Recruiting care homes to a randomised controlled trial
Full text
Figure
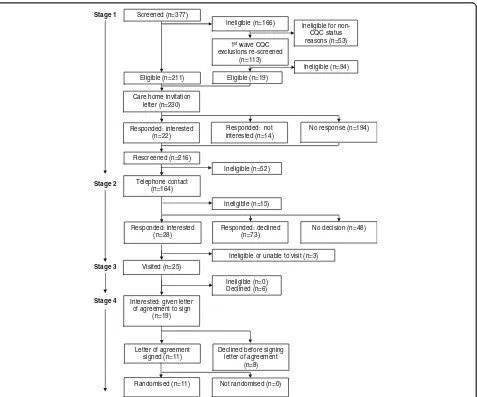
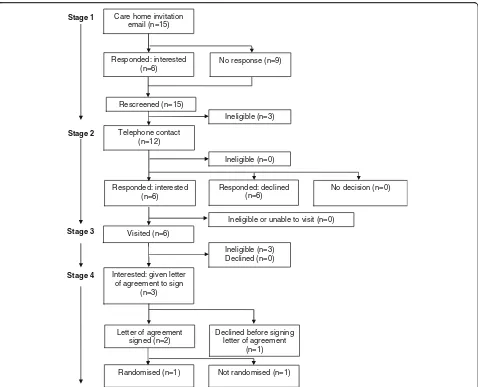

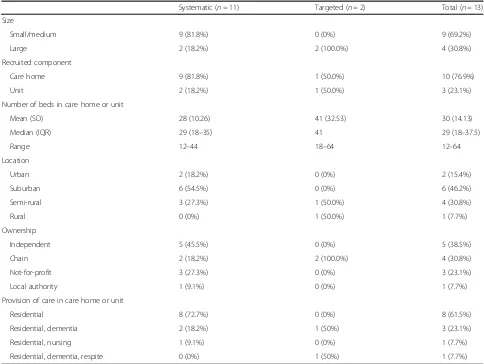
Related documents
AEs: Adverse events; BIC: Bayesian information criteria; BNF: British national formulary; CONSORT: Consolidated standards of reporting trials; CRF: Case report form; CTU:
CBT: Cognitive behaviour therapy; CC: Care Coordinator; CMHTs: Community mental health teams; CONSORT: Consolidated Standards of Reporting Trials cluster trial extension standards;
DEMQoL: Dementia quality of life tool; EMS: Elderly Mobility Scale; ENRICH: Enabling Research in Care Homes; EQ-5D: EuroQoL five dimensions; FAC: Functional Ambulation
CQC: Care Quality Commission; ENRICH: National Institute for Health Research: Enabling Research in Care Homes Programme; GP: general practitioner; MDS: Minimum Data Set; NHS:
AE: Adverse event; AWI: Adults with Incapacity (Scotland) Act 2000; CH: Care home; CHaRT: Centre for Healthcare Randomised Trials; CI: Chief Investigator, confidence interval; CRF:
B-IPQ: Brief Illness Perception Questionnaire; BRTC: Bristol Randomised Trials Collaboration; CCG: Clinical Commissioning Group; CI: Chief investigator; CONSORT: Consolidated
CAT: The COPD Assessment Test; CCQ: The Clinical COPD Questionnaire; CME: Continuing medical education; CONSORT: Consolidated Standards of Reporting Trials; COPD: Chronic
CALPAS: California Psychotherapy Alliance Scales; CI: chief investigator; CMHT: Community Mental Health Team; CONSORT: Consolidated Standards of Reporting Trials; CPA: Care