0022-538X/79/01/0242/08$02.00/0
In
Vitro
Synthesis of Infectious Transforming DNA by the
Avian Sarcoma Virus Reverse
Transcriptase
CAROL H. CLAYMAN, ENAYAT MOSHARRAFA, AND ANTHONY J. FARAS* Departmentof Microbiology, UniversityofMinnesota MedicalSchool, Minneapolis, Minnesota 55455
Received forpublication10July1978
Infectious DNAmolecules,capable of transforming chicken embryo fibroblasts,
canbesynthesizedby the Rous sarcoma virus-associated reverse transcriptase in
vitro. Theoptimal enzymatic conditionsemployedfor infectious DNAsynthesis
also facilitatemaximumsynthesis of genomelength DNA. Analysis of the DNA
product synthesized by detergent-disrupted Rous sarcoma virus under these
conditionsindicatesthat DNAcomplementarytoviral RNA(minus-strandDNA)
isgenomelength in size, whereasDNAcomplementarytogenomelength
minus-strandDNA(plus-strand DNA)appearsassubgenomic-length moleculesranging
between 300and3,500nucleotidesinlength.These features of the DNAproduct
synthesized by the Roussarcomavirusreverse transcriptaseinvitroaresimilar
to those identified in the cytoplasm of cells shortly after infection and lend credence to studiesof the mechanismofreverse transcription in vitro andtheir
significancetoproviralDNAsynthesisin vivo.
Theretrovirusreversetranscriptase plays an
important role in the life cycle of the virus by
converting theviral RNA genome intoproviral
DNAshortlyafter infection. Studiesonthe
na-ture of
proviral
DNA have delineated twopre-dominant structures in virus-infected cells: a
linear molecule anda
covalently
closedcircularmolecule (7, 8, 11, 28, 30). Both appear to be
doublestrandedinnatureandcorrespondtothe
length ofa 35S RNAsubunit ofthe retrovirus
genome. The double-stranded linear molecule
consists of a strand of genome length DNA
which is complementary to the viral genome
(minus-strand
DNA)andpiecesof DNAvaryingin size from 300 to 3,500 nucleotides in length
whicharecomplementarytothe genomelength
minus DNA
(plus-strand
DNA) (8, 28, 30).Theselinearmoleculesrepresenttheinitialviral
DNAstructuresfound in thecytoplasmofcells
shortly after infectionand appeartobe
precur-sors tocovalently closed circular formsof viral
DNAidentified in the infectedcellnucleus (25). Both forms of viral DNA purified from virus-infectedcellshavebeen shown to be infectious
(6).
Although the reverse transcriptase is capable oftranscribing the viral RNA genome into pro-viralDNA invivo and presumably could
inde-pendently be responsible for the synthesis of
double-stranded linear DNA molecules, no dou-ble-strandedstructuressimilar to those observed invivo have been identified in the DNA product
made in vitroby the retrovirusreverse
transcrip-tase. Earlierstudies indicated that the enzyme
could, at best, transcribe the viral genome into
smallpiecesof viral DNA100 to 700nucleotides
in length (10, 27). In fact, only recently have
enzymatic conditions been described that
pro-motedthe transcriptionof theviralgenomeinto
largeDNAmolecules, including genomelength
DNA in vitro(3, 14, 18, 23, 24, 31). In this report, wedemonstrate thatthe avian
retrovirus-asso-ciatedreversetranscriptaseiscapableof
synthe-sizing infectious, transforming DNA under
re-action conditions that facilitate a highyield of
genome length DNA transcripts (ca. 7.5 to 10
kilobases). We also document the conditions
required for maximum full-length DNA
tran-scription in this system and elaborate on the
similarities and differences in reaction
condi-tions between the murine and avian systems
required to facilitate this transcription. Finally,
wepresent dataindicating that the DNA
prod-uct, synthesized under reaction conditions
re-quiredtogenerateinfectious viral DNA, exhibits
featuresclosely resemblingthedouble-stranded linear forms of viral DNA found in the cyto-plasm ofcellsshortly after infection. These latter observations further justify studies on reverse
transcriptioninvitroasaviable means of
delin-eating at least the initial stages of retrovirus
proviralDNAsynthesis in vivo. While this
man-uscriptwasinpreparation,infectious DNA was
synthesized by murine leukemia virus in vitro
242
on November 10, 2019 by guest
http://jvi.asm.org/
underconsiderably different reaction conditions (24). The major differences between these two systems arediscussed.
MATERLALS AND METHODS Radiochemicals.[3H]TTP (50 to 60 Ci/mmol) and
[3H]cdGTP (10 to 25 Ci/mmol) were obtained from Schwarz/Mann; [3H]uridine was from Nuclear Dy-namics; and carrier-free 125I (350 mCi/ml) was from Amersham/Searle Inc. ['25I]dCTP was prepared by previously published procedures (5, 15). Unlabeled deoxynucleoside triphosphates were purchased from P-L Biochemicals, and unlabeled ribonucleoside tri-phosphates were purchased fromSigma.
Cells and virus. B77 strain (subgroup C) of Rous sarcoma virus (RSV) was propagated on gs-chf chicken and Peking duck embryo fibroblastcells (20). Virus was harvested from infected cultures at 24-h intervals and purified by previously published proce-dures (1).
Synthesisof genomelengthviral DNA. Virus-specific DNAwassynthesized in standard endogenous reaction mixtures (1ml) containing4to 5mgof virus protein; 0.05 M Tris-hydrochloride, pH 8.1; 0.01 M
MgCl2; 6x10-5Munlabeled deoxynucleoside triphos-phates(dATP,dGTP, dCTP, dTTP); 0.025% Nonidet P-40 (Shell); 1.4% (vol/vol)2-mercaptoethanol; 1mM
ofeach ribonucleosidetriphosphate (ATP, CTP,GTP, UTP); and 1 ,uCi of ['25I]dCTP. Reaction mixtures wereincubated for 16 h at 41°C andwereterminated by adding0.01 MEDTA, sodium dodecyl sulfate to
0.5%, and Pronase (500,ug/ml).The reaction mixtures
werefurther incubated for30min at37°C, extracted twice withphenol,andprecipitated with ethanol. The total nucleic acid product was nextseparated from free nucleosidetriphosphates by chromatography on
G-50Sephadex. Hydrolysisof RNA fromRNA -DNA
hybridswasperformedeitherby incubation in0.3N
NaOH for4h at37°Cfollowedby neutralization and ethanolprecipitationorby incubationwith RNase A
(Worthington)at100,ug/ml in20mM Tris-hydrochlo-ride, pH 7.4and 10mM EDTA (TE) for45 min at
37°C, followedbyphenolextraction andethanol pre-cipitation.
Transfection assay. Transfection ofvirus-specific DNA was performed by the calcium phosphate
co-precipitation technique asoutlined byKrzyzeketal. (17). Briefly, DNA was dissolved in
N-2-hydroxy-ethyl piperazine-N'-2-ethanesulfonate (HEPES)-saline-phosphatebufferat2to 20,g/ml, adjustedto 0.125M CaCl2,and allowedtoprecipitate for30min
atroomtemperature.Aliquots (0.5ml)of the calcium phosphate-DNA precipitates were addedto chicken
secondaryfibroblasts(0.5x 10'cells per 60-mm tissue
culture plate) previously washed with
HEPES-sa-line-phosphate buffer. After a 30-min incubation at roomtemperature,2.5ml ofgrowthmedium
contain-ing 10% calfserum but without tryptose-phosphate
brothwasaddedtotheculturesandincubated for4h at37°C.TheDNA-calciumphosphate-containing me-dium was then removed and replaced with culture
mediacontaining 10%calfserum and
tryptose-phos-phate broth. Positive transfections were scored by
visible foci oftransformedcells and/or the ability of transfected cultures to either incorporate[3H]uridine into virusparticleswithacharacteristicdensity of 1.16 g/cm3, or exhibit reverse transcriptase activity (16). Hybridization for RSV-specific DNA.
I"I-la-beled DNA was hybridized to unlabeled 70S viral RNA asfollows. Genome lengthDNA(1,200 cpm, 1.2 ng) was heated to 100°C for 2 min and then mixed with increasing amounts of 70S RNA in buffer con-taining 0.6 M NaCl-0.02 M Tris-hydrochloride (pH 7.4)-0.001 M EDTAinthepresence of250,ug of carrier yeast tRNA per ml. The mixture was incubated at
68°C for 40 h to achieve final Crt (concentration of RNA[moles/liter] x time[h]) values ranging from2
x 10-2to 2 x 10' (19). Hybrid formation was deter-mined with thesingle-strand-specific S1 nuclease (19, 22).
RESULTS
Optimum conditions for the synthesis of genome length DNA by RSV in vitro. We
have previously demonstrated the effect of
in-creasing the concentration of
deoxyribonucleo-side triphosphates on the length of the DNA
product synthesized by preparations of
deter-gent-disrupted
RSV in vitro (3). More recentstudies fromourlaboratory have indicated that
high concentrations ofdeoxyribonucleoside
tri-phosphates notonly dramatically affectthe
per-cent of the total DNA product that is greater
than 5,000nucleotides in length (ca. 20%), but thatwhichis genomelength (ca. 7%) insize as
well
(C.Clayman,E. Mosharrafa, andA. Faras,unpublished
data). Underoptimum
conditionsofenzymatic synthesis of DNA (Nonidet
P-40,
deoxyribonucleoside triphosphates,
tempera-ture, etc.) by detergent-disruptedRSV, the ad-dition of 1 mM each of the four ribonucleoside
triphosphates to the reaction mixture
consist-ently facilitated the
synthesis
ofhigh
yields
ofgenome length DNA molecules (Fig. 1). This effect was observed irrespective of whether the deoxyribonucleoside triphosphate concentration was 60,M or 2 mM and thusprovidedameans bywhichgenomelengthDNAcould be
synthe-sized withoutsignificantly reducing the specific
activity of the DNA
product.
Theeffect ofhigh
concentrations of ribonucleoside triphosphates
on thesynthesisofgenome-lengthDNA by
de-tergent-disrupted preparations
isconsistentwiththeobservations ofMyers et al. (21) employing reconstructed reactions containing 35S viral
RNA-
oligodeoxythymidylic
acid template-primer complexes
andpurified
reversetranscrip-tase. Presumably the presence ofhigh concen-trations of ribonucleoside triphosphates during
enzymatic synthesis reduces nicking of RNA,
thusallowinguninterrupted transcriptionof the
on November 10, 2019 by guest
http://jvi.asm.org/
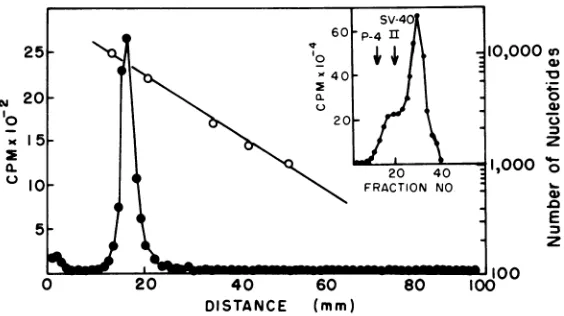
25- A0,000u
00 100
~~~~20
0~~~~~~~~~
o
20-15-
~~~~~~~z
0.
~~~~~~~~~~20
400~~~~~~~~~~~~~~~~~~~~.
14
~~~~~~~~~~E
Wi
~~~~100
0 20 40 60 80 100
DISTANCE (mm)
FIG. 1. Isolation and polyacrylamide gelelectrophoresis of genome length DNA transcripts. DNA tran-scriptssynthesized bydetergent-disruptedRSV underoptimumconditionsforgenomelengthDNAsynthesis
asdescribed in thetext wassubjectedtorate-zonal sedimentation in5to20o alkalinesucrosegradients(see inset). 3H-labeled P4 DNA(Mr 7.26x 106)and'4C-labeled simian virus40formII DNA(Mr - 3.30x 106)
weresedimented inparallelgradientsasmarkers andaredenotedbythearrows.Thosefractions containing
moleculesofapproximately 7,500to10,000nucleotides inlengthwerepooledandsubsequently analyzed by
electrophoresis under denaturing conditions in 98% formamide-3.5%polyacrylamide cylindrical gels by procedurespreviously outlined(9, 14). Nucleotidelengthcalibrationonthegelswasdeterminedbyparallel
electrophoresis ofP4DNA,fX-174DNA,and theHae III restriction endonucleasefragments of
OX-1
74DNA(2, 4).
viralgenome to occur.
Incontrast tothe murine leukemiavirus
sys-tem (22, 23), the ratio of divalent cation to
deoxyribonucleosidetriphosphate concentration
didnothavetobe reducedtopromotesynthesis
ofhigh yieldsof genome length DNA from
de-tergent-disrupted preparations of RSV. Two
other parameters that did, however, exhibit
varying effects on the yields ofgenome length
DNA were temperature and incubation time.
The optimal conditions routinely employedfor
the synthesis of consistently high yields of
ge-nomelengthDNAbydetergent-disrupted
prep-arations of RSVare asfollows: 0.025% Nonidet
P-40, 6 x 10-5 M deoxyribonucleoside
triphos-phates, 1 mM
ribonucleoside
triphosphates, 10mM
Mg2+,
0.02 MTris:hydrochloride,
1.4%(vol/vol) ,B-mercaptoethanol,andincubationfor
16hat410C.
Infectious nature of DNAsynthesized by
detergent-disrupted
RSVinvitro. We haveanalyzed the DNA product synthesized under
optimum conditions for genome length DNA
production for infectivity by the Ca3(PO4)2
co-precipitationmethod ofGraham and van der Eb
(9). The infectivity studies were initially
per-formed on total extracted viral nucleic acids
consisting ofviral RNA and DNA products to
optimizethe conditions formaximum
transfec-tionwith the invitro-synthesizedDNAproduct. Because our purified virus preparations
em-ployedfor thesynthesisofinfectiousDNA
con-tainedamixture of bothtransforming virusand
transformation-defective virus, assays were
de-veloped to determine the production of virus
particles as well as transformed foci. Superna-tants wereassayedfor virusbyboth[3H]uridine
labeling and reverse transcriptase procedures
(16). In ourhands, virusproductionasassayed
byeither of thesetechniques could be detected before overttransformation of cellsbecame
ev-ident. As seen inFig. 2,virusproduction could
be detected in those cell cultures transfected
with the total RSVnucleicacidproductbut not in mock-infected cultures. B77 RSV-infected duck cell DNA was
routinely
employed as apositive controlformaximum virus production
underoptimum transfection conditions.
The kinetics of transfectionareshown inFig.
3A. Virus detection routinely occurred by 12
days posttransfection and only after the cell
cultures were passaged at least twicetofacilitate
secondary virus infection of surrounding
non-transfected cells. Ifsufficient concentrations of viral DNA were employed in the transfection
experiments,transformed fociappearedin a
sim-ilar timeperiod.The minimum concentration of total DNAproductrequiredtoreproducibly in-itiate transfection was 5 ,ug/ml. Chicken cell
cultures transfected with both high and low
concentrationsof viral DNAproductareshown
inFig.4.Althoughbothtransfected cell cultures
actively produceRSV, only thehigher
concen-trations of DNA product weresufficient to
on November 10, 2019 by guest
http://jvi.asm.org/
[image:3.501.124.406.77.235.2]duce
morphological
transformation(Fig.
4). Thedependence of transformationonthe
concentra-tion of viralDNA
employed
inthe transfectionassaypresumablyreflects the presence of
trans-formation-defective variants which are present
in ourpreparations andarise during continuous propagationof large amounts ofRSV for these
studies. The virus obtained from chicken cell
culturestransfected withviralDNAproductwas
analyzed foritshostrange onseveral avian cell
types and was found to exhibit properties of
subgroup C viruses of whichB77RSV, the virus
employed togenerate infectious DNA in these
studies,was amember.
Nature of infectious RSV DNA product.
Although
purified
RSV RNA isnot infectiouswhenassayed with theCa3(PO4)2 technique (M.
Smith and A. Faras, unpublished data), itwas
neverthelessnecessary todeterminewhich
com-ponent of the total viral nucleic acid isolated
from virions after enzymatic synthesis ofDNA
wasresponsiblefor the observedtransfection of
chickembryo fibroblast cells. Therefore, sodium
dodecylsulfate-Pronase-phenol-extracted total
reaction product wasassayedforinfectivityafter
eitherRNase or DNasetreatment (Fig. 3).The
mostdramatic effecton
infectivity
wasexhibitedby
DNasehydrolysis
because both virusproduc-tion and transformaproduc-tion were
completely
elimi-nated by such treatment. On the other hand, RNasetreatment, under conditions of hydrolysis
sufficiently exhaustive to completely solubilize
RNA,includingthatwhich was complexed with
DNA, did not eliminate infectivity. In fact,
RNase-treated product accelerated the rate of
transfection. Because a constant amount of
nu-cleic acid was employed in the transfection
as-says, we attribute theaccelerated rate of
trans-fection observed after RNase hydrolysis to an
increasein the number of infectiousDNA
mol-eculespresentper microgram of product because
of the removal ofthe noninfectious RNA
com-ponent. Alternatively, RNase hydrolysis may
liberate infectious DNA moleculescomplexed in
noninfectiousDNA-RNA hybrids. Nevertheless,
these data demonstrate that infectivity is
de-pendent upon DNA aloneand not DNA-RNA
orDNA-proteincomplexes. Alkaline hydrolysis
of the total in vitro-synthesized virus-specific
nucleicacid product didnotsignificantly affect
infectivity,
although
a slight, butreproducible,
lagwas observedsubsequenttoalkaline hydrol-ysis, suggesting that denaturation may affect
theinfectivityof at leastaportionofthe
mole-cules present in the DNA product. The
impli-cationsofthesefindingswith regard to the
pos-siblestructureof theinfectious DNA molecules
arediscussed below.
o
2n
0LhX1E2>% 1.2 40'
5 10 15 5 10 1I
FRACTION NUMBER
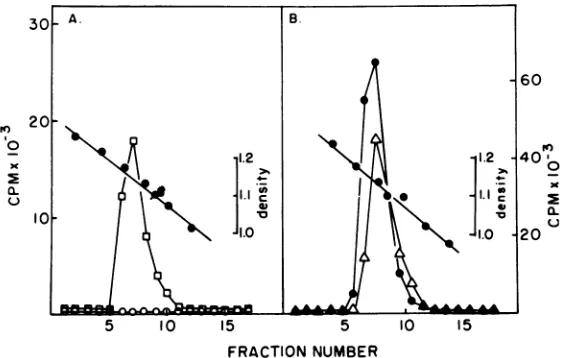
FIG. 2. 3H-uridinelabeling ofRSVfrom transfectedchickenembryofibroblasts. Chickenembryofibroblast
cultureswerelabeledovernightwith20
ICi
of[3H]uridineper mlat30daysposttransfection.Supernatants were collected, clarified by centrifuging at 10,000 x gfor 10min at 4°C, and layeredonto 5-ml sucrosegradientsconsistingof25to55%sucrose(wt/vol)in 0.1 MNaCl-0.02MTris-hydrochloride (pH 7.4)-0.01M
EDTA(STE) overlayedwith3mlof15%sucrose(wt/vol)in STE. Thegradientswerecentrifuged for2.5h at
40,000rpminanSW41rotor at4°C.Fractionswerecollected,and therefractiveindexwasdetermined.Each
fractionwasassayed for[3H]uridineincorporation bytrichloroaceticacidprecipitationasdescribed
previ-ously (28).Data arepresentedas total[3H]uridinecontained within the 1.15- to 1.16-g/cm2 region ofthe
gradient.Symbols: (A)EO 0.48pgof RSV-DNAproductper60-mmplate;0,mock-infectedcultures. (B)
AL,
4.8 pgof RSV-DNAproductper 60-mmplate;0,5,ug ofRSVtransformed Pekingduck cell DNA per60-mmplatepreparedbypreviously publishedmethods(12).
on November 10, 2019 by guest
http://jvi.asm.org/
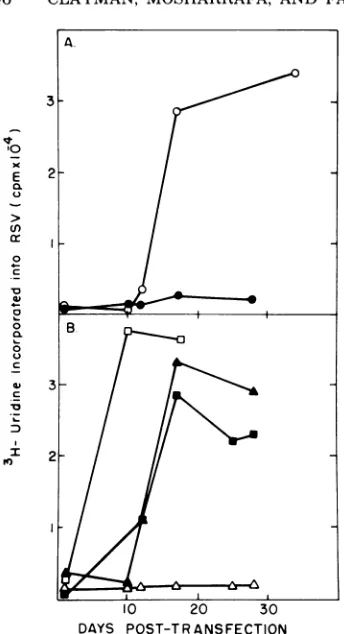
[image:4.501.106.389.380.559.2]2
0
0
DAYS POST-TRANSFECTION
FIG. 3. Kineticsof transfection with DNA synthe-sizedbydetergent-disruptedRSV in vitro. The kinet-ics of transfection with DNA synthesized by deter-gent-disrupted RSVweredeterminedbyassayingfor
virusproduction atdistincttimeintervals posttrans-fection by methods outlined in the legend toFig. 2. Symbols: (A) 0, chicken embryo fibroblasts
trans-fected with total DNA isolated from RSV-trans-formed duck cells: 0,mock-infected cells. (B) Total
nucleic acidwasisolatedfromreactionsafter synthe-sisofDNAbydetergent-disrupted virusasdescribed
inthetextandemployedintransfectionexperiments after eitherno treatment (a); RNase A (100,ug/ml) treatment (O) in 3mMEDTAfor 45min at 37°C; alkaline treatment (A) in 0.3M NaOH for 4 h at
37°C;orDNase(2 pg/ml)treatment(A)in0.2M Tris-hydrochloride (pH 7.4)-0.01 M MgCl2 bufferfor2hat
roomtemperature. In each case5 pgof productwas
addedtothetissueculturedishes.
Inanefforttodetermine whether similarities exist between viral DNA synthesized in vitro and that isolated from virus-infected cells in
vivo,wehaveanalyzedthestructureofthe DNA
product synthesized under our reaction condi-tions in vitro. In these studies viral DNA was sedimented in alkalinesucrose,and thesize and
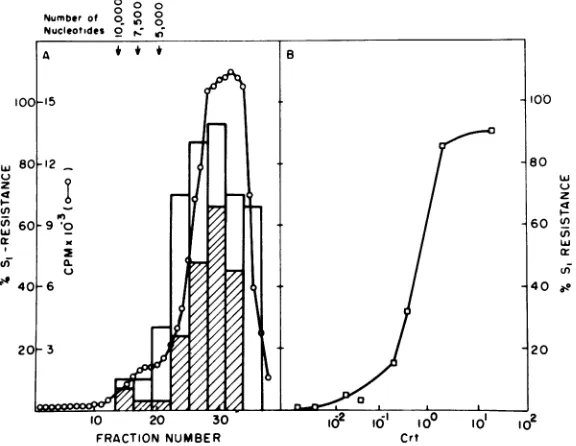
polarity of the DNA were determined (Fig. 5). Reassociation of the DNA present in each
frac-tionindicated that DNA strands of both polari-ties could be observed in the lower-molecular-weight region of thegradient (Fig. 5A) ranging between 300 and 3,500 nucleotides in length. However, very few double-stranded molecules were found after reassociation of the genome length region of thegradient,indicatingthat the bulk (ca. 90 to 95%) of DNA molecules of that size classcontained either minus-orplus-strand molecules butnotboth. Thepolarityof genome length DNA was directly established by dem-onstrating that these molecules could
com-pletely hybridize totheviral RNA genome(Fig. 5B). These data suggest that viral plus-strand DNAmolecules synthesizedunder ourreaction conditions consist ofsubgenomic length DNA
transcripts. To directly prove this contention,
we have reassociated single-stranded, purified, genomelengthminus-strand DNAtoeach frac-tionofanalkalinesucrosegradient andassayed forconversiontoSi-resistantduplexmolecules. Theresults of suchanexperiment are shown in Fig. 5A and demonstrate that DNA molecules
complementary to the genome length
minus-strand DNAmoleculescanonlybefound in the range of300 to 3,500nucleotides inlength.
Fur-thermore, because thesubgenomic length
plus-strand DNA renders the bulk(>80%)of genome length minus-strand DNA resistant to S1-nu-clease, most, if not all, of the viral genomic sequences must betranscribed into plus-strand DNA. Weconclude that the structure,size, and polarity of DNA synthesized under enzymatic conditions that facilitatesynthesis of infectious DNA moleculesare compatible with the
struc-ture ofduplexmolecules identifledinvivo
con-sisting of intact genome length minus-strand
DNA andsubgenomic length plus-strand DNA molecules.
DISCUSSION
In this communication we demonstrate that infectious, transforming DNA can be synthe-sized bythe RSVreverse transcriptase invitro under reaction conditions that promote
maxi-mum synthesis of genome length DNA
tran-scripts.Itappearsthat neither RNA norprotein
isrequired forinfectivity because neither RNase
norproteinasetreatmentaffect theabilityofthe
in vitro-synthesized DNA to transfect chicken embryo fibroblasts.
Thereaction conditionsrequiredfor the max-imum synthesis of RSV infectious DNA are
considerably different from those reported for
murine leukemia virus (22, 23).In ourhands the
Mg2+
concentration does not have to be kept below that of the deoxyribonucleoside triphos-phate concentration for maximum genomeon November 10, 2019 by guest
http://jvi.asm.org/
[image:5.501.74.246.64.381.2]RSV REVERSE TRANSCRIPTASE DNA SYNTHESIS
A
C
*
B
at w- ;.,
e
,
'¼ *. . e.7.
.?.Q ..
.
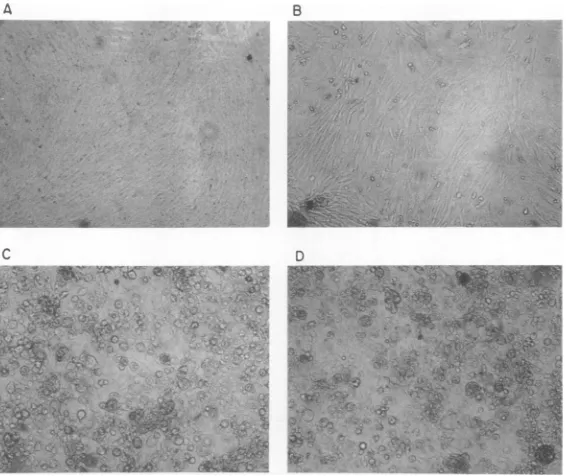
FIG. 4. Morphology of chicken embryo fibroblasts aftertransfection with DNA product synthesized by
detergent-disruptedRSV in vitro. Chickenembryo fibroblastsweretransfected with DNA product synthesized bydetergent-disrupted RSV in vitro as described in thetext. (A) Mock-infected cells; (B) 0.48pg of DNA product per60-mmplate; (C)4.8pgofDNAproduct per60-mmplate; (D)5pgof DNA fromRSV-transformed
duck cells per60-mmplate.Allphotographs weretakenatx80 magnification.
length DNA synthesis. However, the
signifi-cance ofthis observation is not clear, because
althoughtherateof DNA synthesisis maximum
with
Mg2"
forRSV,Mn2"
appearsthe preferreddivalent cation by murine leukemia virus (10).
Furthermore,wehave found that the
concentra-tion of deoxyribonucleoside triphosphates can
be
significantly
reduced withoutanyappreciableaffect on the
yields
ofgenomelength
DNA ifribonucleosidetriphosphatesarepresentduring
enzymatic synthesis. The
advantage
of thisma-neuveris that genomelengthDNA can be
syn-thesized without affecting the extent to which
the molecules can be labeled with radioactive
precursors. The yields of genome length DNA under our conditions of enzymatic synthesis range between 5 and 15% of the total DNA
product and are appreciably better than those
reported for RSVby Junghans et al. (14),
em-ployingsomewhat different conditions.
From preliminary structural studies on the
nature of the infectious DNA product synthe-sized by RSV, it is clear that genome length DNA is of theminuspolarity,whereas the plus-strand DNAproduct is found predominantly as subgenomic pieces ranging from 300 to 3,500 nucleotides in length. These findings suggest
that enzymatic reactions containing
detergent-disruptedRSV may be capable ofsynthesizing
invitrostructuralforms of proviral DNA similar
to the DNAspeciesfound in the cytoplasm of infected cells.Linear, duplex viralDNA isolated
from RSV-infectedcells appearsto contain
ge-nome length minus-strand DNA and
subgen-omic-length plus-strand DNA of the
approxi-matesize rangeasthatidentifiedinourin vitro
product (25, 28, 30).Our analyses, however, do
not ruleoutthe possibility of thepresenceofa
small portion of duplex molecules containing intactplus- and minus-strandDNA components
(see Fig. 5). Furthermore, because
double-stranded linear DNA molecules isolated from
virus-infected cellsareinfectious (6, 13, 26),it is conceivable that the infectious molecules syn-thesized in vitro representstructuresvery simi-lar tothoseidentified in vivo. Our transfection assays employing RNase-treated and DNase-treatedproductsynthesizedin vitroclearly
dem-onstratethat the DNA component is infectious. Webelieve that the acceleratedrateof transfec-tion observed after RNase
hydrolysis
is due toan increase in the total number of infectious molecules present per microgram of reaction product because of the removal of the noninfec-tious RNA genomeand other virion-associated
low-molecular-weight RNA species. On the
otherhand, alkalinehydrolysiswhich results in
aslightdecrease in the
efficiency
of transfection247
on November 10, 2019 by guest
http://jvi.asm.org/
[image:6.501.104.387.77.315.2]248 CLAYMAN,
oi w
z (.
0- 9 30 60
w
40 6 40
20-3
~~~~~~~~~~~~~20
10 20 30
162
16'
l00
o1le
FRACTIONNUMBER Crt
FIG. 5. Natureof viral DNAsynthesizedunderoptimumconditionsforinfectioustransforming DNA and genomelength DNA. Viral DNA productwassynthesizedby detergent-disrupted RSV under conditionsfor maximum genome-length DNA and subjected to alkaline sucrose gradient sedimentation subsequentto
alkaline hydrolysis (A) asdescribed in thelegendtoFig. 1. The resultantprofile is indicatedby the open circles and thepositionofthe various nucleotidelengthmarkers denotedbythearrows.Everyfour fractions
of the gradient werepooled, neutralized, and ethanolprecipitated. The precipitates werepelleted and resuspended in0.02MTris-hydrochloride, pH7.4, andaportionfromeachregionwasheatedat100°Cfor2
minandquenchedonice. The salt concentrationforeachsamplewasadjustedto0.6 MNaCl,and the DNA
wasincubatedat68°Cfor60h(Cot3.33x 10-') (29)topromote reassociationof duplexDNA. The percent double-stranded DNA in eachpooled gradient fractionwasdeterminedby the resistance of the reassociated portionstodigestion with thesingle-strand-specific nucleaseSi (19) andisrepresentedbythe shaded bar graph. Thecomplexity andpolarity ofeach size classof DNAwasascertainedby hybridizing unlabeled DNA
fromeachregionofthegradienttolabeled, single-strandedgenomelength DNA which isofminuspolarity (seeB).Briefly, 1,200 cpmofgenomelengthDNA(1.2 ng)wasmixed with unlabeled DNAfromeachsize class and allowedtoreassociate to a Cot of3.33x 10-1 asoutlined above. Duplexformationwasdetermined by resistancetoSinucleasedigestionanddenotedby the open bar graph. IntrinsicSi nuclease resistanceof the genomelengthDNAwas9.0%o. (B) Complexityandpolarity ofthe genomelength DNA. Labeled DNA pooled from the alkalinesucrosegradient in the range of 7,500to 10,000 nucleotides washybridized to varying
amountsofviral708 RNAbyproceduresoutlined inthetext.Extentof hybridizationwasdeterninedbySi nucleasehydrolysis.
notonlyremovesviral RNA but denaturesany double-stranded DNA present in the reaction product aswell. This relativelag in the rate of transfection after alkaline hydrolysis suggests that thepredominant infectious species isafully or partially double-stranded DNA molecule. Thisinterpretation is compatible with the
pre-liminary observation that double-strandedDNA
synthesized under conditions which facilitate
ge-nome length DNA synthesis exhibits greater infectivity than single-stranded DNA which is
only weakly infectious (Clayman and Faras,
un-published data). Consistent with this
interpre-tation is the demonstration thattheinfectivity
of lineardouble-stranded DNA from avianRNA
tumor virus-infected cells is also affected by
alkaline denaturation(6, 13,26).Further studies
are currently under way toelucidate the exact
natureoftheinfectious DNAmoleculesaswell
as the alkaline-sensitive DNA present in these preparations.
Finally, although these studies indicate that
virions of RSV contain all the necessary com-ponents for thesynthesisofinfectious DNA and it istherefore temptingtospeculate onthe sig-nificance of these observations with respectto
therelevance ofproviralDNAsynthesisinvivo, further studiesconfirming the restriction
endo-nuclease maps of the in vitro productwith the
in vivo structures willberequiredbefore we can
unequivocally conclude that in vitro
transcrip-tion accurately reflects the mechanism of
on November 10, 2019 by guest
http://jvi.asm.org/
[image:7.501.120.408.81.304.2]RSV REVERSE TRANSCRIPTASE DNA SYNTHESIS 249
viral DNA synthesis in vivo. Such studies are
currentlyinprogress inourlaboratory.
ACKNOWLEDGMENTS
We thank B. Bulbulian for excellent technical assistance. We also thank R. Ostrow and C.Thuilfor readingand typing this manuscript, respectively.
Thisinvestigation wassupported by Public Health Service Research grants CA 18303-02 and CA 20011-01 from the National Cancer Institute. C.H.C. is currently a predoctoral fellowsupported bytraining grant GM OD966 from the Na-tionalInstitute of General Medical Sciences.
LITERATURE CITED
1. Bishop, J., W. E.Levinson, N. Quintrell, D. Sullivan, L.Fanshier, and J. Jackson. 1970. The low molecular weight RNAs ofRoussarcoma virus 1. The 4S RNA. Virology 42:182-195.
2. Clayman,C. H., M. S. Collett, and A. J. Faras. 1977. Invitrotranscription of the avianoncornavirusgenome by the RNA-directed DNApolymerase: effect of acti-nomycin D on the extent of transcription. J. Virol. 23:209-212.
3. Collett,M.S.,and A. J. Faras.1975.Invitro transcrip-tion of70S RNA by theRNA-directed DNA polymerase ofRous sarcoma virus: lack of influence of RNase H. J. Virol. 17:291-295.
4. Collett,M.S.,and A. J. Faras.1977.In vitro transcrip-tion of theavian oncornavirus genome by the RNA-directed DNA polymerase: analysis of DNA transcripts synthesized in reconstructed enzymatic reactions. J. Virol. 22:86-96.
5. Commerford,S. L. 1971.Iodination of nucleic acids in vitro.Biochemnistry10:1973.
6. Fritsch, E., and H. M. Temin. 1977. Formation and structure ofinfectious DNA ofspleen necrosis virus. J. Virol. 21:119-130.
7. Gianni,A.M., D.Smotkin,and R. A.Weinberg.1975. Murine leukemia virus: detection ofunintegrated dou-ble-stranded DNA forms of the provirus. Proc. Natl. Acad. Sci. U.S.A.73:447-451.
8. Gianni, A. M., and R. A. Weinberg. 1975. Partially single-stranded form of freemoloneyviral DNA. Nature (London) 255:646-648.
9. Graham, F.L., and A. J.van der Eb. 1973. A new technique for the assay ofinfectivityof human adeno-virus 5DNA.Virology 52:456-467.
10.Green, M., and G. F. Gerard.1974.RNA-directed DNA polymerase-propertiesand functions inoncogenic RNA virusesand cells. Prog. Nucleic Acid Res. Mol. Biol. 15:187-334.
11. Guntaka, R.V., 0.C.Richards,P. R.Shank,H. J. Kung, N.Davidson,E.Fritsch,J. M.Bishop, and H. E. Varmus.1976.Covalentlyclosed circular DNA of aviansarcomavirus:purificationfrom nuclei of in-fectedquailtumorcells andmeasurementby electron microscopy and gel electrophoresis. J. Mol. Biol. 106:337-357.
12. Haase,A., B. Traynor, P.Ventura, and D.Ailing.
1976.Infectivityof visna virus DNA.Virology70:65-72. 13.Hillova, J., G.Goubin, andM.Hill.1972.Transfection des fibioblastes de Poule par l'acide d6soxyribonu-cleiquedenature decellulestransformeesde Rous. C. R.Acad.Sci. Paris274:1970-1973.
14. Junghans,R.P.,P. H.Duesberg,and C.A.Knight. 1975.Invitrosynthesis offull-length DNA transcripts ofRoussarcomavirusRNAbyviralDNApolymerase.
Proc.Natl. Acad. Sci. U.S.A. 72:4895-4899.
15. Kamen, R. J. Sedat, and E. Ziff. 1976. Orientation of thecomplementary strands of polyoma virus DNA with respect to the DNA physical map. J. Virol. 17:212-218. 16.Kieras, R. M., and A. J. Faras.1975. DNA polymerase ofreticuloendotheliosis virus: inability to detect endog-enous RNA-directed DNA synthesis. Virology 65:514-523.
17. Krzyzek, R. A., A. F. Lau, A. J. Faras, and D. H. Spector. 1977. Post transcriptional control of avian oncornavirus transforming gene sequences in mamma-liancells. Nature (London) 269:175-179.
18. Lai, M.M.C., and S. S. F. Hu. 1978. In vitro synthesis and characterization of full- and half-genome length complementary DNA from avian oncomaviruses. Na-ture(London) 271:481-483.
19. Leong, J.-A., A.-C. Garapin, N. Jackson, L. Fanshier, W.Levinson, and J. M. Bishop. 1972. Virus-specific ribonucleic acid in cells producing Rous sarcoma virus: deWectionandcharacterization. J. Virol. 9:891-902. 20. Levinson, W. E. 1967.Fragmentationof thenucleusin
Rous sarcoma virus-infected chickembryo cells. Virol-ogy32:74-83.
21. Myers, J.C., S. Spiegelman, and D. L.Kacian. 1977. Synthesis of full-length DNA copiesof avian myelo-blastosis virus RNA inhigh yields.Proc. Natl. Acad. Sci. U.S.A.74:2840-2843.
22. Rothenberg, E., andD.Baltimore.1976.Synthesis of long, representative DNA copies of themurineRNA tumorvirusgenome. J. Virol.17:168-174.
23. Rothenberg, E., and D. Baltimore. 1977. Increased length of DNA made by virions of murine leukemia virus atlimiting magnesium ion concentration. J.Virol. 21:168-178.
24. Rothenberg,E., D.Smotkin,D.Baltimore,and R. A. Weinberg.1977.In vitrosynthesisof infectious DNA of murine leukemia virus. Nature (London) 269:122-126.
25. Shank, P. R., and H. E. Varmus.1978. Virus-specific DNAinthecytoplasm of aviansarcomavirus-infected cellsis aprecursortocovalently closed circular viral DNAinthe nucleus.J.Virol.25:104-114.
26. Svobada, J., I.Hlozanek,0. Mach,A.Michlova, J. Riman, and M. Urbankova. 1973. Transfection of chicken fibroblasts withsingle exposuretoDNA from virogenicmammaliancells.J. Gen. Virol.21:47-55. 27. Taylor, J. M., A. J. Faras, H. E. Varmus, W. E.
Levinson,and J. M.Bishop. 1972.Ribonucleic acid directeddeoxyribonucleicacidsynthesisbythepurified deoxyribonucleicacidpolymeraseof Roussarcoma vi-rus. Characterization of the enzymatic product. Bio-chemistry11:2343-2351.
28. Varmus, H.E., S. Heasley, J.Linn,andK.Wheeler. 1976.Use ofalkalinesucrosegradientsin azonalrotor to detect integrated and unintegratedavian sarcoma virus-specific DNA incells. J. Virol.18:574-585. 29. Varmus,H. E., W. E. Levinson,and J. M. Bishop.
1971.Extent oftranscription by theRNA-dependent
DNApolymeraseof Roussarcomavirus.Nature (Lon-don)NewBiol. 233:19-21.
30. Varmus,H., andP. R.Shank. 1976.Unintegratedviral DNA issynthesizedin thecytoplasmofaviansarcoma
virus-transformed duckcells byviral DNApolymerase. J. Virol. 18:567-573.
31. Verma,L.M. 1978.Genomeorganizationof RNAtumor
viruses.I. In vitrosynthesis offull-genome-length sin-gle-stranded and double-stranded viral DNA tran-scripts. J. Virol. 26:615-629.