Copyright@ 1968 AmericanSocietyforMicrobiology Printed in U.S.A
Specific Role of
Each Human
Leukocyte
Type
in
Viral
Infections
II.
Phytohemagglutinin-treated
Lymphocytes
as
Host Cells for Vesicular Stomatitis
Virus
Replication
In
Vitro
ROBERT EDELMAN AND E. FREDERICK WHEELOCK
Departments of Preventive Medicine andMedicine, Case Western Reserve UniversitySchool of Medicine, and UniversityHospitals, Cleveland, Ohio 44106
Received for publication 12February1968
Themitogenicagent, phytohemagglutinin (PHA), addedtohuman mixed
leuko-cytecultures andtolymphocytecultures converted smalllymphocytesinto
lympho-blasts and increased lymphocyte susceptibilitytovesicular stomatitis virus (VSV).
Maximum virus yieldswere30-to1,000-fold higher inPHA-treated thanincontrol
cultures. VSV replicated to peak titers before lymphocytes were morphologically
transformed by PHA, and virus titers fellas lymphoblastdestruction began. PHA
neitherinduced significantVSVreplicationinpolymorphonuclear leukocyte cultures,
norincreased the large virus yields inmonocyte cultures. The treatment of PHA
withheat, digestiveenzymes, rabbit anti-PHA serum and serial dilutions failedto
dissociate thatportionof the PHAextractresponsiblefor the conversion of
lympho-cytesintovirus-susceptiblecellsfromthose componentsresponsiblefor
leukoagglu-tinationorlymphocyte transformation.
Numerousnonviral agents have
been shown
toenhance
animal virus
replication
inmonolayer
cell cultures (2, 3, 8, 10,
18, 25).
Inhumanleuko-cyte
cultures,
phytohemagglutinin (PHA),
an extractof
theredkidney
bean Phaseolusvulgaris,induces
thereplication
of
someviruses,
such asherpes
simplex (13)
and mumps(4),
whichother-wise would
notreplicate,
andalso increases
theyield of vesicular stomatitis virus (VSV)
(5)
and 17Dyellow fever
virus(Wheelock
andEdelman,
unpublished data),
whichotherwise would
repli-cateto
low
titer.PHA
agglutinates
humanleukocytes
(11, 14)and produces a
variety
of alterations inlympho-cytes, including:
(i)
stimulation of ribonucleicacid (RNA) and
protein synthesis
(7, 11); (ii)transformation into blast cells (14, 27); (iii)
stimulation of
deoxyribonucleic
acid (DNA)replication
andmitosis
(7,
14,27) and (iv)induc-tion
of
interferonproduction (24).
PHAhas beenshown to enhance
lymphocyte-mediated
immuneresponses invitro (11), and eithertoenhance (22)
orsuppress
(23)
immune responses invivo.
The presentstudyinquires into the mechanism
by
which PHA enhances VSVreplication
inhuman leukocyte cultures. The
relationship
ofPHA-induced leukoagglutination, blast cell
for-mation, and enhancement
of
VSVreplication
is described, and the white cell types which
undergo cytopathic alterations in PHA-treated
virus-infected cultures
are identified. The smalllymphocyte, acell in which VSV
replicates
tolowtiter, is
shown to betransformed by
PHAinto
alymphoblast, a cell
which
can support VSVreplication
tohigh titer.
MATERIALS AND METHODS
Virus. VSV, Indiana serotype, was obtained from Frederick C. Robbins. The virus seed was prepared
by replication in a PPLO (pleuropneumonia-like
organism) -free L-cell strain ofmouseskinfibroblasts;
supernatant fluids were quick-frozen and stored at -70 C. The VSV seed contained 3.2 X 108 tissue culture infective doses, 50% (TCID50) per ml, when titered in L-cellmonolayercultures.
Culture medium. Humanleukocytes and theL-cell
monolayers used for virus assay were cultured in Eagle's minimum essential medium (MEM) supple-mentedwith: TryptosePhosphate Broth,4%(Difco);
unheated fetal calf serum, 10%;sodium bicarbonate,
1.75 g/liter; glutamine, 1%; penicillin and strepto-mycin. All cell-culture vessels were gassed with 5%
CO2inair and incubatedstationaryat37C.
Preparation of mixed leukocyte cultures. The venous blood of healthy adults was drawn into
plastic disposable syringes and then placed in glass
440
on November 11, 2019 by guest
http://jvi.asm.org/
vessels containing 0.2 ml of phenol-free heparin (Upjohn) for each 10to14ml ofblood.After incuba-tion at 37 C for 30 to 60 min, the leukocyte-rich plasma upper phasewasaspirated and centrifuged at 225 X gfor 10 mintosediment theleukocytes. The plasma supernatant fraction, now containing prin-cipally platelets, wasaspirated. The cell pellet in the bottom of thetube wasgently resuspended inalarge volume ofphosphate-bufferedsaline(PBS),centrifuged
again, and theremaining plasma, heparin, and plate-lets were drawn off with the supernatant fluid. The washedleukocytes were thenresuspended inculture medium. Theyield ofleukocytes from 50 ml ofvenous blood averaged 150 X 106 cells; more than 99.5%
were viable. The procedures employed in obtaining
leukocyte cultures did not significantly alter the cell differential count from that of the donor's whole blood, by using the morphological criteria outlined by Wintrobe (26). Erythrocytes were present, and their quantity varied from one-tenth to 10 times the leukocyteconcentration.Plateletswereabsent, having
been removed during preparation of the leukocyte
suspensions. Cell cultures which were prepared
directlyfromperipheralbloodasdescribed aboveare
designated mixed leukocyte cultures. In mixed
leu-kocyte cultures treated with PHA, the identification ofblast cells and oflymphocytes in transformation was based on the morphological criteria of Yoffey
et al. (27), and both cell types will be referred to
collectively as "blast cells" or "lymphoblasts" to
distinguishthem from smalllymphocytes (seeFig.2).
Unless indicated otherwise, a given experiment was
performed with freshly preparedcells from a
single
healthy adult donor, although donors of human
leukocytesvaried fromexperimenttoexperiment.
Determination of cell viability. Immediately after
being placedinculture,andatselected intervalsduring
thecourseof
experiments
describedbelow,
the leuko-cytes were counted in a hemocytometer chamberemploying an erythrosin Bvital dye exclusion
tech-nique(15). Eachcountrepresentedtheidenfication of 100to250cells whichexcludeduptakeoferythrosinB vital dye. Cell concentrations in this study are
ex-pressedin terms ofviable
leukocytes
per milliliter ofculture medium.
Leukocyte morphology. Samples of leukocyte
cultures were
prepared
forstudy
ofcell morphologyand for differential counts in the
following
manner. Samples (1 to 2 ml) ofleukocyte
cultures werecentrifuged at200 X gfor10 min,the mediumwas
aspirated, and the
leukocyte resuspended
in 0.1 to0.2 mlof medium bygentleagitation of thetube. A large drop of the cell suspension was
carefully
dis-bursed betweentwoglasscover slips (15 X 15 mm), airdried, andstained withWright'sstain. Atotal of 500 cellswascounted from eachculture;
atleast 100 leukocytes on eachof fourcoverslips
wereidentified as tocell type. Three to five separateareas oneach coverslipwereexaminedtominimize the effect of cellaggregationandunequalcelldistribution.Leukocytes
showingevidence of cell
damage
werenotcounted,andtherareleukocytethat
appeared
intactbutnotidenti-fiableas tocell typewas notincluded in thedifferential count. The differential counts were reproducible;
there wasclose agreementbetween each offive sets of 100cells countedfrom each culture, and there was no significant variation when the same cover slip was countedontwoseparateoccasions.
Separation of leukocyte fractions by using silicone-treatedglass-beadcolumns. Thewhite blood-cell types were separated on the basis of their differential adherence to and elution from siliconized glass bead columns (17). Lymphocytes did not adhere to glass beads, whereaspolymorphonuclear (PMN) leukocytes and monocytes did adhere and wereselectivelyeluted by buffered ethylenediaminetetraacetic acid (EDTA) in saline.After removal fromaglassbeadcolumn,the cell fractionswerewashedonce toremoveautologous plasma or EDTA, resuspended in culture medium, adjusted to acell concentration of 106cells/ml, and samples of each of thesuspension cultures were taken fordifferential leukocyte counts. Loss of cell viability during column separation was less than 0.5%.
Virus growth curves and infectivity assay. After VSVinoculation into leukocyte cultures at a virus-to-cell multiplicity of 1, the cultures were placed in a stationary position for 1 hr at 37 C to allow virus absorption. The leukocytes were then washed by 1
cycle of centrifugation at 225 X g for 10 min; the
cellular pellets were resuspended in fresh culture mediumattheoriginal volumeandincubatedat37 C. PHAwas notaddedtothe culturesthat had earlier received PHA, since preliminary studies indicated there was no further virus enhancement when PHA wasaddedagain after the cell wash. Attimedintervals, the sedimented cells and cells adhering to the glass vessels were resuspended by pipetting, and 1-ml samples of the infected cultures wereremoved, frozen rapidly, and stored at -70 C. Immediately before virus assay, the frozen samples were thawed and frozen rapidly twice to disrupt cells, a procedure whichproducednodetectable loss of VSVinfectivity.
Infective virus was assayed by the tube dilution method in monolayers of human diploid fetal lung cellsorintheL-cellstrain ofmouseskinfibroblastsin screw-cap glass culture tubes (16 X 125 mm). Both cell linesareequallysusceptible to VSV; PHA does not influence the replication of VSV in thecelllines
used for virus assays (5). Titrations employed fiveto
eight tubes per 10-fold dilution, and the 50% end
point was calculated by the method of Reed and Muench (19). Differences in titer of more than 0.5
log1o (3.2-fold) wereconsidered tobesignificant.
Assay of human interferon. Samples of leukocyte cultures obtained for interferon assaywerecentrifuged to removecells,andthe culturemediumwasstoredat 4 C. Interferonwas consideredtobe present whena 1:2dilution (or more) of theculture medium protected monolayer cultures of human fetal lung fibroblasts against Sindbis viruschallenge (24).
PHA. Lyophilized commercial PHA-P (Difco
lot % 475973) was dissolved in PBS (10 mg/ml),
sterilized by filtration, and storedat 4 C. A freshly
prepared stock of PHA was used every 4weeks. A 25- to
50-,gg
amount of PHA inan 0.05-ml volume was added to each 1-ml amount of leukocytesuspension containing 106
cells/ml.
The time ofaddi-tion of PHA with respecttoVSVinoculationwas not
441
on November 11, 2019 by guest
http://jvi.asm.org/
crucial; virus enhancement occurred if PHA was
added either withVSVor5daysbefore virus
inocula-tion.
Preparation and titration ofanti-PHA serum. An
adult white female rabbit was injected both sub-cutaneouslyandintramuscularlywith 25mgof PHA insalineand completeFreund's adjuvant, onceeach week for 5 weeks; one-half of the final inoculating dose,free ofadjuvant,wasadministeredintravenously. Serum-obtained1week after thelastinjection-was
heated (56 C, 30min) to remove leukoagglutinating activity. Serialdilutions ofserum werethenincubated in PHA (50 ,g of PHA/ml of culture medium) at
37C for1 hr; leukocytes wereaddedtothe incuba-tion mixture (106 cells/mnl, finaldilution), kept
over-nightat4 C, and the whitecellaggregates werethen
gradedastosize andnumber.Anantiserumdilution of 1:40 inhibited leukoagglutination by PHA.
Data showing the effect of anti-PHA serum on
PHAactivity (Table 1) wereobtained inthe follow-ingmanner. Anti-PHAserum(0.2 ml) wasincubated
with2.0 ml ofasolution containing 50,ugofPHA/ml
ofculture medium for 1 hr at 37 C. The flocculent
precipitate thatformedwasthenremovedby
centrifu-gation (5,000 rev/min for 30 min), the supernatant
fluidaddedto anequalvolume of mixedleukocytes in culture medium, and the residual PHA activity determinedaspreviouslydescribed. PHApreincubated with heat-inactivated serum from a nonimmunized rabbitservedasacontrol.
RESULTS
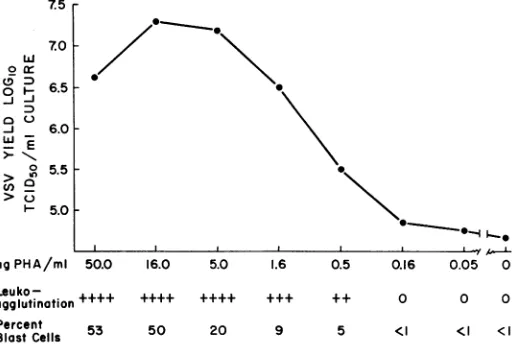
Correlationoftheleukoagglutinating, blastogenic,
and VSV-enhancing activities of PHA in mixed
leukocyte cultures. To elucidate the mechanism
of PHA-induced enhancementof VSVreplication
inhuman mixedleukocytecultures (5),aseries of
experiments was performed to correlate the
various effects of PHA on leukocytes and on
virusreplication.
7.5
w
C g D)
_i E
D
_ L) >i->0
>in
7.0
6.5 6.0 5.5 5.0
,ug PHA/mI 50.0 16.0 5.0 1.6 0.5 0.16 0.05 0
PHA was added in
0.5-logio
(3.2-fold)incrementstoaseriesof mixed leukocyte cultures
containing106cells/ml.Thedegree of
leukoagglu-tination (graded 0 to +++ +, shown on the
abscissa, Fig. 1) was based onthe number and
size of microscopic white-cell aggregates in
un-infectedculturesincubatedat37Cfor16 hrafter
the addition of PHA. The uninfected cultures
wereharvested 64 hr after the additionof PHA,
and thepercentage of blast cells (also shownon
theabscissa) wascalculated fromatotal count of
1,000 cells from eachculture.Additional leukocyte
cultures, prepared in an identical fashion to
cultures described above, received VSV 16 hr
after the addition ofPHA,and thecultureswere
washed1hrafter virusinoculation(seeMaterials
and Methods). The virus titersexpressedon the
ordinate inFig. 1 arethosepresentat57hrafter
VSV inoculation, which is a time on the virus
growthcurveswhen the differences between VSV
yields in PHA-treated and control cultures are
expectedtobeatamaximum (5).
With decreasing doses of PHA, starting at a
concentrationof50,g/mlofculture, therewas a
linear decrease in PHA-induced VSV enhance-ment, leukoagglutination, and blast-cell
forma-tion (Fig. 1); all oftheseeffectsdisappearedatthe
same terminal dilution. Thelower limit ofPHA
activity was detected attheconcentration of 0.5
,Mgof
PHA/ml.
Additional studiescorrelating
thedose of PHA with the formation of interferonin
mixed leukocyte cultures showed that interferon
was detectable inleukocyte culturestreated with
a minimum of1.6 ,g of PHA/mlin one
experi-mentandwith aminimumof 5.0 Mg ofPHA/ml
ina secondexperiment.
Inadditiontoalteringtheconcentration of the
Leu ko +
agglutination + Percent 53
Blast Cells
++++ ++++ +++ ++ 0 0 0
50 20 9 5 (I <1 <I
FIG. 1. Correlation of the leukoagglutinating, blastogenic, and vesicular stomatitis virus (VSV)-enhancing
activitiesofphytohemagglutilnin inhumanleukocyte cultures.
0~~~~~
I\
--\0
0~~~Ya 442
on November 11, 2019 by guest
http://jvi.asm.org/
[image:3.487.127.387.452.624.2]crude PHAextract, othermeans wereutilized to
eliminate selectively one or more of the
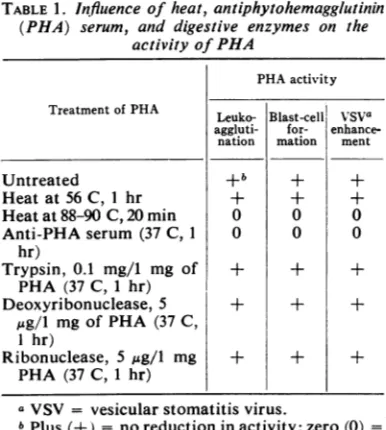
PHA-induced effects. Table 1 summarizes the effect of
treatment of PHA with heat, anti-PHA serum,
and several digestive enzymes on PHA-induced
leukoagglutination, blastogenesis, and virus
en-hancement in mixedleukocyte cultures. The three
PHA-induced effectsweredestroyed by heat (88to
90 C for 20
min)
and were neutralized withserumfromaPHA-immunerabbit(37 C for1hr),
whereas heat(56 C for 1 hr), trypsin, and nucleases
had no effect. The ability of PHA to induce
interferon was abolished by prior treatment of
PHA with heat(88to90C for 20
min)
and withanti-PHAserum.These resultssuggestthat
blast-cell transformation,
leukoagglutination,
andenhancement of VSV replication areinterrelated
events.
Effectof VSVoncellmorphologyand survival in
PHA-treated mixed leukocyte cultures. To
elucidatefurther thepossible relationshipbetween
virus enhancement and blast-cell formation, the
effect of VSV on leukocyte morphology and
survival was studied, and the cell type or types
destroyed as a consequence of infection were
determined (Table 2).
A freshly prepared mixed leukocyte culture
containing 2 x 10i
cells/ml
was divided intofour cultures. Onewasinoculatedwith PHA and
VSV simultaneously, while a companion culture
received only PHA; the two remaining cultures
were treated with culture medium instead of
PHA (controls), and one of these cultures was
TABLE1. Influence of heat,antiphytohemagglutinin (PHA) serum, and digestive enzymes on the
activityofPHA
PHAactivity
Treatment ofPHA VSVa
Leuko- Blast-cell
aggluti- for- enhance-nation mation ment
Untreated +b + +
Heat at56 C, 1 hr + + +
Heatat88-90C,20 min 0 0 0
Anti-PHAserum (37 C, 1 0 0 0
hr)
Trypsin, 0.1 mg/i mg of + + + PHA (37 C, 1 hr)
Deoxyribonuclease, 5 + + +
,4g/l
mgofPHA (37 C,I hr)
Ribonuclease, 5 jsg/l mg + + + PHA (37 C, 1 hr)
aVSV = vesicularstomatitis virus.
bPlus(+) = noreduction inactivity;zero(0) =
absence of detectable activity after treatment.
inoculated
with
VSV.After
1 hrof
incubation at37C, the
cells
werewashed
once and thecultureswere
incubated
in a stationaryposition at 37 C for 3additional
days to allow adequate time forformation
of lymphoblasts
and to permit VSV toreach
maximum titer
(5). Three days after VSVinoculation, samples
of
each culture were takenfor cell
viability
counts(cell survival), for
differen-tial
counts(cell
morphology), and for virus assay.Differential
counts wereperformed
on a total of 500cells
from each culture.
Although
leukoagglu-tination did
not permit accurate cell counts 1 dayafter
theaddition of
PHA, by 2 or 3days most ofthecell aggregates had partially
dissociated
andreproducible cell
counts could beobtained.
Lymphoblast transformation was noted after
an
incubation period
of 72 hr in all cultures (Table 3). In the uninfected PHA culture, therewere 20
times
more lymphoblasts than in theinfec-ted PHA
culture,
and 40 times morelymphoblasts
thanin either
of
thecontrolcultures.
Acompari-son
of
the twoPHA-treated cultures
revealed a 95% loss of blast cells
in thevirus-infected
cultureat 72
hr; this
loss ranged
between 75 and 97% inadditional experiments.
There was no detectableloss oflymphoblasts in the VSV-infected control
culture
ascompared with
theuninfected control
culture.
Incontrast tothe marked destruction
of lymphoblasts in the infected PHA culture, thereTABLE2. Effect ofvesicularstomatitis virus (VS V) on cell survivalin mixed leukocyte cultures
Totalno.of each cell typea/mlof cultureX 10'
Treatment 0hr 72 hr
(Tciteo/ml)b
P+L B P+L B
+M +M
Control
Uninfected 2,000 0 760 5.3
Infected 2,000 0 710 6.4 2.3 X 104 PHAc
Uninfected 2,000 0 290 210
Infected 2,000 0 263 11 6.8 X 105
a Total number of each cell type was obtained by multiplying the percent differential count for each culture bythetotal number of viable leuko-cytes in that culture at either the time of VSV inoculation
(0
hr) orat 72 hrafter VSV inoculation (72 hr). Symbols P + L + M = combinedwhite cell fractions of polymorphonuclear leukocytes(P), lymphocytes (L), and monocyte-macro-phages (M). Lymphoblasts (B) are listed sepa-rately.
b Assayed 72 hr after virus inoculation.
-Culture was inoculated with
phytohemagglu-tinin (PHA) and VSV simultaneously.
443
VOL.
on November 11, 2019 by guest
http://jvi.asm.org/
[image:4.487.242.440.380.619.2] [image:4.487.41.234.420.635.2]was only a minimal loss of
leukocytes
in thecombined nonblast cell fraction (P + L + M;
Table 2) of both virus-infected cultures. This cell
loss,
equivalent to a6.5%- and a9.3%/o
reduction,
respectively,
in the control and PHA-treatedcultures,
could
be attributedtocytopathic
effectsin the monocyte-macrophage
fraction
of themixed
leukocytes
(6). PHA treatment wasas-sociated with a
30-fold
enhancementof
the virustiter
compared
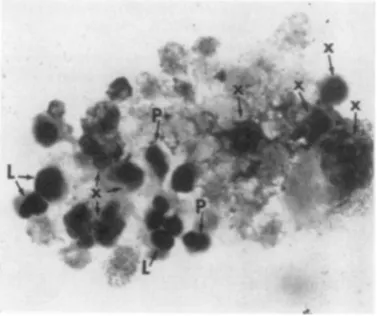
tothecontrol.Figure 2 shows
lymphocyte transformation
inmixed leukocyte cultures treated
with PHAfor
3days;
Fig.
3illustrates
cytopathic alterations
inasimilar
culture 3days
after thesimultaneous
inoculation of
both VSV and PHA. Thesmall
lymphocytes
andpolymorphonuclear
leukocytes
werewell preserved in the
infected
culture(Fig. 3),
but there were
fewer
intacttransformed
lympho-cytes present as
compared
with theuninfected
culture
(Fig.
2).Monocyte-macrophages
are notshown in either
Fig.
2 orFig.
3.Theresults presented above suggest that, asa
consequence
of
infection,
PHA-stimulated
lymphocytes
were destroyed either before theirtransformation into
blast cells orshortly
there-after,
and that increased virusyields
in thesePHA-treated
mixed
cultures may beattributed
to VSV
replication
in thetransforming
lympho-cyte
fraction.
Studies were next undertaken toelucidate
thetemporal
relationship
betweenFiG. 2.
Photomicrograph
ofanoninfected
keukocyte
culture treated with phytohemagglutinin for 3 days;
lymphocytes (L), lymphocytes in transformation (T),
andlymphoblasts (B). Noattemptismade in the text
to distinguish between lymphoblasts and transforming
lymphocytes, and both cell types are referred to
col-lectivelyas "blast cells" or "lymphoblasts." Wright's
stain,
>X
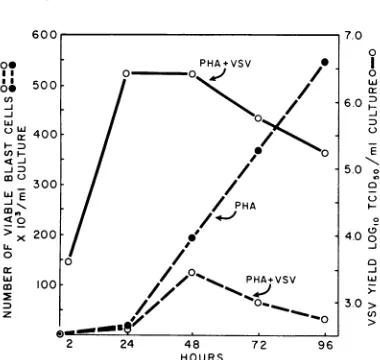
1,924.VSVreplication and lymphoblast production and
destruction.
Duplicate
mixedleukocyte
cultures wereprepared, treated with PHA for several minutes,
and then one culture was inoculated with VSV.
At 24, 48,
72,
and 96hr after VSVinoculation,
determinations were made of the total numbers
oflymphoblasts present in each of the cultures
by utilizing
methods outlined above. A virusgrowth curve was constructed from samples of
the VSV-infected cultures. In the uninfected
culture, lymphocyte transformation was first
detected at 48hr after theaddition ofPHA, and
increasing numbers of blast cells appeared
between 48 and 96 hr (Fig. 4). Bycontrast, in the
VSV-infected culture, fewer blast cells were
detected at 48hr, and there was a steady decline
in the number of
transformed
cells over thesubsequent 48 hr. Excess blast-cell loss in the VSV-infected culture (as compared with the
control culture) was first apparent 24 hr after
peak virus titers were reached, and virus titers
declined as the loss of blast cells increased
between 48 and 96 hr.
These results indicate that VSV can replicate
peak titers before lymphocytes are
morpho-logically transformed into
lymphoblasts,
andthat virus titers fall shortly after lymphoblast
destruction begins.
Effect
of PHA on the replication of VS V inlymphocyte cultures. The results of the studies
presented thus far clearly implicated the
PHA-stimulated lymphocyte in the enhancement
FIG. 3. Photomicrograph of a mixed leukocyte culture3daysafterinoculation withvesicularstomatitis virus and phytohemagglutinin; lymphocytes (L), polymorphonuclear leukocytes (P), and disrupted leukocytes (X) which cannot be precisely identified. No macrophages orblast cells can be seen. Wright'3 stain, X 1,924.
J. VIROL.
on November 11, 2019 by guest
http://jvi.asm.org/
[image:5.487.55.245.384.568.2] [image:5.487.259.449.420.578.2]INFECTINOS
0O PHA+VSV * l
0-500 /
<2
/ 6.z0 300; /// 5.0 .
o X 400 ;
cn6.-:
eD~~~~~~~100/5_PH.0 >
4O00
2x0 24 48 209
0~~~~~~~~~~~~~~50
p > 100-/
PHA(vsV
4.0*JPH-tmuae
PHAphcyte
uneg yoD O.~~~~~~0. 30>
z 0 cn
2 24 48 72 96
HOURS
FiG. 4.
Temporal relationship
betweet virusreplica-tion, lymphocyte
transformation,
and lymphoblastdestruction in mixed leukocyte cultures treated with
phytohemagglutinin
(PHA). VSV = vesicularstoma-titisvirus.
phenomenon
previously
noted in mixedleuko-cyte cultures
(5).
Thecorrelation
of virusen-hancement andblast-cell
transformation, together
with the
observation
that thelarge
majority
ofPHA-stimulated
lymphocytesundergo
cyto-pathic
alterations in infectedcultures, suggested
that VSV
replicates
in this celltype.
Therefore,
interest was focused next on the effects of PHA
in
cultures
ofsmall
lymphocytes.
Lymphocyte
cultures wereprepared
by
passageof mixed
leukocytes through
silicone-treated
glass-bead
columns
according
to thetechnique
outlined
inMaterials
and Methods.Duplicate
lymphocyte
cultures
werecomposed
of
99.7%
small
lymphocytes
and 0.3%
PMNleukocytes.
In
addition, duplicate
mixed
leukocyte
culturescontaining 30%
lymphocytes,
derived
from asample of
the samebatch ofbuffy-coat cels
thatserved as the source of
lymphocyte
cultures,were established without passage
through
theglass-bead
column. After
thecultures
wereadjusted
to 10 x106 cels
in a 10-mivolume,
PHA was added to one culture of each of the
two
paired
sets of cultures.After
16hrof incuba-tion at 37C,
asample
wasobtained
from eachculture
forinterferon
assay. VSV was theninoculated
into the fourleukocyte
cultures,and
virus
growth
curves were constructed asde-scribed above.
Figure
5 illustrates thereplication
of VSV inpaired
cultures oflymphocytes
and of mixedleukocytes.
inthe presenceandabsence of PHA.Intheuntreated mixed
leukocyte
culture,
apeak
virus titer Of
10"'"
TCID,5/Ml
was reached at15 hr after virus
inoculation, whereas,
in thePHA-treated mixed leukocyte culture, a
maxi-mum titer of 107 '
TCID50/ml
was achieved at24 hr,
which
represents a100-fold
increase invirus yield. Significantly, the maximum virus
titer in the PHA-lymphocyte culture (107 50
TcID50/ml)
was equal to, orslightly
greaterthan, the
maximum titer
in the PHA-mixedleukocyte culture, and the time course of virus
replication in
both PHA-treatedcultures
weresimilar.
There was nosignificant
VSV replicationin the non-PHA-treated lymphocyte
culture.
New
virus
wasfirst detected
6 hr after VSVinoculation in the untreated
mixed culture and
after
8 hrin
both PHAcultures.
Theextension
by
2hrof
theviral eclipseperiod
inPHA-treatedcultures, together
with
additional studies showing
extensions of
aslong
as 10 hr in PHAcultures,
maybe attributed to the viral inhibitory effect
of
interferon produced by lymphocytes in response to PHA.
Interferon
wasdetected
inboth
PHA-treated
cultures but
notin theuntreated
cultures.
It
is evident, however,
that the level of antiviralactivity
was insufficient to prevent VSV fromreplication
tohigh
titers in PHA-treatedlympho-cytes.
VSV enhancement in PHA-treated mixed
leukocyte
culturescanbeattributedtothepropa-gation of
VSV inPHA-stimulated
lymphocytes,
and
this
cellfraction
isincapable
ofsupporting
significant
virusreplication
without PHA(Fig.
5).
, PHA+LYMPHOCYTES (99.7%) /
_j ~~~~0
D)
/
6 -INOCULUM /PHA+MIXED CULTURE
o /
8/
E
/ /-MIXED CULTURE
25
_jjo
1.
/ ^4LYMPHOCYTES
(99.7%)C,)
O 2 4 6 8 15 24 48
HOURS AFTER VSV INOCULATION
FIG. 5. Effect ofthe additionofphytohemagglutinin (PHA) on vesicular stomatitis virus (VSV) replica-tion inboth humanmixedleukocyte cultures
(contain-ing 30% lymphocytes) and lymphocyte cultures
(containing 99.7% lymphocytes). Each culture
con-tained approximately 10 leukoyctes at the time of virusinoculation.
VOL.
2,
1968on November 11, 2019 by guest
http://jvi.asm.org/
[image:6.487.41.231.61.241.2] [image:6.487.245.437.376.590.2]AND
WHEELOCK
Previous studies have shown that, in the absence
of PHA,
VSVreplicates principally
in themonocyte-macrophage fraction of mixed
leuko-cyte cultures (6).
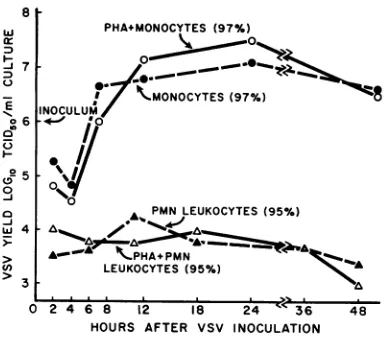
Effect
of PHAon VSVreplicationinmonocytesand polymorphonuclear
leukocytes.
To excludethe
participation of the other circulating
whiteblood cell
typesin
the enhancementphenomenon,
predominantly
purecultures of
PMNleukocytes
and
of
monocytes wereprepared by using
sili-conized glass-bead
columns (17). Theexperi-mental procedures were
identical
to thosedescribed
above. Monocytecultures
contained
97% monocytes and 3% PMN
leukocytes, and
the PMN
leukocyte cultures
werecomposed of
95
%
PMNleukocytes
and
5%
small
lymphocytes.
Figure
6illustrates
theeffect
of
PHA on VSVreplication
innearly
purecultures
ofboth
mono-cytes and PMN
leukocytes.
VSVreplicated
tohigh maximum titer in
theuntreated
monocyteculture (10715
TcID5o/ml).
Theslightly higher
maximum titer
in the PHA-monocytecultures
(107.50
TCID5o/m1)
waswithin
the error oftitration.
There was nosignificant virus
replica-tion in
the PMNleukocytecultures, whether
ornot the cultures were treated with PHA. PHA
neither induced virus
replication in PMN leuko-cytecultures
norenhanced
virusyields in
monocyte
cultures.
Interferon
was notdetected
in either the PHA-treated
monocytes or PMN leukocyte cultures.DIscussION
The
presentstudy
demonstrates thatsignificant
VSV
replication
does
not occurin small
lympho-8
w
Ir
]7
o6
05 CT
-J >w4
>3
PHA+MONOCYTES (97%)
I 'HMONOCYTES( 9;°/e)
INOCULUMX
.
A PMNLEUKOCYTES (95%)
*--A .PHA+PMN~MN
LEUKOCYTES (95%)
0 2 4 6 8 12 18 24&' 36 48
HOURS AFTER VSV INOCULATION
FiG. 6. Effect of the addition ofPHA on VSV replicationinhwnanmonocytecultures(97%monocytes)
and in polymorphonuclear (PMN) leukocyte cultures (95S% PMN kukocytes). Virus yieldis expressed in termsofTCID5o/Ml ofculturecontainingapproximately 106leukocytesatthetimeofvirusinoculation.
cytes in vitro except when these cells are
stimu-lated by PHA. Furthermore, the previously
reported enhancement of VSV yields in human mixed leukocyte cultures by PHA (5) can be
ascribed
to the induction of virusreplication
inlymphocytes that comprise approximately 30%
of the total cell population of mixed leukocyte
cultures.
The
addition of
PHAfailed
toinduce
significant
VSV
replication
in PMNleukocyte
cultures or toincrease
thehigh
virusyields
in monocytecul-tures. These
findings give
further support to theconcept that
PHA-stimulated lymphocytes play
the
key role
in the virus enhancement phe-nomenon. The PHA-mediated conversion oflymphocytes into cells capable of supporting
VSV
replication
occurswithin
8 to 16hr,
since
newly
synthesized
virus is first detected 8 to 16 hrafter
thesimultaneous
inoculation of
lympho-cyte
cultures with
VSV and PHA. Peak VSVtiters
arereached
at 24 to 48hr, beforemorpho-logical
changesindicative
of lymphocytetrans-formation
haveoccurred.
The process
by which
PHA converts anon-productive virus infection
inlymphocyte
culturesinto
ahighly productive
oneremains
to beelucidated.
Several
factors
maycontribute,
dependent
upon PHAand
operating within
thefirst
8 to 16 hrafter virus inoculation.
The moreobvious
possibilities include: enhanced virus
absorption
orpenetration
into
lymphocytes,
orachange
incell metabolism that
permits
VSVreplication
toproceed
inlymphocytes already
infected
with virus. Since
anincreased
rateof
RNA
and
protein synthesis
occurswithin
2 to10
hr
after
lymphocytes
arestimulated with
PHA (11, 7), and DNA synthesis is increased onlyafter
24to 48hr(27,
21),
VSVreplication
maybe
associated with
theearlier
change
in RNA orprotein metabolism.
The
percentageof
cellsproducing
infectivevirus
particles
inPHA-treated
lymphocyte
cultures is
notknown.
Theobservation that
enhanced VSV
replication
is
associated with
destruction
of
75 to 97%of
thelymphoblasts
in PHA-treated
leukocyte cultures
is,
onthe one.hand,
compatible
with thehypothesis
thatvirtually
all blastcells
produced infectious virus
particles prior
totheirdestruction.
On theotherhand,
thedestruction
of blast cells
inVSV-infected
cultures may have been theresult of
apoorly productive cytocidal infection
in thelarge
majority
of
infected,
PHA-stimulated
lymphocytes, andonly a small percentage
of
theblast cells
destroyed
may have beenresponsible
for
thelarge yields of
VSV.PHA has
recently
been shown tocontain
atleast three
physically
distinct and activecompo-446
on November 11, 2019 by guest
http://jvi.asm.org/
[image:7.487.56.248.424.594.2]nents: a leukoagglutinin (16, 20), a factor which induces RNA synthesis in lymphocytes (16, 20),
and a factor which induces DNA replication
(mitogenic factor; 16). In the present study
several methods were employed in an attempt to isolate a possible fourth factor responsible for
enhancementof VSV. Initially, the concentration
ofPHAwas altered in thehopeof diluting outits
blastogenic (possible RNA-synthesizing factor)
and leukoagglutinating activities. Next,
anti-PHA serum was employed to separate theactive,
antigenic factors of PHA (1), from possible active, nonantigenic components. Finally, PHA
was exposed to heat, trypsin, ribonuclease, and
deoxyribonuclease. It wasfound that the
portion
of the PHA extract responsible for converting
lymphocytes into virus-susceptible cells could
not be dissociated from the components
re-sponsible
forleukoagglutination
orblast-cell
transformation. Thus, virus enhancement may
beintimately relatedto theactivity ofone of the
aforementioned components of PHA (20). The
complete separation
of PHA into componentparts hasnotbeen
achieved,
and theexistenceof
as yet unrecognized factors responsible for the
induction of
virusreplication
in humanlympho-cytescannotbeexcluded.
Aproblem raised in the present study and by
past reports (13, 4) concerns the PHA-related
specificity
ofthevirus-enhancementphenomenon.Is enhancement of VSV
replication peculiar
tothe interaction of PHA and
lymphocytes,
orwill any
lymphocyte mitogen
enhance virusreplication?
Twomitogenic substances,
rabbitanti-human
lymphocyte
serum(ALS;
9)
andstreptolysin (SLS; 12)
have been testedfor
theirVSV-enhancing
properties
inhumanlymphocyte
cultures. Both ALS
(6a)
andSLS(in
preparation)
significantly
enhancedvirusreplication;
butSLS,
in contrasttoboth ALS and
PHA,
enhancedvirusreplication
inlymphocytes
without associatedleukoagglutination.
Thus, the enhancement of VSVreplication
inlymphocytes
isnotpeculiar-to
PHA, nordoes it
depend
uponlymphocyte
leuko-agglutination; rather,
it isprobably
moregener-ally
relatedtostimulation oflymphocyte
metabo-lism.
ACKNOWLEDGMENT
Wethank Juanita RuffierandEthel Blair for their abletechnical assistance.
Thisworkwasconductedundertheauspicesof the Commission on Acute Respiratory Diseases of the Armed Forces Epidemiological Board and was
supported by contract DA-49-193-MD-2090 of the U.S. Army Medical Research and Development
Command, and by Public Health Service grant 5-F2-AI-30, 193-02 from the National Institute of Al-lergyandInfectious
Diseases,
and5-K03-CA31815-02 from theNationalCancerInstitute.LITERATURE CITED
1. Byrd, W. J., K. Hare, W. H. Finley, and S. C. Finley. 1967. Inhibition of themitogenicfactor in phytohemagglutinin by an antiserum. Nature 213:622-624.
2. DeMaeyer-Guignard, J., and E. DeMaeyer. 1965. Effects of carcinogenic and non-carcino-genic hydrocarbons on interferon synthesis and virus plaque development. J. Natl. Cancer. Inst. 34:265-276.
3. DeMaeyer-Guignard, J., and E. DeMaeyer. 1965. Inhibition of interferon synthesis and stimulation of virus plaque development in mammalian cell cultures after ultra-violet ir-radiation. Nature 205:985-987.
4. Duc-Nguyen, H., and W. Henle. 1966. Replica-tion of mumps virus in human leukocyte cultures. J. Bacteriol. 92:258-265.
5. Edelman, R., and E. F. Wheelock. 1966. Vesicular stomatitis virus replication in human leukocyte cultures: enhancement by phytohemagglutinin. Science 154:1053-1055.
6. Edelman, R., and E. F. Wheelock. 1967. Specific role of each human leukocyte type in viral infections. I. Monocyte as host cell for vasicular stomatitis virus replication in vitro. J. Virol. 1:1139-1149.
6a. Edelman, R., and E. F. Wheelock. 1968. En-hancementofreplication of vesicular stomatitis virusin human lymphocyteculturestreated with
heterologous anti-lymphocyte serum. Lancet.
1:771-775.
7. Epstein, L. B., and F. Stahlman, Jr. 1964. RNA synthesis in cultures of normalhumanperipheral blood.Blood 24:69-75.
8. Fiala, M., and G. E. Kenny. 1966. Enhancement of rhinovirus plaque formation in human heteroploid cell cultures by magnesium and calcium. J. Bacteriol.92:1710-1715.
9. Grasbeck, R., C. T. Nordman, and A. DeLa
Chapelle. 1964. The leukocyte mitogenic
effect of serum from rabbits immunized with human leukocytes. Acta Med. Scand. Suppl. 412:39-47.
10. Grossberg, S. E., M. Lwoff, andA.Lwoff. 1966. Exaltation of the development ofpoliovirus by semicarbazide. J. Bacteriol. 92:1473-1477. 11. Hirschhorn, K., F. Bach, R. L. Kolodney, T. L.
Firschein, and N. Hashem. 1963. Immune response and mitosis of human peripheral
blood lymphocytes in vitro. Science 142:1185-1187.
12. Hirschhorn, K., R. R. Schreibman, S.Verko, and R. H. Gruskin. 1964. The action ofstreptolysin S on peripheral lymphocytes of normal sub-jects and patients with acute rheumatic fever.
Proc.Natl. Acad. Sci. U.S. 52:1151-1157. 13. Nahmias, A. J., S. Kilbrick, and R. C. Rosan.
1964. Viral leukocyte interrelationships. I.
Multiplicationof a DNAvirus herpessimplex in human leukocyte cultures. J. Immunol.93: 69-74.
14. Nowell, P. C. 1960. Phytohemagglutinin: an
VOL. 447
on November 11, 2019 by guest
http://jvi.asm.org/
initiator of mitosis in cultures of normal humanleukocytes. Cancer Res. 20:462. 15. Paul,J. 1960.Cellandtissue culture, p. 784.The
Williams and Wilkins Co., Baltimore, Md. 16. Punnet, T., and H. H.Punnet.1963. Induction of
leukocyte growthincultures ofhuman periph-eral blood.Nature198:1173-1174.
17. Rabinowitz, Y. 1964. Separation oflymphocytes, polymorphonuclear leukocytes andmonocytes on glass columns, including tissue culture
observations.Blood23:811-828.
18. Rapp, F., and D. Vanderslice. 1965.
Enhance-ment of the replication of measles virus by mitomycin C. J. Immunol.95:753-758. 19. Reed, L. J., and H. A. Muench. 1938. A simple
method ofestimating fifty percent endpoints. Am. J.Hyg.27:493-497.
20. Rivera, A.,and G. C. Mueller. 1966. Differentia-tion of the biological activities of phyto-hemagglutinin affecting leukocytes. Nature 212:1207-1210.
21. Salzman, N. P.,M.Pellegrino,and P.Franceschini. 1966. Biochemical changes in
phytohemagglu-tinin stimulated human lymphocytes. Exptl. Cell Res. 44:73-83.
22. Singhal, S. K., C. K. Naspitz, and M. Richter. 1967. The action of phytohemagglutinin in rabbits. I. The enhancement of the primary
immune response to human serum albumin,
bovinegammaglobulin, and sheep erythrocytes. Intern.Arch. Allergy 31:390-393.
23. Spreafico, F., and E. M. Lerner. 1967. Suppres-sion of the primary and secondary immune
responseofthemousebyphytohemagglutinin.
J.Immunol.98:407-416.
24. Wheelock, E. F. 1965. Interferon-like virus-inhibitor induced in human leukocytes by phytohemagglutinin. Science 149:310-311. 25. White, D. O., and I. M. Cheyne. 1965.
Stimula-tion of Sendai virus multiplication by
puro-mycin andactinomycin D. Nature 208:813-814. 26. Wintrobe,M. M. 1961. Clinical hematology, 5th
ed. Lea & Febiger, Philadelphia.
27. Yoffey, J. M., G. C. B. Winter, D. G. Osmond, and E. S. Meek. 1965. Morphological studies in the culture ofhuman leukocytes with phyto-hemagglutinin. Brit. J. Haematol. 11:488-497.