹
T lymphocytes but also allogeneic primary CD4⫹T cells, as
[image:4.603.52.543.74.413.2]well as human lymphoid cell lines. We compared the cytocidal FIG. 2. Analyses of CTL-mediated lysis of different targets in relation to CD4⫹-T-cell counts, VL, and duration of known HIV seropositivity. (A) CTL-mediated lysis of allogeneic CD4⫹-T-cell, CEM.NKR, and K562 targets. The percentage of specific lysis of allogeneic CD4⫹T cells (obtained from eight HIV-1-seronegative donors) by purified CTL effectors from 21 HIV-infected patients is compared with that of CEM.NKR and K562 cell line targets. Controls represent the cytolysis of these targets by CTL from six age-matched, healthy donors. The values for cell killing activity were obtained at an E:T ratio of 40:1 (killing at an E:T ratio of 20:1 produced relatively similar results for the targets used [data not shown]). (B) Negative correlation between peripheral blood CD4⫹-T-lymphocyte counts of HIV-1-infected CTL donors and the specific lysis of allogeneic CD4⫹T, CEM.NKR, and K562 cells. The correlation values (rs) for the three targets were 0.79 (P⬍0.0001), 0.79 (P⬍0.0001), and 0.96 (P⬍0.0001), respectively. (C) Correlation between VL of CTL donors and specific lysis of allogeneic CD4⫹-T-cell, CEM.NKR, and K562 targets. The correlation values (rs) for the three targets were 0.92 (P⬍0.0001), 0.50 (P⫽0.02), and 0.47 (P⫽0.032), respectively. (D) Correlation between the duration of infection and specific lysis of allogeneic CD4⫹-T-cell, CEM.NKR, and K562 targets. The correlation values (rs) for the three targets were 0.76 (P⬍0.0001), 0.59 (P⫽0.005), and 0.59 (P⫽0.005), respectively. All data were obtained from triplicate experiments.
on November 8, 2019 by guest
http://jvi.asm.org/
effector function of different CTL subsets either by specific MAb-mediated TCR blocking or by elimination of the coun-terpart subset(s) from the effector population. Our data clearly indicate that peripheral blood ␥␦ CTL in HIV-infected pa-tients are abnormally expanded and are predominantly
respon-sible for MHC-unrestricted lysis of various human cell types, whereas the␣TCR⫹effectors display only a minor level of
cytolytic activity. Our data also show that the V␦1 CTL subset represents the main effectors of this cytolysis and that targets with and without CD4⫹or MHC class I or II molecules are
susceptible to their cytolytic activity.
Normally, ␥␦ T cells constitute only 5 to 10% of human peripheral blood CTL (39), but abnormal expansions, espe-cially of the V␦1␥␦T-cell subset, in several disease conditions
[image:5.603.316.542.206.543.2]FIG. 3. CTL immunophenotypes and their relationship to HIV in-fection and VL. (A)␣and␥␦CTL populations in the activated CTL from HIV-infected patients and HIV-seronegative controls. As de-scribed in the legend to Fig. 1, CTL effectors were analyzed for ex-pression of TCR phenotypes. Enriched T cells (depleted of CD4-, CD16-, CD19-, CD36-, and CD56-positive populations) obtained from HIV-positive and HIV-seronegative (age-matched, healthy) donors were washed thrice in cold RPMI 1640 medium with 2% FBS and labeled with␣and␥␦pan-TCR specific MAbs according to standard protocols. FACScan data, presented as means plus SEM, were ob-tained from five independent determinations that included three groups as follows: LVL (patients with VL of⬍log102.70 copies of HIV-1 RNA/ml), MVL (patients with VL between log102.70 and log10 3.70 copies of HIV-1 RNA/ml), and HVL (patients with VL of⬎log10 3.70 copies of HIV-1 RNA/ml). Asterisks represent statistically signif-icant mean difference from the respective control value for each TCR phenotype. (B) V␦1 and V␦2␥␦CTL subsets in HIV-positive patients and HIV-seronegative controls. For these determinations, PBMC from 10 HVL HIV-positive individuals and six HIV-seronegative controls were purified for ␥␦ CTL and stained with anti-␦1 and anti-␦2 ␥␦ TCR-specific MAbs as well as isotype control antibodies. FACScan data are presented as the percentages of positive cells of each subset for HIV-infected patients and control individuals. For each phenotype, the differences between the values for HIV-infected patients and those for the controls were statistically significant (P⬍0.05; Mann-Whitney nonparametricttest).
FIG. 4. Analyses of anti-CD4⫹effector CTL phenotypes. (A) Sup-pression of target cytolysis following CTL labeling by TCR-specific MAbs. In vitro-activated and purified CTL from six HIV-infected patients were mixed with51Cr-labeled targets in the cytotoxicity assays. The effectors (CTL) were pretreated with anti-human ␣ and ␥␦ pan-TCR-specific isotype control antibodies by using recommended optimum concentrations before the CTL were mixed with the targets. The percentage of specific lysis (cytotoxicity) was determined from the amount of radioactivity released into the supernatants after 8 h of incubation at 37°C. Each bar in the figure represents mean cytotoxicity plus SEM from triplicate experiments. Asterisks indicate statistically significant differences (P⬍0.05) from the values for the respective controls for each target used. (B) Comparative lysis by purified TCR ␣, V␦1, and V␦2 ␥␦ CTL effectors. Shown is the percentage of specific lysis of allogeneic CD4⫹T lymphocytes by purified CTL ef-fectors from four HVL patients at an E:T ratio of 20:1, as determined by 8-h microculture cytolytic assays. The difference (nonparametrict
test) between the mean percentage of lysis by the␦1 and␦2 effectors and ␣CTL-mediated lysis was found to be statistically significant (P⬍0.05).
on November 8, 2019 by guest
http://jvi.asm.org/
including HIV-1 infection (53) and neoplasia (29) have been reported. Although selective expansion of peripheral blood␥␦
T cells has been reported during HIV-1 infection (8, 53), the immunological consequences of this phenomenon have not yet been explained. We speculate that␥␦ T cells induced in the early stages of acute HIV-1 infections are maintained as pre-cursors of their effector subsets during progression. Conse-quently, ex vivo expansion of these precursors would yield reactivated effectors that may cause depletion of various cell targets, as demonstrated in our microculture cytotoxicity as-says. Since these CTL lyse both infected and uninfected tar-gets, it seems obvious that their effector function is directed not towards a virus-specific antigen(s) but rather against the cellular ligand(s) commonly expressed on targets as a sequel to immune activation and/or dysregulation. It may be noted here that, as part of the indirect consequences of HIV infection, sustained overstimulation of the immune system during infec-tion has been reported as a factor leading to cumulative, det-rimental effect (i.e., CD4⫹-T-cell depletion) (Feinberg et al.,
letter). Given the potential of immortalized human lymphoid cell lines for continuous division, the target ligand(s) may al-ready be highly expressed on CEM.NKRand K562 cells
com-pared with the primary CD4⫹-T-cell targets. This is perhaps
the reason why, as our results indicate, these cell lines (unlike CD4⫹ T cells) do not require further reactivation for their
CTL-mediated cytolysis.
To our knowledge, there are no reports presently available that define either the cause or the mechanism of selective expansion of␥␦CTL in the peripheral blood of HIV progres-sors or identify the target molecule(s) mediating this cytolytic activity. Most human ␥␦ CTL (V␥2V␦2 or V␥9V␦2 subset) recognize non-peptide phosphorylated antigens, isopentenyl phosphate, and related prenyl pyrophosphate molecules (46, 47) as well as alkylamine antigens (13). Both the phosphate and amine antigens are important products of microbes and self-antigens, and their presentation does not involve MHC class I or class II peptide antigen-presenting molecules (31, 33, 38). It might be speculated that these antigens may be recog-nized much as haptens are recogrecog-nized by immunoglobulins and TCRs of cytotoxic T cells (12). We show that both activated autologous and allogeneic primary CD4⫹T cells, as well as the
CEM.NKRand K562 cell lines, are lysed by␥␦ CTL. Which
target molecules are involved in this cytolysis by␥␦CTL from HIV-positive individuals is not known. However, it has previ-ously been reported (37) that CD4⫹ T cells activated with
either PHA or anti-CD3 antibodies express MHC class I-re-lated surface MICA and MICB (28, 29, 44), whereas the CEM.NKRand K562 cell lines express ULBPs, with the
ex-pression of ULBPs being independent of the exex-pression of surface MHC class I and class II antigens on these cells (17, 45). Therefore, it is probable that MICA and MICB and/or ULBP ligands are expressed on these targets and mediate cytolysis by delivering both TCR-dependent signal 1 and NKG2D-dependent signal 2 (5) to the V␦1 T effectors (54) and signal 2 to the V␥9␦2 CTL effectors (18). Widely expressed nonpolymorphic CD1c molecules may also be involved in TCR-mediated recognition and target cytolysis by the V␦1␥␦
CTL (6, 42). In any event, it remains to be determined which type of ligand molecules are specifically induced or modified on the target cells during HIV infection and to establish
whether these molecules play a role in␥␦CTL-mediated cy-tolysis.
In view of their broad-scale ex vivo killing potential as shown by this study, the V␦1␥␦CTL effectors may be associated, at least in part, with the immunopathology observed in HIV pro-gressors. Furthermore, it is interesting that functional anergy of the V␦2 T subset in as many as 60% of asymptomatic HIV-infected patients has also been reported (53). Our results showing the correlation of CTL-mediated killing with the var-ious clinical parameters are also corroborated by prevvar-ious studies (10, 34). Persistent decline in CD4⫹-T-cell counts is
both a hallmark and a prognostic indicator of HIV progression. Subsequent to the onset of symptomatic HIV infection, sharp declines in CD4⫹-T-cell counts and consistent ascension of VL
are the major features indicating progression. Based on our results, it is tempting to suggest that continued selection of diverse progeny of viral variants (25) may not be sufficient to account for the ineffectiveness of existing CTL repertoires in containing viral replication and that the progressive immune dysregulation observed in HIV-infected patients may lead to (or result from) the induction and expansion of V␦1␥␦CTL. These CTL seem to be capable of contributing to immuno-pathogenesis through depletion of vital cell types involved in the adaptive immune response to infection. Interestingly, lack of full-blown AIDS in chimpanzees has been associated in part with the failure to develop MHC-unrestricted CTL in blood (55) as well as the absence of a Th1-to-Th2 cytokine shift (30). This clearly contrasts with the characteristic features of HIV infection in humans.
Finally, it is noteworthy that although this study was carried out with patients on HAART, MHC-unrestricted, CTL-medi-ated cytolysis of HIV-infected and bystander cells in individu-als both on and not on HAART (23, 24) has been reported; however,␥␦T-cell-mediated cytolytic activity was not investi-gated in those previous studies. It is not known at present whether different therapeutic regimens prescribed to HIV-1-infected patients and AIDS patients have any effect on CTL-mediated cytopathicity. Previous observations, together with the present data, clearly emphasize the need for further studies on this matter. Such studies could provide important insights on the pathogenic mechanisms involved in the progression of HIV infection and AIDS. Taken together, our data suggest that abnormal expansion of ␥␦ CTL, especially of the V␦1 subset, in HIV-1-infected patients may contribute to lysis of bystander CD4⫹T lymphocytes as well as other cells, leading
eventually to exacerbation of immunopathology and AIDS in these individuals.
ACKNOWLEDGMENTS
We thank the Canadian Institutes of Health Research for support. We thank Marc Dumont for advice on statistical analyses of the data and Micheline Patenaude and Isabelle Champagne for secretarial as-sistance.
REFERENCES
1. Ahmad, A., A. Ladha, E. A. Cohen, and J. Menezes.1993. Stable expression of the transfected HIV-1envgene in a human B cell line: characterization of gp120-expressing clones and immunobiological studies. Virology192:447– 457.
2. Anderson, R. W., M. S. Ascher, and H. W. Sheppard.1998. Direct HIV cytopathicity cannot account for CD4⫹decline in AIDS in the presence of
homeostasis: a worst-case dynamic analysis. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol.17:245–252.
on November 8, 2019 by guest
http://jvi.asm.org/
3. Aries, S. P., K. Weyrich, B. Schaaf, F. Hansen, R. H. Dennin, and K. Dalhoff. 1998. Early T cell apoptosis and Fas expression during antiretroviral therapy in individuals infected with human immunodeficiency virus-1. Scand. J. Im-munol.48:86–91.
4. Badley, A. D., J. A. McElhinny, P. J. Leibson, D. H. Lynch, M. R. Alderson, and C. V. Paya.1996. Upregulation of Fas ligand expression by human immunodeficiency virus in human macrophages mediates apoptosis of unin-fected T lymphocytes. J. Virol.70:199–206.
5. Bauer, S., V. Groh, J. Wu, A. Steinle, J. H. Phillips, L. L. Lanier, and T. Spies.1999. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science285:727–729.
6. Beckman, E. M., A. Melian, S. M. Behar, P. A. Sieling, D. Chatterjee, S. T. Furlong, R. Matsumoto, J. P. Rosat, R. L. Modlin, and S. A. Porcelli.1996. CD1c restricts responses of mycobacteria-specific T cells. Evidence for an-tigen presentation by a second member of the human CD1 family. J. Immu-nol.157:2795–2803.
7. Bienzle, D., F. M. Smaill, and K. L. Rosenthal.1996. Cytotoxic T lympho-cytes from HIV-1 infected individuals recognize an activation dependent, non-polymorphic molecule on uninfected CD4⫹lymphocytes. AIDS10:247–
254.
8. Boullier, S., M. Cochet, F. Poccia, and M. L. Gougeon.1995. CDR3-inde-pendent␥␦V␦1⫹T cell expansion in the peripheral blood of HIV-infected
persons. J. Immunol.154:1418–1431.
9. Brinchmann, J. E., G. Gaudernack, and R. Vardtdal.1991. In vitro replica-tion of HIV in naturally infected CD4⫹T cells is inhibited by rIFN
␣2and by
a soluble factor secreted by activated CD8⫹T cells but not by rIFN , rIFN␥,
or rTNF␣. J. Acquir. Immune Defic. Syndr.4:480–488.
10. Bruisten, S. M., P. H. Frissen, P. Van Swieten, P. R. Harrigan, I. Kinghorn, B. Larder, H. M. Weigel, E. De Vries, R. M. Regez, and J. H. Henrichs.1997. Prospective longitudinal analysis of viral load and surrogate markers in relation to clinical progression in HIV type 1 infected persons. AIDS Res. Hum. Retrovir.13:327–335.
11. Bucht, A., K. Soderstrom, T. Hultman, M. Uhlen, E. Nilsson, R. Kiessling, and A. Gronberg.1992. T cell receptor diversity and activation markers in the V␦1 subset of rheumatoid synovial fluid and peripheral blood T lympho-cytes. Eur. J. Immunol.22:567–574.
12. Bukowski, J. F., C. T. Morita, Y. Tanaka, B. R. Bloom, M. B. Brenner, and H. Band.1995. V␥2V␦2 TCR-dependent recognition of non-peptide anti-gens and Daudi cells analyzed by TCR gene transfer. J. Immunol.154:998– 1006.
13. Bukowski, J. F., C. T. Morita, and M. B. Brenner.1999. Human␥␦T cells recognize alkylamines derived from microbes, edible plants, and tea: impli-cations for innate immunity. Immunity11:57–65.
14. Casella, C. R., and T. H. Finkel.1997. Mechanisms of lymphocyte killing. Curr. Opin. Hematol.4:24–31.
15. Centers for Disease Control and Prevention.1992. 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. Morb. Mortal. Wkly. Rep.41:1–19. 16. Conti, L., G. Rainaldi, P. Matarrese, B. Varano, R. Rivabene, S. Columba, A.
Sato, F. Belardelli, W. Malorni, and S. Gessani.1998. The HIV-1 vpr protein acts as a negative regulator of apoptosis in a human lymphoblastoid T cell line: possible implications for the pathogenesis of AIDS. J. Exp. Med.187: 403–413.
17. Cosman, D., J. Mullberg, C. L. Sutherland, W. Chin, R. Armitage, W. Fanslow, M. Kubin, and N. J. Chalupny.2001. ULBPs, novel MHC class I-related molecules, bind to CMV glycoprotein UL16 and stimulate NK cytotoxicity through the NKG2D receptor. Immunity14:123–133. 18. Das, H., V. Groh, C. Kuijl, M. Sugita, C. T. Morita, T. Spies, and J. F.
Bukowski.2001. MICA engagement by human V␥2V␦2 T cells enhances their antigen-dependent effector function. Immunity15:83–93.
19. Forrester, J. M., L. S. Newman, Y. Wang, T. E. J. King, and B. L. Kotzin. 1993. Clonal expansion of lung V␦1⫹ T cells in pulmonary sarcoidosis.
J. Clin. Investig.91:292–300.
20. Frost, S. D., and C. A. Michie.1996. Lymphocyte dynamics, apoptosis and HIV infection. Trends Microbiol.4:77–82.
21. Gerencer, M., V. Burek, B. A. Crowe, N. P. Barrett, and F. Dorner.1998. The role of complement and gp120 specific antibodies in virus lysis and CD4⫹T
cell depletion in HIV-1 infected patients. Microb Pathog.25:253–266. 22. Gomez, A. M., F. M. Smaill, and K. L. Rosenthal.1994. Inhibition of HIV
replication by CD8⫹T cells correlates with CD4⫹counts and clinical stage
of disease. Clin. Exp. Immunol.97:68–73.
23. Grant, M. D., F. M. Smaill, and K. L. Rosenthal.1993. Lysis of CD4⫹
lymphocytes by non-HLA-restricted cytotoxic T lymphocytes from HIV-infected individuals. Clin. Exp. Immunol.93:356–362.
24. Grant, M. D., F. M. Smaill, and K. L. Rosenthal.1994. Cytotoxic T lympho-cytes that kill autologous CD4⫹lymphocytes are associated with CD4⫹
lymphocyte depletion in HIV-1 infection. J. Acquir. Immune Defic. Syndr. 7:571–579.
25. Grant, M. D., F. M. Smaill, D. P. Singal, and K. L. Rosenthal.1992. The influence of lymphocyte counts and disease progression on circulating and inducible anti-HIV cytotoxic T cell activity in HIV positive humans. AIDS 6:1085–1094.
26. Grant, M. D., F. M. Smaill, K. Lauri, and K. L. Rosenthal.1993. Changes in cytotoxic T cell repertoire of HIV-1 infected individuals: relationship to disease progression. Viral Immunol.6:85–95.
27. Grant, M. D., M. S. Weaver, C. Tsukos, and G. W. Hoffmann.1990. Distri-bution of antibodies against denatured collagen in AIDS risk groups and homosexual AIDS patients suggests a link between autoimmunity and im-munopathogenesis of AIDS. J. Immunol.144:1241–1250.
28. Groh, V., A. Steinle, S. Bauer, and T. Spies.1998. Recognition of stress-induced MHC molecules by intestinal epithelial␥␦T cells. Science279: 1737–1740.
29. Groh, V., R. Rhinehart, H. Sercist, S. Bauer, K. H. Grabstein, and T. Spies. 1999. Broad tumor-associated expression and recognition by tumor-derived ␥␦T cells of MICA and MICB. Proc. Natl. Acad. Sci. USA96:6879–6884. 30. Heeney, J., W. Bogers, L. Buijs, R. Dubbes, P. ten Haaft, W. Koornstra, H.
Niphuis, P. Nara, and V. Teeuwsen.1996. Immune strategies utilized by lentivirus-infected chimpanzees to resist progression to AIDS. Immunol. Lett.51:45–52.
31. Holoshitz, J., N. C. Romzek, Y. Jia, L. Wagner, L. M. Vila, S. J. Chen, J. M. Wilson, and D. R. Karp.1993. MHC-independent presentation of mycobac-teria to human␥␦T cells. Int. Immunol.5:1437–1443.
32. Imberti, L., A. Sottini, A. Bettinardi, M. Puotti, and D. Primi.1991. Selective depletion in HIV infection of T cells that bear specific T cell receptor V sequences. Science254:860–863.
33. Lang, F., M. A. Peyrat, P. Constant, F. Davodeau, J. David-Ameline, Y. Poquet, H. Vie, J. J. Fournie, and M. Bonneville.1995. Early activation of human V␥9V␦2 T cell broad cytotoxicity and TNF production by nonpep-tidic mycobacterial ligands. J. Immunol.154:5986–5994.
34. Legrand, E., I. Pellegrin, D. Neau, J. L. Pellegrin, J. M. Ragnaud, M. Dupon, B. Guillemain, and H. J. Fleury.1997. Course of specific T lymphocyte toxicity, plasma and cellular viral loads and neutralizing antibody titers in 17 recently seroconverted HIV type 1 infected patients. AIDS Res. Hum. Ret-rovir.13:1383–1394.
35. Mackewicz, C., and J. A. Levy.1992. CD8⫹cell anti-HIV activity: non-lytic
suppression of viral replication. AIDS Res. Hum. Retrovir.6:1039–1050. 36. Mittler, J. E., B. R. Levin, and R. Antia.1996. T cell homeostasis,
compe-tition and drift: AIDS as HIV-accelerated senescence of the immune rep-ertoire. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol.12:233–248. 37. Molinero, L. L., M. B. Fuertes, G. A. Rabinovich, L. Fainboim, and N. W.
Zwirner.2002. Activation-induced expression of MICA on T lymphocytes involves engagement of CD3 and CD28. J. Leukoc. Biol.71:791–797. 38. Morita, C. T., E. M. Beckman, J. F. Bukowski, Y. Tanaka, H. Band, B. R.
Bloom, D. E. Golan, and M. B. Brenner.1995. Direct presentation of non-peptide prenyl pyrophosphate antigens to human␥␦ T cells. Immunity 3:495–507.
39. Poccia, F., M. Malkovsky, M. L. Gougeon, M. Bonneville, M. Lopez-Botat, J. J. Fourine, and V. Clizzi.1997.␥␦T cell activation or anergy during infections, the role of nonpeptide TCR ligands and HLA class I molecules. J. Leukoc. Biol.62:287–291.
40. Sewell, A. K., D. A. Price, A. Oxenius, A. D. Kelleher, and R. E. Phillips. 2000. Cytotoxic T lymphocyte responses to human immunodeficiency virus: control and escape. Stem Cells18:230–244.
41. Shimonkevitz, R., C. Colburn, J. A. Burnham, R. S. Murray, and B. L. Kotzin.1993. Clonal expansion of activated␥/␦T cells in recent-onset mul-tiple sclerosis. Proc. Natl. Acad. Sci. USA90:923–927.
42. Spada, F. M., E. P. Grant, P. J. Peters, M. Sugita, A. Melian, D. S. Leslie, H. K. Lee, E. van Donselaar, D. A. Hanson, A. M. Krensky, O. Majdic, S. A. Porcelli, C. T. Morita, and M. B. Brenner.2000. Self-recognition of CD1 by␥␦T cells: implications for innate immunity. J. Exp. Med.191: 937–948.
43. Spencer, J., T. T. MacDonald, T. C. Diss, J. A. Walker-Smith, P. J. Ciclitira, and P. G. Isaacson.1989. Changes in intraepithelial lymphocyte subpopula-tions in coeliac disease and enteropathy associated T cell lymphoma (malig-nant histiocytosis of the intestine). Gut30:339–346.
44. Steinle, A., V. Groh, and T. Spies.1989. Diversification, expression, and␥␦ T cell recognition of evolutionarily distant members of the MIC family of major histocompatibility complex class I-related molecules. Proc. Natl. Acad. Sci. USA65:12510–12515.
45. Sutherland, C. L., N. J. Chalupny, K. Schooley, T. VandenBos, M. Kubin, and D. Cosman.2002. UL16-binding proteins, novel MHC class I-related proteins, bind to NKG2D and activate multiple signaling pathways in pri-mary NK cells. J. Immunol.168:671–679.
46. Tanaka, Y., S. Sano, E. Nieves, G. De Libero, D. Rosa, R. L. Modlin, M. B. Brenner, B. R. Bloom, and C. T. Morita.1994. Nonpeptide ligands for human␥␦T cells. Proc. Natl. Acad. Sci. USA91:8175–8179.
47. Tanaka, Y., C. T. Morita, Y. Tanaka, E. Nieves, M. B. Brenner, and B. R. Bloom.1995. Natural and synthetic non-peptide antigens recognized by human␥␦T cells. Nature375:155–158.
48. Tsubota, H., C. I. Lord, D. I. Watkins, C. Morimoto, and N. L. Letvin.1989. Cytotoxic T lymphocytes inhibit acquired immunodeficiency virus replication in peripheral blood lymphocytes. J. Exp. Med.169:1421–1434.
49. Uyemura, K., R. J. Deans, H. Band, J. Ohmen, G. Panchamoorthy, C. T. Morita, T. H. Rea, and R. L. Modlin.1991. Evidence for clonal
on November 8, 2019 by guest
http://jvi.asm.org/
tion of␥/␦ T cells in response to a human pathogen. J. Exp. Med. 174:683–692.
50. Walker, B. D., S. Chakrabarti, and B. Moss.1987. HIV-specific cytotoxic T lymphocytes in seropositive individuals. Nature328:345–347.
51. Walker, C. M., D. Moody, D. Stites, and J. A. Levy.1986. CD8⫹lymphocytes
can control HIV replication in vitro by suppressing viral replication. Science 234:1563–1566.
52. Walker, C. M., A. L. Erickson, F. Hsueh, and J. A. Levy.1991. Inhibition of HIV replication in acutely infected CD4⫹cells does not involve cytotoxicity.
J. Virol.65:5921–5927.
53. Wallace, M., A. M. Scharko, C. D. Pauza, P. Fisch, K. Imaoka, S. Kawabata, K. Fujihashi, H. Kiyono, Y. Tanaka, B. R. Bloom, and M. Malkovsky.1997.
Functional␥␦ T lymphocyte defect associated with human immunodefi-ciency virus infections. Mol. Med.3:60–71.
54. Wu, J., V. Groh, and T. Spies.2002. T cell antigen receptor engagement and specificity in the recognition of stress-inducible MHC class I-related chains by human epithelial␥␦T cells. J. Immunol.169:1236–1240.
55. Zarling, J. M., J. A. Ledbetter, J. Sias, P. Fultz, J. Eichberg, G. Gjerset, and P. Moran.1990. HIV-infected humans but not chimpanzees have circulating cytotoxic T lymphocytes that lyse uninfected CD4⫹cells. J. Immunol.144:
2992–2998.
56. Zinkernagel, R. M.1988. Virus triggered AIDS: a T-cell-mediated immuno-pathology. Immunol. Today9:370–372.