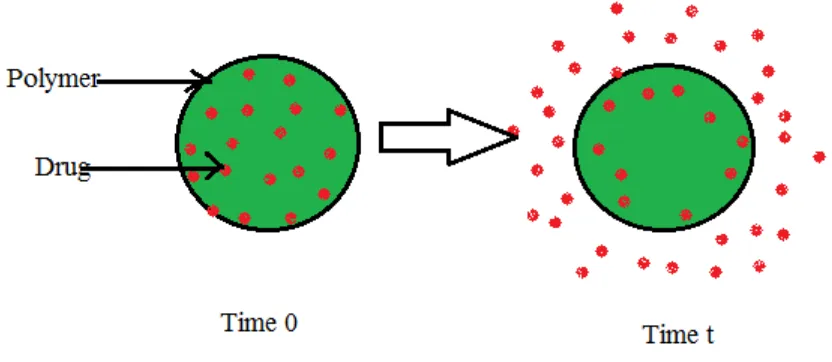
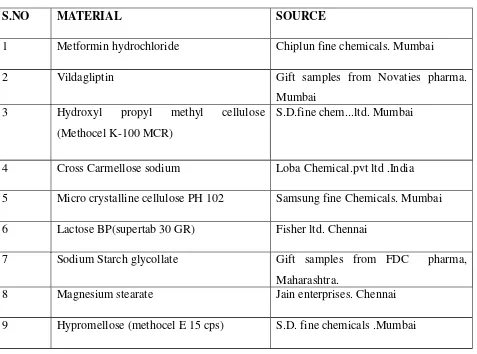
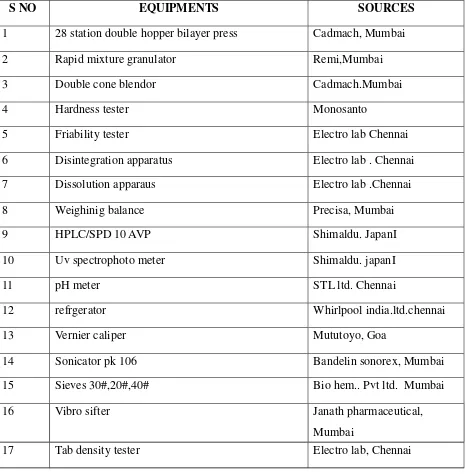
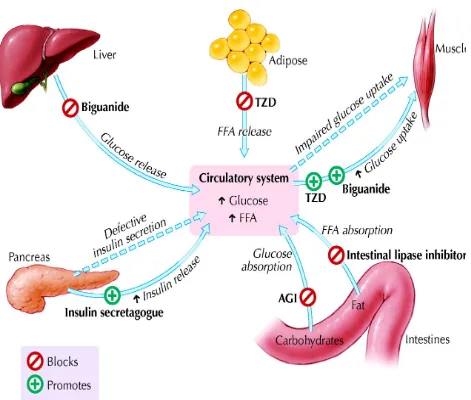
Formulation and Evaluation of Bilayer Tablets of Metformin Hydrochloride SR and Vildagliptin IR
Full text
Figure
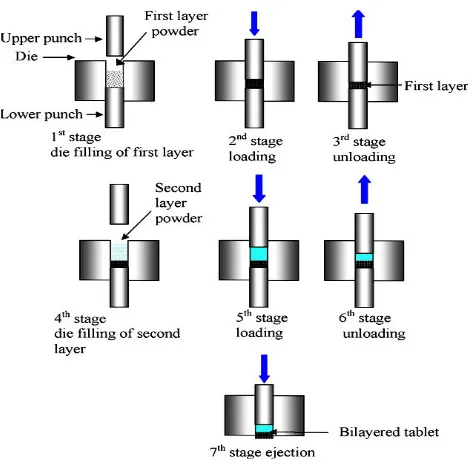
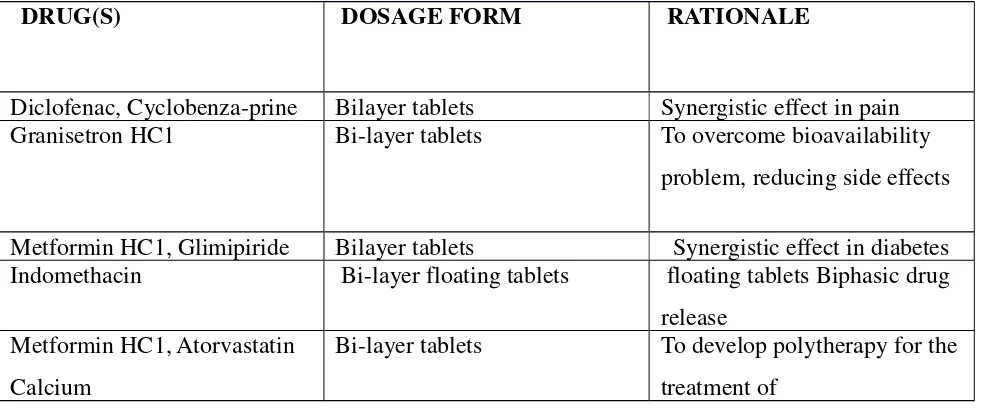
Related documents
FORMULATION AND IN VITRO EVALUATION OF METFORMIN HYDROCHLORIDE DIRECT COMPRESSIBLE TABLET USING BY.. HICEL TM MICROCRYSTALLINE
In-vitro evaluation and optimization of controlled release floating drug delivery system of metformin hydrochloride. Hollow microspheres for use as
Vol 9, Issue 6, 2016 Online 2455 3891 Print 0974 2441 STUDIES ON FORMULATION AND EVALUATION OF ETHYL CELLULOSE BASED EXTENDED RELEASE METFORMIN HYDROCHLORIDE MATRIX TABLETS SUNDARA
Research Article DEVELOPMENT AND VALIDATION OF A STABILITY?INDICATING HPLC UV METHOD FOR THE DETERMINATION OF PIOGLITAZONE HYDROCHLORIDE AND METFORMIN HYDROCHLORIDE IN BULK DRUG
Shukla KV, Vishwakarma P, Pathak R, Formulation Development and Evaluation of Gastroretentive Floating Tablets of Trazodone Hydrochloride Using Natural Polymer, Journal of
modified release metformin hydrochloride matrix tablets: Influence of some hydrophilic polymers on release rate and invitro evaluation. Rohini D, Alexander S
Table 1: Composition of pioglitazone hydrochloride (30 mg) effervescent floating matrix tablets.. Table 3: Dissolution parameters, and f2 factor of pioglitazone hydrochloride
The tablets were subjected to weight variation, thickness, hardness, friability, drug content and invitro release studies.. The TG/DTA analysis reveals that there is a