JOURNAL OFVIROLOGY, Dec. 1971, p. 910-918
Copyright ©1971 American SocietyforMicrobiology PrintedVol.in8,U.S.A.No.6
Structure
and
Development
of
Viruses
as
Observed
in
the
Electron Microscope
XI. Entry and
Uncoating
of
Herpes
Simplex
Virus
KANEATSU MIYAMOTO'AND COUNCILMAN MORGAN
Department ofMicrobiology, College of PhysiciansandSurgeons, Columbia University, New York,New York
10032
Receivedforpublication9August 1971
Two morphologically distinct types ofcapsids are described. The dense capsid appearedto bedisrupted nearthe cellular membrane with release ofcorematerial. Thelight capsidwas more stable andwas frequently encounteredclose tothe
nu-cleus, where emptycapsids werealso found.Pretreatment ofcells before infection with either puromycin or actinomycin D markedly decreased the percentage of emptycapsids. It is suggested that thetwo types ofcapsidsplay different roles in theprocess ofinitiatinginfection. One (the dense capsid) releasesdeoxyribonucleic acid(DNA) shortly afterentry.This DNA is transcribed intoavirus-specific
ribo-nucleicacid, which codes foranenzymecapableofalteringthepermeability of the secondtypeofcapsid (thelight capsid). In proximitytothenucleus, the infectious DNA thenescapeswithoutgrossdisruption of the capsid.
Herpes simplex virus has been reported to enter cellsbyfusion of the viral envelope to the cellmembrane with release of thecapsid directly into the cytoplasm (5). In
experiments
with iso-tope-labeled virus,deoxyribonucleic acid (DNA) wasfound to be uncoated in the cytoplasm and transferred to the nucleus (2). Although initial studies showed capsids at stages of dissolution with releaseofcores to thecytoplasm
(5),
subse-quentinvestigation alsorevealedsignificant num-bers of intact capsids devoid of cores in close proximityto the nucleus.The purpose of thepresentstudywas to exam-ine indetail the sequentialevents occurring after entryofthe
capsid
intothe cell. In anattemptto clarify the mannerinwhich infection isinitiated,
astatisticalstudyof thedifferentviralformsseen at intervals afterentry was undertaken. In addi-tion, the effects onviral
uncoating
ofpuromycin (inhibitor of protein synthesis), actinomycin D [inhibitor of DNA-dependent ribonucleic acid (RNA)synthesis],
and ultraviolet (UV) irradia-tion wereobserved.MATERIALS AND METHODS
Cells and culture media. Monolayer cultures of HeLa cells were maintained in Eagle's minimal essential medium (MEM; Grand Island Biological Co.,GrandIsland,N.Y.) supplementedwith5% calf
serum.
1Postdoctoral Fellowof The DamonRunyonMemorialFund forCancerResearch.
Virus. The Miyama strain of herpessimplex virus
wasused.
Preparation of inocula. The cells wereinfected with
a10-1dilution ofstock virus. Afterincubation for 48 hr, the cells were scraped into MEM and sonically treated for 10 min, and the debris was removed by low-speedcentrifugation. Thevirus-containing super-natant fluid was used immediately.
Virusinfection for time course experiments. Sparse monolayers (5 X 105 cells) in 30-ml plastic flasks (FalconPlastic, Los Angeles, Calif.) werecooled by placing them in an ice bath for 15 min. The cell layers were washed with cold Earle's solution and infected with 0.5 ml of cold fresh virus preparation containing 108 TCIDwo/ml. After placing the flasks on arocker at 4 C for 1hr, the cells were washed with coldEarle'ssolutionand, to synchronize the initiation ofvirus entry, warmedquicklyto37Cbyadding pre-warmed MEM.Theywereincubatedatthis tempera-turefor 0, 2, 5, 10, 20, 30, 40, 60, and 120 minand then cooled to 4 C, fixed for 30 min in situ with cold 1%glutaraldehyde bufferedatpH 7.2, scrapedfrom thesurface of theflask, andpelleted by centrifugation. Metabolic inhibitors. Puromycin dihydrochloride
was obtained from Nutritional Biochemicals Corp., Cleveland, Ohio, and the final concentration was adjusted to 20
pig/ml.
Actinomycin D was kindly supplied by Merck Sharpe and Dohme Research Laboratories, Rahway, N.J., and was used at a final concentration of 5 ,g/ml.Virus infectionforinhibitorexperiments.After
pre-treatmentwith inhibitors in3ml ofMEMfor different periods at 37 C, cell layers were washed once with prewarmed Earle's solutioncontaining inhibitor and inoculated with 0.5 ml of prewarmed, fresh virus 910
on November 11, 2019 by guest
http://jvi.asm.org/
UNCOATING OF HERPES SIMPLEX VIRUS
(108 TCID5o/ml). The flasks were then placed on a rocker for 1 hr at 37 C. At the end of theincubation period, cell layerswerewashed,scraped, pelleted, and then fixedfor 1 hr with cold 1% glutaraldehyde.
Ultraviolet irradiation. A 5-ml amount of a fresh virus suspension wasplacedin apetriplate(5cm) on anice bath at adistanceof 10 cm from theultraviolet source (GE germicidal 30-w UV light). The virus suspension was agitated frequently throughout the period of irradiation. UV irradiation for 5 and 10 mindecreased theinfectivitytiter from 108TCID5o/ml to 102 and 101TCID50o/ml,respectively.Noinfectivity was detectable when irradiation was prolonged for 30 min.
Isolation of cytoplasmic capsids. Suspensions of fresh virus were concentrated by centrifugation (105,000 X g) and resuspended in Earle's solution. Monolayers of HeLa cells were inoculated with concentrated virus and incubated for 1 hr at 37 C. The cells were then washed with Sorenson's buffer, harvested, frozen, thawed, and subjected to sonic vibration for 10 min. The suspensions werecleared by low-speed centrifugation. Paraformaldehyde at a
final concentration of3% was added to portionsof the supernatant fluid, whichwerekeptat4C for 15 hr, and thensubjectedtoisopycniccentrifugation in CsClasdescribedbySpringandRoizman(9).Bands werecollectedandnegatively stained with 1% phos-photungstic acidfor electronmicroscopy.
Ferritin-conjugated antibody. The 7S antiherpes antibody was kindly provided by B. Hampar (Na-tional Cancer Institute). Rabbits received, over a
period of 8weeks, onecorneal and fiveintravenous injections of the CHR-HSV-3 strain of herpesvirus grown in primaryrabbit kidney (PRK) cells.The7S antibodywasisolatedfrom the serumbygel filtration on Sephadex G-200 (1). Ferritin conjugation was
kindly carried out by K. C. Hsu by a previously described procedure (8).
Application offerritin-conjugatedantibody.Infected cellswerefixed in situwith 1.6%o paraformaldehyde for 5 min, immersed in
10%0
dimethyl sulfoxidefor 30 min, collected by scraping, and pelleted. The pelleted cells were frozen in a CO2-alcohol bath, thawed, resuspended in the ferritin conjugate for 1 hr at room temperature, washed by pelleting and resuspension four times in Sorenson's buffer, and fixedfor 1 hrwith cold 1%glutaraldehyde.Preparation for electron microscopy. The cell pellets, after fixationinglutaraldehyde, werewashed withSorenson'sbuffer, fixedfor 30 min in1%osmium tetroxide, dehydrated, andembedded in epoxy resin (Epon 812). The sections were stained with uranyl acetate and lead citrate before examination in an RCA 3G or aPhilips200 electron' microscope.
RESULTS
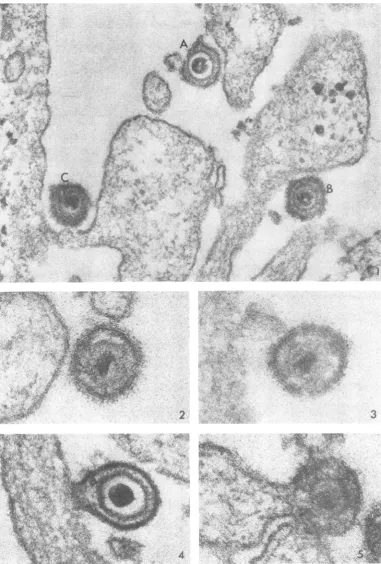
Virusparticles on the cell surface. Twotypes of virionswereobserved atthesurfaceofcellsafter the inoculum was added. Figure 1 was taken 2 minafterwarming the cells to 37 C and illustrates three enveloped particles on the cell surface. In onevirion (marked A), a translucent zone
sepa-ratesthe capsid from the dense core. In the virion marked B, the translucent zone contains finely granular, dense material. (The opacity of this granular material to the electron beam varies
considerably
fromoneviriontoanother.) Forthe sake of simplicity, the former will be termed a lightcapsid andthelatter will be termed a dense capsid. Light capsids constituted 53% and dense capsids constituted 47% of the total particles ob-servedonthecell surfaceatthe end of1 hr at 4C (time 0). Half of the latter exhibited poorly de-fined capsids, as seen inparticle C and as illus-trated athigher
magnification
inFig.
2 and 3.Virus entry. At time0, no free virus could be found in thecytoplasm.Inthesampleswarmed to 37 C,the site of fusion oftheviral envelope with thecell membranewasoften difficulttofind, sug-gesting that the process occurredrapidly. (Com-pounding the problem is the critical angle and levelatwhich thesectionmustpass throughboth viral envelope and cell membrane toreveal their
continuity.)
Figures 4 and 5 illustrate two characteristic examples offusion, which are believed to consti-tutetheinitialstepof
capsid penetration.
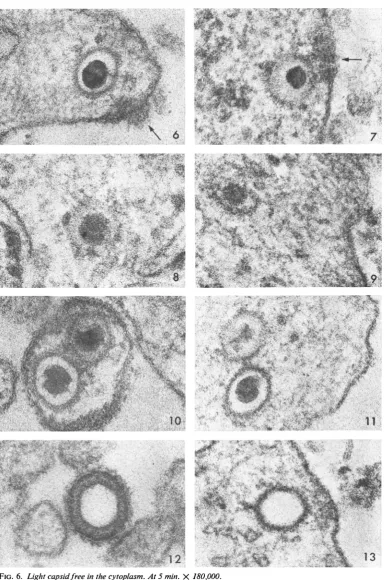
Figures 6and7showlight and dense capsids,respe'
tively,
just beneath the cell membrane. Theaggregateof dense material at the surface (see arrows) was frequently encountered and presumably either represents remnantsof theviral envelopeor signi-fiesanalteration of the cell membraneatthesite ofentry.Note thatthedense
capsid
(Fig. 7) ap-pears tobelesswelldefined, suggesting thatit is in process ofdissolution. More advanced stages of capsid dissolution are seen in Fig. 8 and 9. Figure 10 shows both types ofcapsids enclosed within a single viral envelope, whereas Fig. 11illustrates
similar capsids side by side in the cytoplasm close to the cell surface. In the latter example, it is again evident that the dense capsid appears to be disintegrating. Through-out the study, only the dense capsids were seen atvarious stagesofdisruption
in the cytoplasm, an event most commonly encountered 20 min afterwarming
the cells to 37 C. Presumably, the core is released soon after thecapsid
enters the cytoplasm, since it was almost always ob-served closetothe cell surface.Figure12
illustrates
aviralparticlethatlacks a core. Such particles constituted 4 to7%O
ofany given inoculum. It is hardly surprising that empty capsids gained entry to the cytoplasm (Fig. 13), since theirenvelopes appearedto be no different from thoseof viral particles possessing the dense core, whichispresumed toconsist of nucleopro-tein. Pinocytotic vesicles could be readily distin-guished from such capsids, because the former exhibit aclosely spaced double membrane and the latterdonot.911
VOL.8,
1971on November 11, 2019 by guest
http://jvi.asm.org/
;,:./tA
*8u , .4t~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~3Os I
t;;ts*it
iE s t
I
;'';
~ ~
'';' rfl
sst;>- ai t -1 - XI
..t
.'AsP
'e;sI4
.. ;*E_,'
0
.'½
Si
2
.^S-
4 s .FIG. 1. Thlree characteristictypesofvirions. In thispaper,particleA hasbeenttermed thelight capsid formand particleBthedensecapsid. Asignificacnt proportion ofthe latterexhibitedpoorly defined capsids,asshownl byC.
At2 miii. X 100,000.
FIG.2and 3. Viralparticleswithill-defined capsidsclosetothecellsurface.At 2min. X180,000.
FIG.4and 5. Twoexamples offusionbetweenthecell(totheleft)andtheviralenvelope.At2and5mini, respec-tively. X 180,000.
912
3
on November 11, 2019 by guest
http://jvi.asm.org/
[image:3.486.64.446.34.599.2]~~~~4~~~~~~~4
v
. e
A?A
44
it'ste'-5get.-..I< - vt>; \
[image:4.486.51.436.24.605.2]..4~~~~~.
FIG. 6. Liglhtcapsidfreeinthecytoplasm.At5 min. X 180,000. FIG. 7. Deniseill-defined capsid.At5 mitt.X 180,000.
FIG. 8 and 9. Capsids undergoinigdissolutionat30anid10min,respectively.X 180,000.
FIG. IO. Lightanddensecapsidswitliinasingle envelopeatthecellsurface.At 10 min. X180,000.
FIG. 1 1. Liglhtand densecapsidsfreewithinith1ecytoplasmclosetothe cellsurface. Thelattercapsidappears to
beinprocessofdissolution. X 180,000.
FIG. 12. Emptycapsidatthe cellsuirface.At2mini. X 180,000.
FIG. 1 3. EmptycapDsidwithinz thecytoplasm.At5 min. X180,000. 913
on November 11, 2019 by guest
http://jvi.asm.org/
MIYAMOTO AND MORGAN
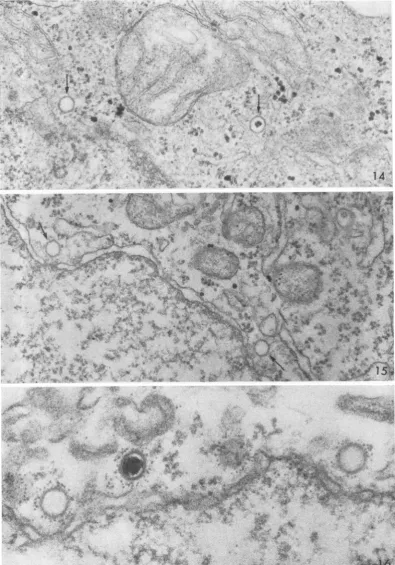
Tenminutes after warmingthecells, light
cap-sids wereencountered in the cytoplasm nearthe
cellsurface, but by 20to 30min the majority had passedtotheperinuclear region and had lost their densecores.In Fig. 14, for example (at 30min),a
light capsid containing a core is visible on the
right about halfwaybetween the cell surface (up-per right corner) and the nuclear membrane
(lower left). Another
capsid,
onthe left in closeproximitytothe nuclear membrane,appearstobe intactbut is devoid ofa core.During theensuing
30min,empty capsidsincreased in number. Two typical empty capsids, each close to a nuclear pore, aremarked byarrowsinFig. 15. With
fer-ritin-conjugated 7S antibody, itwas possible to
demonstrate by specific tagging that such struc-turesareindeed viralcapsids (Fig. 16). Asseenin
this micrograph, occasional capsids inproximity
to the nucleus contained a core, but they were
greatly in the minority.
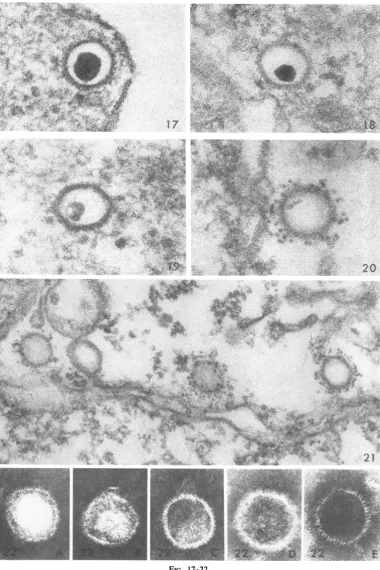
Theprobablesequenceofeventsleading tothe
empty capsids is shown in the following five
fig-ures. Closeto the surface, after 5 min (Fig. 17), the cores exhibited a density and size that are
characteristic of infectious virus. During migra-tiontothenucleus,mostbecamesmaller (Fig. 18,
20min)and less dense (Fig. 19, 30min).Closeto
thenucleus,althoughafewcapsids still retaineda
fragment of densematerial (Fig. 20, 2 hr, ferritin-tagged), the majority were empty (Fig. 21, 2 hr, ferritin-tagged). Dense intranuclear clumps of granular material frequently were seen in
prox-imitytonuclearpores.Suchstructuresmay repre-sentaggregates ofnucleoprotein thataccumulate
atspecificsites afterreleasefromthe capsids. To ascertain whether changes in thecore
visu-alized by negative staining could be correlated with those observed in thinsections, viruswas
pre-pared as described aboveand examined. Figure
22(A-E) illustrates characteristic virions and shows that all of the gradations from full to empty capsids could readily beseen.
Timecourse of cell-associated virus. To obtain
quantitative data concerning the sequentialevents
associated with viralentry, statisticalcountswere
made of thelocation of viralstructuresinsections of 100 consecutivecells examinedat5,10, 20, 30, 40, 50,60, and 120min after warming thecultures
to 37 C.
Figure 23 shows that the disappearance of enveloped virus at the cell surface was
accom-panied by the accumulationofcapsidsinthe cyto-plasm. Significantly, the number of virions in cytoplasmic vacuoles (pinocytosis) remainedata
relatively low level. It will alsobenoted thatthere
was a discrepancy between disappearance of
ex-tracellular virus and increase inintracytoplasmic
capsids; i.e.,after 10, 20, and 30 min, 52, 44,and 31%,respectively, of the viruslostfromthe sur-face could not be seenin the cytoplasm. Lastly, theprocess ofentryalmostentirelyceasedtoward the end of the first hour, the extracellular virus thenremainingonthe cellular surface.
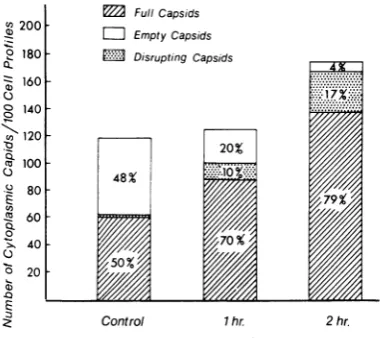
Figure24 showsthe fate of theintracytoplasmic particlesatsequential intervals afterentry. Dense capsids in the process of disruption were most frequently encountered at early stages before emptycapsidshadbeguntoincrease significantly. The fact that the number of empty capsids was inversely proportional to the number of full cap-sidssuggeststhat thelatterwere derived fromthe former. It should be emphasized that, as stated above, only 4 to 7% of the inoculum contained emptycapsids,whereas by 1 hr65%, and by 2 hr 90%oftheintracytoplasmic capsidswereempty. Effectsofinhibitorsoncapsiddisruptionand the accumulation of empty capsids. Cells were fixed for1 hrafter addition of virusat 37 C and differ-entialcountsin100consecutivecells werecarried outin amanner similar to thatdescribedabove. Puromycin. Pretreatment ofthecells for1 or 2 hr had no effect on viral entry, but there was a marked change in the number as wellas thetype of intracellularcapsids (Fig. 25). The percentage of fullcapsids increased from 50% inthecontrols to
79%O
in cellspretreated with the inhibitorfor 2 hr, whereas the percentage ofemptycapsids fell from48%o
in the controlsto 4% (2% ofthe cap-sids inthe controls and 17% in the treated cells wereobserved inprocess ofdisruption). Itis evi-dent that puromycin markedly inhibited the loss ofcores from capsids and slowed down capsid dissolution.Actinomycin D. The effects of the drug were similarto those of
puromycin.
Incells pretreated for2 hrwith actinomycinDbeforeinfection,81% ofviralcapsids
werefullas compared with 48% in the controls, and only9% ofthecapsids were empty as compared with 50% in the controls. Whenactinomycin
Dwasadded 10minafter the cells were infected, the percentages of full and empty capsids were similar to those in the con-trols.UV-irradiated virus. Irradiation of the inoc-ulumfor5mindidnotaltercapsid
entry,
but the percentage offull capsidsatthe end of1 hrrose from 35 (inthe controls) to 70% Irradiationfor 10minreduced
the number ofintracellular cap-sidsby
30%,
but 97% ofthem contained cores. After 30 min ofirradiation, whichdestroyed
all infectivity, no free intracytoplasmic capsidswere encountered, although the rate ofviropexis
re-mained unchanged in comparison with the con-trols.914 J. VIROL.
on November 11, 2019 by guest
http://jvi.asm.org/
.. *; / ..K
*|'
A
¢~ ;4~**v'.t~~~~~~~~~WIf*.*.;5.
e'tkt'
e . ,^ ;'PtZ ii r-*< m .5
*<
n t lit AW*D k,~~P1~4
5',. %tt 2It.
.^ '''s}w'i,,. r' S a i h
'"''g
;'t4! %4L <;: .
AL,
s
-88]rms
rip;....$
a
~~~~~~~'k412
.
M,f
Ac*$P
XZ,:I, } ,f'.
a
.RS: W
* >S S,@ \.e
*
.}s....r.
.>s-> 84
.'....SiS..
V"
...1a4
FIG. 14. Full, light capsid halfwaybetween the cell surface(upperright) and the nucleus (lower left). Close to the nuclearmembraneisanemptycapsid.Thecapsidsareindicated by arrows. At 30min.X60,000.
FIG. 15. Twoemptycapsids (marked by arrows) close to pores of the nucleus at the lower left. The hexagonal
shapeof the capsids isevident.At 60min. X 60,000.
FIG. 16. Two emptycapsidsand one containing a core have been tagged with ferritin and are in proximity to the
nucleus, whichoccupiesthe lowerportionofthefield. X100,000.
915
..4
Pt, dt4_
14
'
,,R -1- ;~,
r M
.z .4
-5~ -I".
r
..
?I. '.
on November 11, 2019 by guest
http://jvi.asm.org/
[image:6.486.44.440.48.614.2]916 MIYAMOTO AND MORGAN
J.
VIROL.1
7
20
FIG. 17-22.
on November 11, 2019 by guest
http://jvi.asm.org/
[image:7.486.62.443.69.640.2]UNCOATING OF HERPES SIMPLEX VIRUS
DISCUSSION
The presence oftwo distinct types of capsids raises the possibility that theyservedifferent
func-tions in the process ofinitiating infection. Only dense capsids aredisrupted, aneventthatoccurs
v,
250
-Q.
00 150
-_l
0Q
50
Cytoplas,imcCaps,ds 0
-- -Pa,ices -scles
0 10 20 30 40 50 60 120
TIME(minutes,
FIG. 23. Extracellular virions, intracellullar capsids, anid virions within vesiclesat intervals after infectioni. The cells were inoculated with conicentrated virus anid heldat4Cfor Ihrtoallow atiachmenit. The cultures
were warmed rapidly, anid 100 conisecutive cells were
examinledatthe inidicated times.In thisanzd the follow-in2gtwofigures,4to5%G ofcapsidscomprisinigthe iniocu-lumwereempty.
--
-A0-o~~~~~~~~~~~~7
c -,A
A--_ _-- -Al Dsu, -ri Ca3s
_~~~~~--- _
C 10 20 30 40 50 60 120
[image:8.486.246.437.68.237.2]TIME(minutes'
FIG. 24. Capsids containzing cores, empty capsids,
antd capsids in process ofdisrutptioti at intervalsafter inzfectioni. The cellsweretreatedasdescribedinFig.23. The timne inidicatesperiodsat 37 C. Onehiundred
conI-secutive cellswereexaminied ineachpreparationi.
9, 200 ._?
° 180
Q.
, 160
Q.)
° 140 \ 120
cu 100
C.)
- 80
2 60
Q.
0
>. 40 ° 20
a)
E
3 FullCapsids
= Empty Capsids
M DisruptingCapsids
Control 1hr 2 hr.
TimeofPretreatment with 20pg/mlofPuromycin
FIG.25. Effect ofputromycin oni capsid disruption anzd loss ofcorematerial. The cellswereexamined1hr
after inoculation of virus at 37 C, durinig which time the treated cells were incubatediin thecontinzued
pres-ence of puromycini. Thepercentages show the relative
n2umbers of capsidsconitainiingcores, capsidsdevoidof
cores,and capsidsinprocessofdisruiptioni.
rapidly and closetothe site ofentry. Presumably, theyare unstable. (Instability ofthedense capsid
is further suggested by the fact that, inanygiven
inoculum,
nearly half of themare poorlydefinedandappeartohave undergone partial disintegra-tion even before entry into the cell.) The light capsids, on the otherhand, are relatively stable,
for theyarerarely disrupted and, during thefirst hour at least, accumulate within the cytoplasm andmigrate toward thenucleus.The progressive
increase in the percentage of empty capsids, as
opposed tothosecontaining acore, suggeststhat thelight capsidsbecomepermeablewithresulting
escape ofthenucleoprotein. Incontrastto adeno-virus, whose core material can actually be
ob-served at stages ofrelease from capsids close to
the nucleus (6), the deoxyribonucleoprotein of herpes simplexvirusappearstobereleasedslowly, rather thanejectedatagivenmoment,anddirect
FIG. 17. Capsidclose to the cell membrane shortly after entry. Thecore islarge, dense, anidsharply defined. At5miin. X 180,000.
FIG. 18. Capsidmidwaybetweeln the cellsuirfaceand thenucleus. Tlhecore issmallbuitstilldenlse.At 20miii.X
180,000.
FIG. 19. Capsidconitainiiigasmall, irregularly shapedcoreoflowdenisity.At 2hr.X 180,000.
FIG. 20. Ferritin-taggedcapsid closetothe nulcleus (attheleft). Afragmenit ofthecore isvisible. At 2 hr. X
180,000.
FIG.21. Thlreeempty capsids closeto the niuclearmembrane (lower portiont ofthefigure). Thecapsidscanl be distiiiguished from the vesicle ofsimilarsize (left third) by thepresenceof specificJerritin label. Qfparticular
inlterest isthepreseiice of ferritiln within thecapsidiiearthe center, suggestingthatthecapsidis ntowpermeable. At 2hr. X 100,000.
FIG. 22. (A-E)Negativelvstainiedpreparatioiisshowing capsidssimilarto thoseenIcounlteredinsectioiis.
VOL. 8, 1971 917
on November 11, 2019 by guest
http://jvi.asm.org/
[image:8.486.41.230.128.270.2] [image:8.486.43.234.356.486.2]MIYAMOTO AND MORGAN
passage ofaggregated nucleoprotein into the nu-cleus has not been detected. Studies with ferritin-labeledanti-DNAantibodies, which are currently in progress, may help to clarify the manner in whichthe corematerial gainsentry to the nucleus. UV irradiation appears to have sequential effects. Initially the permeability of the capsid or theconfigurationofthenucleoprotein is presum-ably altered so that release of core material is in-hibited or prevented. Further irradiation appar-ently affects the envelope so that attachment occurs but fusion with the cell membrane does notresult.[It is of interest that cellular fusionwas alsoprevented byUVirradiation,anobservation which supports aprevious suggestion (7) that in the case of herpes simplex virus, at least, viral entryandcellfusionmay berelated phenomena.] The effects ofpuromycin and actinomycin D are consistent with the hypothesis that protein synthesis, presumably mediated by viral
entry,
is necessary for the release of nucleoprotein from thelight capsids.Theforegoing datasuggestthefollowing mech-anisms whereby herpes
simplex
virus initiates infection. After attachment, the viral envelope fuseswith the cellmembrane,permittingegressof capsidsto the cytoplasm. Thecapsids
are oftwo distinct types, light and dense. The former are stable andmigrate
toward the nucleus, whereas the latter are unstable andrapidly disrupt
near the cell surface. Two different interpretations of subsequent events can be proposed. (i) The dense particlesare inertanddonotcontributeto theinfectiousprocess. In this event, DNA inthe light particles codes for avirus-specific
enzyme, which willultimatelyrenderthecapsidpermeable
to theviral DNA. If such is the case, either the virions contain an as yet undiscovered DNA-dependent RNA
polymerase
similar to the en-zyme found in vaccinia virus(3,
4)
or a host RNA polymerase is able to reach the enclosed viralDNAandtranscribe it there.(The
latter pos-sibility seems remote since this reaction would have to occurin thecytoplasm,
whichis poorin RNApolymerase.)(ii)
Bothlight
anddense par-ticles arerequiredfor the infectious process. The denseparticles,
which are unstable after removal of theviralenvelope,releasetheirDNA nearthe cellsurfaceveryshortly
afterentry. This DNAisthentranscribedinto a virus-specific RNA, which in turn codes for an enzyme capable of rendering thelightparticles permeable and thereby allowing release ofinfectious DNA in close proximity to the nucleus. It is of interest in thisconnection that Hochberg andBecker (2) reported that only about 40% of the viral DNA becomes deoxyribonu-clease-sensitive inthe presence of puromycin. This probably corresponds to the DNA in the dense cores, which we suggest is uncoated shortly after entry. The remainder of the DNA would remain within the capsid if, in fact, an enzyme must be synthesizedto permit release.
Itistempting to postulate that toinitiate infec-tiontwo types ofparticlesmay benecessary in the case ofcertain other viruses, such as members of the poxvirus group.
ACKNOWLEDGMENTS
This investigation was supported by Public Health Service grantAI-06814from the National Institute ofAllergyand Infec-tious Diseasesand bygrant GB-6013 from theNationalScience Foundation.
LITERATURE CITED
1. Hampar,B.,A. L.Notkins,M.Mage, andM. A. Keehn.1968. Heterogeneity in thepropertiesof7Sand19S rabbit-neutra-lizing antibodiestoherpes simplex virus. J. Immunol. 100: 586-593.
2. Hochberg, E., andY.Becker. 1968. Adsorption, penetration and uncoating ofherpessimplexvirus.J.Gen.Virol. 2:231-241.
3. Kates, J.R.,and B. R.McAuslan. 1967.MessengerRNA syn-thesis bya "coated" viral genome. Proc. Nat. Acad. Sci. U.S.A. 57:314-320.
4. Kates, J. R., and B. R. McAuslan. 1967. Poxvirus DNA-dependent RNA polymrease. Proc. Nat. Acad. Sci. U.S.A.
58:134-141.
5. Morgan, C.,H. M.Rose,and B.Mednis. 1968. Electron micros-copy ofherpes simplex virus.1.Entry.J.Virol. 2:507-516. 6. Morgan,C.,H.S.Rosenkranz,and B.Mednis. 1969. Structure
and development of viruses as observed in the electron microscope. X.Entry anduncoatingof adenovirus.J.Virol. 4:777-796.
7. Nii,S.,C.Morgan,H. M.Rose, andK.C.Hsu.1968.Electron microscopyofherpes simplex virus.IV.Studies with ferritin-conjugated antibodies.J.Virol.2:1172-1184.
8.Rifkind,R.A.,K. C.Hsu,andC.Morgan. 1964. Immuno-chemical staining for electron microscopy. J. Histochem. Cytochem.12:131-136.
9. Spring, S. B., and B. Roizman. 1967. Herpessimplex virus productsinproductiveand abortiveinfection.I.Stabilization with formaldehyde and preliminary analyses by isopycnic centrifugationinCsCI.J. Virol.1:294-301.
918 J. VIROL.