Original Article
Protective effects of curcumin on hepatocytes in cecal
ligation and puncture-induced sepsis in rats
Hai-Yan Yin1, Pei Tao1, Jian-Rui Wei2, Rui Zhang1, Yong-Hui Ma1
1Department of Intensive Care Unit, Guangzhou Red Cross Hospital, Medical College, Jinan University, Guangzhou 510220, PR China; 2Department of Cardiology, Guangzhou Red Cross Hospital, Medical College, Jinan University, Guangzhou 510220, PR China
Received November 14, 2015; Accepted August 13, 2016; Epub December 15, 2016; Published December 30, 2016
Abstract: We aim to observe the protective effects of curcumin in hepatocytes in septic rat. Sprague-Dawley rats were randomly divided into six groups of 20 rats each: sham group; model group; xuebijing group; low dose group (50 mg/kg), middle dose group (100 mg/kg) and high dose group (150 mg/kg). Another 60 rats were randomly divided into the six above-mentioned experimental groups of 10 rats each to determine the survival rate. Our results showed that, treatment with curcumin and Xuebijing significantly attenuated the CLP-induced hepatocyte edema and inflammation indicators, especially in the Xuebijing and 100-mg/kg curcumin groups. This result was also sup -ported by the histopathologic examination findings. Additionally, curcumin improved the survival rate of rats with CLP. Curcumin has a protective effect on CLP-induced liver dysfunction and that the effect varies at different doses; the 100-mg/kg dose was optimal for protection of hepatocytes in septic rats. This protective effect can be attributed to the ability of curcumin to counteract inflammatory cell infiltration and regulate cytokines.
Keywords: Curcumin, sepsis, hepatocyte, cecal ligation, puncture
Introduction
Sepsis is a systemic, deleterious host response to infection and a common complication of burns, trauma, shock, and major surgery. It may lead to septic shock, multiple organ dysfunc-tion syndrome, and other serious condidysfunc-tions. The mortality rate associated with sepsis is extremely high; the overall hospital mortality rate is reportedly 28.6%, while the mortality rate associated with severe sepsis and septic shock are higher at 25% to 30% and 40% to 70%, respectively [1, 2]. Sepsis has become the leading cause of death in the intensive care unit, causing more deaths than acute myocar-dial infarction, lung cancer, and breast cancer and killing 1,400 people worldwide each day [3-5]. Moreover, the cost associated with sep-sis is remarkably high. According to an epide-miological survey, more than 370,000 people die of sepsis in Europe and the United States each year, and the cost of treatment reach up to $25 billion [6]. Sepsis has thus become a serious problem and challenge to human health [7]. The development of effective treatments
and drugs for sepsis is significant and
emergent.
As the central organ of metabolism, the liver is one of the most important organs participating in the elimination of bacteria and endotoxins from the body [8-10]. Numerous studies have
identified a close relationship between the
development of liver injury and the occurrence and aggravation of sepsis and septic shock
[11]. Liver dysfunction plays a critical role in the
disease course [9]. Patients with sepsis have
significantly high levels of oxygen free radicals and release of inflammatory mediators, which
occurs secondary to increased membrane per-meability caused by damage to the liver cell structures and mitochondrial membranes. Sodium pump dysfunction leads to sodium
retention, hepatocyte swelling, and finally hepa -tocyte apoptosis [12-15].
anti-oxi-dation, and anti-inflammation effects [16].
Curcumin also exhibits relatively low toxicity, has a simple extraction process, and is low in
cost. Curcumin has been confirmed to be
extremely safe in various animal models and clinical studies, even at the maximum daily dose of 12 g, and can be administered topical-ly, oraltopical-ly, and by inhalation. Therefore, curcumin shows great potential for clinical applications [17]. Curcumin has a dual role in apoptosis, showing different reactions in different cells. Previous studies have revealed that curcumin exerts a protective effect against liver cell inju-ry caused by multiple factors including ethanol-induced and carbon tetrachloride-ethanol-induced liver cell damage [18-20]. The underlying mecha-nism involves increased superoxide dismutase
vitality, regulation of inflammatory and anti-inflammatory cytokine expression, inhibition of
cytochrome C activity, and other processes. Although curcumin has been shown to be
ben-eficial in sepsis, its potential value in protection
of sepsis-induced hepatocyte dysfunction has not been evaluated [20, 21].
Therefore, the present study was designed to demonstrate the protective effect of curcumin and its possible mechanism on hepatocytes in septic rats. We also explored the potential clini-cal use of curcumin for treatment of sepsis-induced liver dysfunction.
Materials and methods
Experimental animals
Male 3-month-old specific-pathogen-free
Spra-gue-Dawley rats weighing 250 to 350 g were
obtained from the Laboratory Animal Center of
Sun Yat-sen University (Guangzhou, China). The rats were maintained in cages in a tempera-ture-controlled room at 25°C±1°C, under an
artificial 12-h light-dark cycle, and on a stan -dard diet and water. Animal housing and all experimental procedures were approved by the
Institute of Laboratory Animal Science, Jinan
University.
Chemicals and reagents
Curcumin of analytical grade was purchased
from Sigma-Aldrich (St. Louis, MO, USA).
Xuebijing (XBJ) (batch number 1308221) was supplied by Tianjin Chase Sun Pharmaceutical
Co., Ltd. (Tianjin, China). All other chemicals
and biochemicals used in this study were of high analytical grade.
Animal groups
Sprague-Dawley rats were randomly divided into six groups of 20 rats each: animals
under-going sham CLP (Sham group), animals under
-going CLP (CLP group), animals under-going CLP
and treated with XBJ (XBJ group), and animals
undergoing CLP and treated with curcumin at 50 mg/kg (low-dose curcumin [L-Cur] group),
100 mg/kg (middle-dose curcumin [M-Cur] group), or 150 mg/kg (high-dose curcumin [H-Cur] group). Curcumin was diluted with nor-mal saline solution to 10 ml/kg and freshly pre-pared on the day of the experiment. It was
administered intraperitoneally after CLP. The
rats received an additional dose of curcumin at
8, 16, and 24 h post-CLP.
Another 60 experimental rates were observed to determine the survival rate. The rats were randomly divided into the six above-described experimental groups of 10 rats each. XBJ and curcumin were intraperitoneally injected three times a day beginning after surgery and con-tinuing for 3 days. The survival rates were recorded at 12, 24, 36, 48, and 72 h after surgery.
Establishment of animal model
The rat models of sepsis in this study were
layer by layer [22]. After the surgery, sterile
nor-mal saline solution (100 mL/kg of body weight) was administered for fluid resuscitation.
In the Sham group, the abdominal cavity was
opened and the cecum was flipped over and
placed back into the abdominal cavity without ligation or puncture; the remaining steps were the same.
Specimen collection
The animals were anesthetized with 2% sodium
pentobarbital at 2, 6, 12, and 24 h post-CLP (n
= 5 per time point), and the abdominal cavity was opened to collect liver tissue specimens and portal venous and peripheral venous blood samples. Some samples were stored at -80°C until analysis.
Histological evaluation
The harvested liver specimens were fixed in
10% paraformaldehyde for 24 h, dehydrated in
a graded alcohol series, embedded in paraffin,
and serially sectioned. Some of the specimens were stained with hematoxylin and eosin to assess morphological changes. The remaining specimens underwent terminal deoxynucleoti-dyl transferase-mediated dUTP nick end
label-ing (TUNEL) stainlabel-ing uslabel-ing an In Situ Cell Death Detection Kit (Hoffmann-La Roche, Ltd., Basel,
Switzerland). The occurrence of tissue damage was evaluated independently by a pathologist and histologist blinded to the experiment.
Serological evaluation
The serum concentrations of procalcitonin
(PCT), tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β) were analyzed by
enzyme-linked immunosorbent assay (Boster Biological
Engineering Co., Ltd., Wuhan, China). The plas -ma samples were centrifuged to separate the serum. All experimental procedures were car-ried out according to the manufacturer’s instructions.
Statistical analysis
All experimental data were statistically pro-cessed using SPSS 13.0 software (SPSS, Inc.,
Chicago, IL, USA). Measurement data are
expressed as mean ± standard error, and a
t-test was performed. Multiple comparisons
were analyzed for significant differences using
one-way analysis of variance with Tukey’s post hoc test for multiple comparisons. A P level of <
0.05 was considered statistically significant.
Results
Histopathologic findings (Figure 1)
In the Sham group, the liver tissues exhibited normal histological features at all postopera-tive time points, including normal hepatocytes, portal areas, and parenchyma.
[image:3.612.91.523.71.247.2]observed. The central vein and hepatic sinus were conspicuous, sinusoids were mildly dilat-ed, and ballooning degeneration of
hepatocy-tes was partly visible among periportal inflam
-matory cell infiltration. The XBJ and curcumin
groups also showed varying degrees of histo-logical changes, central vein and hepatic sinus congestion, and sinusoidal dilation. Neverthe-
less, this congestion and dilation were signifi
-cantly less severe than those in the CLP group. Six hours after surgery in the CLP group, central
vein and hepatic sinus congestion and sinusoi-dal dilation were widespread. Hepatocyte
hypertrophy and infiltration of inflammatory
cells were more visible. A small amount of liver cell degeneration and punctate eosinophilic necrosis could be seen at this time point. In the XBJ and curcumin groups, the degrees of cen-tral vein and hepatic sinus congestion and sinu-soidal dilation were aggravated, but still less
severe than those in the CLP group.
Twelve hours after surgery, the range of dam-age continued to expand in all groups. Hepa- tocytes showed varying degrees of irregular
arrangements. Fragmentation of hepatic cell
cords and multiple spotty hepatocyte necroses
were widespread in the CLP group. Lesions in the XBJ and curcumin groups were still signifi
-cantly less severe than those in the CLP group.
Twenty-four hours after surgery in the CLP
group, the injury was further aggravated, show-ing a large area of hepatocyte edema and necrosis, accumulation of swollen hepatocytes,
and inflammatory cell infiltration. No worsening
was noted in the XBJ group or curcumin groups.
TUNEL results
[image:4.612.91.522.86.190.2]Stained apoptotic nuclei appeared as blue-black granule deposits [23]. The apoptotic index was calculated with a formula, the ratio between the number of aptptotic cells and the
Table 1. The changes of apototic index (%) in different time phase
Group Different Time Phase
2 H 6 H 12 H 24 H
A 2.02±0.13 2.10±0.13 2.20±0.05 2.14±0.06
B 23.59±2.00* 30.92±1.69* 50.18±2.11* 52.05±1.31*
C 11.89±1.34*,# 17.76±1.73*,# 27.08±1.64*,# 28.95±1.40*,#
D 17.59±1.43*,#,▲ 23.07±1.18*,#,▲ 33.76±1.58*,#,▲ 34.89±1.76*,#,▲
E 11.56±0.96*,# 18.36±1.10*,# 28.25±1.20*,# 30.35±1.20*,#
F 17.48±1.94*,#,▲ 23.45±2.01*,#,▲ 34.25±1.41*,#,▲ 35.92±1.79*,#,▲
A: Sham group. B: CLP group. C: XBJ group. D: L-Cur group. E: M-Cur group. F: H-Cur group. L-Cur, low-dose curcumin, 50 mg/kg i.p.; M-Cur, middle-dose curcumin, 100 mg/kg i.p.; H-Cur, high-dose curcumin, 150 mg/kg i.p. Data are presented as the mean ± standard error (n = 5). *P < 0.05 vs. Sham group; #P < 0.05 vs. CLP group; ▲P < 0.05 vs. XBJ group.
[image:4.612.91.527.243.417.2]total number of cells analyzed multiplied by
[image:5.612.93.372.69.275.2]100 (Table 1). In the Sham group, few to no lower than those in the L-Cur and H-Cur groups (P < 0.05) (Figures 4, 5). Figure 3. Effects of curcumin on the apoptotic index at 6, 12, and 24 h after
surgery. A: Sham group. B: CLP group. C: XBJ group. D: L-Cur group. E: M-Cur group. F: H-M-Cur group. L-M-Cur, low-dose curcumin, 50 mg/kg i.p.; M-M-Cur, middle-dose curcumin, 100 mg/kg i.p.; H-Cur, high-dose curcumin, 150 mg/ kg i.p. Data are presented as the mean ± standard error (n = 5). *P < 0.05 vs. Sham group; #P < 0.05 vs. CLP group; ▲P < 0.05 vs. XBJ group.
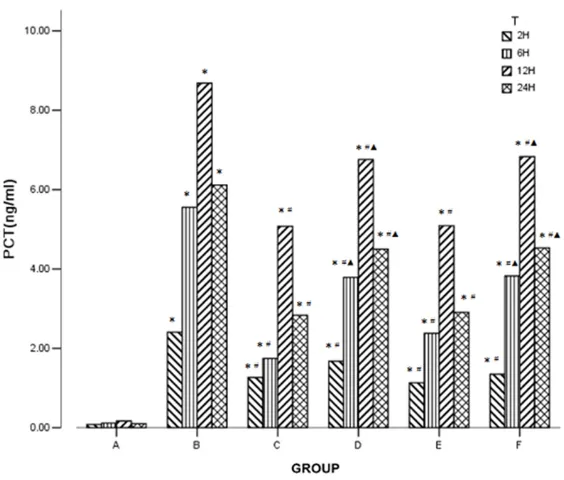
Figure 4. Effects of curcumin on PCT level in peripheral blood at 6, 12, and 24 h after surgery. A: Sham group. B: CLP group. C: XBJ group. D: L-Cur group. E: M-Cur group. F: H-Cur group. L-Cur, low-dose curcumin, 50 mg/ kg i.p.; M-Cur, middle-dose curcumin, 100 mg/kg i.p.; H-Cur, high-dose cur-cumin, 150 mg/kg i.p. Data are presented as the mean ± standard error (n = 5). *P < 0.05 vs. Sham group; #P < 0.05 vs. CLP group; ▲P < 0.05 vs. XBJ group.
apoptotic cells were found at each time point. More apop-totic cells were observed in the other groups, with varying de-grees of cytoplasm concentra-tion and karyopyknosis (Figure 2).
Precise information was re-vealed by quantitative analy-sis. The apoptotic index was
significantly higher in the CLP
group than in the XBJ and cur-cumin groups (P < 0.05). The index continued to rise with
time in the CLP group (P < 0.05), while the upward trend ended between the 12- and 24-h time point in the XBJ and curcumin groups (P > 0.05). Additionally, the index was not
significantly different between
the XBJ and M-curcumin groups (P > 0.05), whereas it
was significantly lower than in the L-curcumin and H-curcumin
groups (P < 0.05) (Figure 3).
Serological evaluation
The PCT, TNF-α, and IL-1β lev -els were elevated in all groups
from 2 h post-CLP and were significantly higher in all groups
than in the Sham group (P < 0.05); the most obvious
in-crease was in the CLP group (P
< 0.05). The PCT, TNF-α, and IL-1β levels changed only
sli-ghtly at each time point in the Sham group (P > 0.05), while they continued to rise in the
CLP group (P < 0.05). The lev-els of each parameter peaked
at 12 h post-CLP in the XBJ
group and curcumin groups
and were significantly lower than those in the CLP group (P
< 0.05). Additionally, the differ-ence between the XBJ and M-Cur groups was not
statisti-cally significant (P > 0.05), but
[image:5.612.91.373.366.606.2]Survival study results
The rats in the CLP group of the survival study
exhibited a 72-h survival rate of only 20%. Conversely, only rats in the XBJ group and cur-cumin groups showed 30% to 50% mortality,
which was significantly lower than that in the CLP group (P < 0.05). All rats in the sham group survived to the 72-h time point (Figure 6).
[image:6.612.92.520.71.237.2]easily cause abdominal infection. This disease process is similar to that of peritonitis second-ary to perforating appendicitis in clinical prac-tice. Studies have shown that the animal exhib-its a high-power cycle and metabolic state in the early stage, and then enters a low-power cycle later. Additionally, compared with other animal models both the endotoxin detection rate in peripheral blood and bacterial culture-Figure 5. Effects of curcumin on cytokine production in peripheral blood at 6, 12, and 24 h after surgery. A: Sham group. B: CLP group. C: XBJ group. D: L-Cur group. E: M-Cur group. F: H-Cur group. L-Cur, low-dose curcumin, 50 mg/ kg i.p.; M-Cur, middle-dose curcumin, 100 mg/kg i.p.; H-Cur, high-dose curcumin, 150 mg/kg i.p. I: TNF-α. II: IL-1β. Data are presented as the mean ± standard error (n = 5). *P < 0.05 vs. Sham group; #P < 0.05 vs. CLP group; ▲P < 0.05 vs. XBJ group.
Figure 6. Effects of curcumin on survival of rats with CLP-induced liver dys -function. L-Cur, low-dose curcumin, 50 mg/kg i.p.; M-Cur, middle-dose curcum -in, 100 mg/kg i.p.; H-Cur, high-dose curcum-in, 150 mg/kg i.p. The survival rates were observed within 72 h. The results are expressed as cumulative survival, n = 10. The survival rate was estimated by the Kaplan-Meier method and compared by the log-rank test.
Discussion
Sepsis is a disease syn-drome that involves many different aspects of the host immune system. The initial trigger of the disease may be multifaceted, but the most common source is bac-terial infection, especially from the abdominal cavity. Consequently, various ani-mal models of abdominal infection have been devel-oped to mimic the disea- se progression in sepsis,
among which the CLP model
[image:6.612.94.381.325.513.2]positive rate are higher. The serum cytokine
elevation is relatively flat and longer in dura
-tion. Therefore, CLP animal models have strong
clinical relevance [24, 25]. We thus chose to
perform CLP in the present experiment.
At each time point in this study, animals were
sacrificed by laparotomy. The rats that had undergone CLP exhibited a black, edematous
cecum with purulent exudates in the abdominal
cavity. These findings combined with the histo -logical changes indicated that the septic shock model was successfully established. After treated by XBJ and curcumin, all the parame-ters and histological changes are better than
those of CLP only group, which showed that XBJ
and curcumin were effective.
Liver, as the center of metabolism, is one of the
most important organs of toxins clearance. Numerous studies indicate that liver damage
significantly correlates with the occurrence and
the progression of septic shock. Previous stud-ies suggest that curcumin performs a protec-tive role in relieving hepatic cell damage caused by a variety of factors. This protective effect carried out via the power of increasing superox-ide dismutase (SOD) vitality and inhibiting cyto-chrome C activity [18-20]. Whereas, the effect of curcumin to liver cell in sepsis is still unclear, which is also the aim of the present study. XBJ injection is a traditional Chinese medicine used to treat sepsis and has been approved by
the State Food and Drug Administration of China. The injection combines five traditional Chinese medicines, including Flos Carthami
(the corolla of Carthamus tinctorius L.), Radix
Paeoniae Rubra (the root of Paeonia veitchii
Lynch), Rhizoma Ligustici Chuanxiong (the root
of Ligusticum chuanxiong Hort.), Salviae Miltiorrhizae (the root of Salvia miltiorrhiza
Bge.), and Angelicae Sinensis Radix (the root of
Angelica sinensis (Oliv.) Diels.) [26]. XBJ is wide-ly used in the treatment of sepsis and multiple organ dysfunction syndrome [27-29]. Therefore, we choose the XBJ group as the positive control group to identify the effect of curcumin in sepsis.
PCT, a precursor of calcitonin, is a 116-amino-acid protein secreted in the neuroendocrine cells of the thyroid, lung, and pancreas. PCT can be broken into three distinct molecules with different enzymatic functions: calcitonin
(32 amino acids), katacalcin (21 amino acids), and an N-terminal fragment called aminopro-calcitonin (57 amino acids) [30].
The circulating PCT levels in healthy individuals are very low (usually < 0.1 ng/ml), but rapidly
and significantly increase under conditions of
bacterial infection (> 1000 ng/ml). In sepsis, a rise in the PCT level can be detected at 3 to 4 h, and serial measurements are useful to monitor the response to therapy, embodying good clini-cal acumen [31]. PCT is therefore considered to be a valuable adjunct in the diagnosis and man-agement of sepsis. In the present study, the
PCT level significantly increased in the rats that underwent CLP, whereas it decreased after
treatment with XBJ and curcumin [32]. Therefore, curcumin may effectively control infection, and middle-dose curcumin could work as well as XBJ.
TNF-α is a cytokine with a wide range of biologi -cal effects. It is secreted by macrophages, monocytes, T lymphocytes, and other cells and is synthesized in many tissues and organs
including the heart, liver, and lungs. TNF-α is
mainly secreted by macrophages and plays critical roles in many biological processes, such
as host resistance to infection and the inflam -matory response [33-35].
In one animal experiment, sepsis initiated extensive apoptosis of gastrointestinal epithe-lial and liver cells. This phenomenon then occurred in other organs, resulting in multiple organ failure [36]. Numerous studies have shown that the major promoters of sepsis are
lipopolysaccharides, which finally induce apop
-tosis. TNF-α, as a proinflammatory factor, is the first multifunctional cytokine produced from
lipopolysaccharide-stimulated monocytes and
macrophages [37]. It activates inflammatory cells, which then release IL-1, IL-6, and other inflammatory mediators to extend and expand the inflammatory response, eventually inducing a systemic inflammatory response. IL-1β, a member of the IL-1 cytokine family, is a pleiotro -pic cytokine. It plays a role in the regulation of
systemic and local inflammatory responses
and functions in almost all cells of the human body [38]. Previous studies have suggested
that IL-1β is involved in the apoptosis pathway
physiologi-cal processes such as cell proliferation, differ-entiation, apoptosis, and necrosis [39-41].
TNF-α and IL-1β may therefore elicit the inflam -matory cascade and contribute to the severity of liver injury in sepsis. In the present study,
significantly greater increases in the expres
-sion of TNF-α and IL-1β in the peripheral blood were seen in the CLP group than in the Sham
group, as expected. After treatment with XBJ
and curcumin, the expression of both TNF-α and IL-1β significantly decreased, also as expected. Furthermore, XBJ and M-Cur showed
the best treatment effect.
This study also demonstrated that the protec-tive effect of curcumin varies with the dose used; the middle concentration of curcumin was most effective for septic rat hepatocytes. This might be correlated with the pharmacoki-netics of curcumin. Curcumin is metabolized
mainly by the liver, and the first pass effect is
obvious. After enterohepatic circulation, most ingredients are transformed and cleared [42,
43]. For this reason, the low concentration of
curcumin lacked effective protection. Therefore, in severe sepsis, when large numbers of liver
cells are damaged by inflammatory cytokines
and mediators, a high concentration of curcum-in could curcum-increase the burden of liver metabo-lism. Thus, the middle concentration of curcum-in was most effective for protection of septic rat hepatocytes.
In conclusion, the present study has shown
that curcumin can protect against CLP-induced liver dysfunction by decreasing the TNF-α and IL-1β levels. The pathological examination find
-ings and changes in the PCT level confirmed this conclusion. Furthermore, the present study
found that the protective effect of curcumin may be related to the drug concentration; nei-ther a low nor high dose of curcumin will achieve
the optimal effect. Although we confirmed the beneficial effect of curcumin on hepatocytes in
septic rats, the precise mechanisms remain to be fully elucidated. Clinical trials are also nec-essary to fully realize the potential use of cur-cumin in patients with sepsis.
Disclosure of conflict of interest
None.
Address correspondence to: Dr. Jian-Rui Wei, De- partment of Cardiology, Guangzhou Red Cross
Hospital, Medical College, Jinan University, No. 396 Tongfuzhong Road, Guangzhou 510220, Guandong, PR China. Tel: +8618928900398; E-mail: jianrui_ wei@sina.com
References
[1] Yang Y, Yang KS, Hsann YM, Lim V and Ong BC. The effect of comorbidity and age on hospital mortality and length of stay in patients with sepsis. J Crit Care 2010; 25: 398-405. [2] Schlichting D and McCollam JS. Recognizing
and managing severe sepsis: a common and deadly threat. South Med J 2007; 100: 594-600.
[3] Riedemann NC, Guo RF and Ward PA. The enig -ma of sepsis. J Clin Invest 2003; 112: 460-7. [4] Martin GS, Mannino DM, Eaton S and Moss M.
The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003; 348: 1546-54.
[5] Awad SS. State-of-the-art therapy for severe sepsis and multisystem organ dysfunction. Am J Surg 2003; 186: 23S-30S; discussion 31S-34S.
[6] Yao YM and Sheng ZY. Fresh advances in basic scientific research on sepsis in China. Chin J Trauma 2003; 19: 9-12.
[7] Wang P, Ba ZF and Chaudry IH. Hepatocellular dysfunction occurs earlier than the onset of hyperdynamic circulation during sepsis. Shock 1995; 3: 21-6.
[8] Weis SN, Pettenuzzo LF, Krolow R, Valentim LM, Mota CS, Dalmaz C, Wyse AT and Netto CA. Neonatal hypoxia-ischemia induces sex-relat-ed changes in rat brain mitochondria. Mitochondrion 2012; 12: 271-9.
[9] Gantner F, Uhlig S and Wendel A. Quinine in -hibits release of tumor necrosis factor, apopto-sis, necrosis and mortality in a murine model of septic liver failure. Eur J Pharmacol 1995; 294: 353-5.
[10] Xie J, Lv R, Yu L and Huang W. Hydroxyethyl starch 130/0.4 exerts its anti-inflammatory ef -fect in endotoxemic rats by inhibiting the TLR4/ NF-kappaB signaling pathway. Ann Clin Lab Sci 2010; 40: 240-6.
[11] Deutschman CS, Cereda M, Ochroch EA and Raj NR. Sepsis-induced cholestasis, steatosis, hepatocellular injury, and impaired hepatocel-lular regeneration are enhanced in interleu-kin-6 -/- mice. Crit Care Med 2006; 34: 2613-20.
[12] Crouser ED. Mitochondrial dysfunction in sep-tic shock and multiple organ dysfunction syn-drome. Mitochondrion 2004; 4: 729-41. [13] Sakaguchi S and Furusawa S. Oxidative stress
during endotoxemia. FEMS Immunol Med Microbiol 2006; 47: 167-77.
[14] Albuszies G, Radermacher P, Vogt J, Wachter U, Weber S, Schoaff M, Georgieff M and Barth E. Effect of increased cardiac output on hepatic and intestinal microcirculatory blood flow, oxy -genation, and metabolism in hyperdynamic murine septic shock. Crit Care Med 2005; 33: 2332-8.
[15] Ammon HP and Wahl MA. Pharmacology of Curcuma longa. Planta Med 1991; 57: 1-7. [16] Sharma RA, Gescher AJ and Steward WP.
Curcumin: the story so far. Eur J Cancer 2005; 41: 1955-68.
[17] Yun SS, Kim SP, Kang MY and Nam SH. Inhibitory effect of curcumin on liver injury in a murine model of endotoxemic shock. Biote-chnol Lett 2010; 32: 209-14.
[18] Sompamit K, Kukongviriyapan U, Nakmareong S, Pannangpetch P and Kukongviriyapan V. Curcumin improves vascular function and alle-viates oxidative stress in non-lethal lipopoly-saccharide-induced endotoxaemia in mice. Eur J Pharmacol 2009; 616: 192-9.
[19] Memis D, Hekimoglu S, Sezer A, Altaner S, Sut N and Usta U. Curcumin attenuates the organ dysfunction caused by endotoxemia in the rat. Nutrition 2008; 24: 1133-8.
[20] Chen HW, Kuo HT, Chai CY, Ou JL and Yang RC. Pretreatment of curcumin attenuates coagu-lopathy and renal injury in LPS-induced endo -toxemia. J Endotoxin Res 2007; 13: 15-23. [21] Sun J, Yang D, Li S, Xu Z, Wang X and Bai C.
Effects of curcumin or dexamethasone on lung ischaemia-reperfusion injury in rats. Eur Respir J 2009; 33: 398-404.
[22] Osakabe N, Yasuda A, Natsume M, Sanbongi C, Kato Y, Osawa T and Yoshikawa T. Rosmarinic acid, a major polyphenolic component of Perilla frutescens, reduces lipopolysaccharide (LPS)-induced liver injury in D-galactosamine (D-GalN)-sensitized mice. Free Radic Biol Med 2002; 33: 798-806.
[23] Maier S, Traeger T, Entleutner M, Westerholt A, Kleist B, Huser N, Holzmann B, Stier A, Pfeffer K and Heidecke CD. Cecal ligation and punc-ture versus colon ascendens stent peritonitis: two distinct animal models for polymicrobial sepsis. Shock 2004; 21: 505-11.
[24] Schabbauer G. Polymicrobial sepsis models: CLP versus CASP. Drug Discov Today Dis Models 2012; 9: 17-21.
[25] Qi F, Liang ZX, She DY, Yan GT and Chen LA. A clinical study on the effects and mechanism of xuebijing injection in severe pneumonia pa-tients. J Tradit Chin Med 2011; 31: 46-9. [26] Sun J, Xue Q, Guo L, Cui L and Wang J. Xuebijing
protects against lipopolysaccharide-induced lung injury in rabbits. Exp Lung Res 2010; 36: 211-8.
[27] Wang Q, Yao YM, Wang WJ, Xian LM, Dong N, Xu S and Dou KF. Effect of Xuebijing injection on renal high mobility group box-1 protein ex-pression and acute kidney injury in rats after scald injury. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2007; 29: 478-83.
[28] Cao SH and Wang JD. Protective effects of Xuebijing on tissue and endothelial cells in rats with septic multiple organ dysfunction. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2002; 14: 489-91.
[29] Whang KT, Steinwald PM, White JC, Nylen ES, Snider RH, Simon GL, Goldberg RL and Becker KL. Serum calcitonin precursors in sepsis and systemic inflammation. J Clin Endocrinol Metab 1998; 83: 3296-301.
[30] Oberhoffer M, Karzai W, Meier-Hellmann A, Bogel D, Fassbinder J and Reinhart K. Sensitivity and specificity of various markers of inflammation for the prediction of tumor necro -sis factor-alpha and interleukin-6 in patients with sepsis. Crit Care Med 1999; 27: 1814-8. [31] Chen LY, Xu XJ and Fang CW. Comparision of
procalcitonin and C reactive protein in treating infection of newborn. Zhejiang Practical Me-dicine 2007; 12: 122-3.
[32] Murwani R, Hodgkinson S and Armati P. Tumor necrosis factor alpha and interleukin-6 mRNA expression in neonatal Lewis rat Schwann cells and a neonatal rat Schwann cell line fol-lowing interferon gamma stimulation. J Neuroimmunol 1996; 71: 65-71.
[33] Kapadia S, Lee J, Torre-Amione G, Birdsall HH, Ma TS and Mann DL. Tumor necrosis factor-al -pha gene and protein expression in adult feline myocardium after endotoxin administration. J Clin Invest 1995; 96: 1042-52.
[34] Pennica D, Kohr WJ, Fendly BM, Shire SJ, Raab HE, Borchardt PE, Lewis M and Goeddel DV. Characterization of a recombinant extracellu-lar domain of the type 1 tumor necrosis factor receptor: evidence for tumor necrosis factor-alpha induced receptor aggregation. Biochemi-stry 1992; 31: 1134-41.
[35] Chopra M, Reuben JS and Sharma AC. Acute lung injury: apoptosis and signaling mecha-nisms. Exp Biol Med (Maywood) 2009; 234: 361-71.
[36] Giebelen IA, van Westerloo DJ, LaRosa GJ, de Vos AF and van der Poll T. Local stimulation of alpha7 cholinergic receptors inhibits LPS-induced TNF-alpha release in the mouse lung. Shock 2007; 28: 700-3.
[37] Dinarello CA. Biologic basis for interleukin-1 in disease. Blood 1996; 87: 2095-147.
[39] Raingeaud J, Whitmarsh AJ, Barrett T, Derijard B and Davis RJ. MKK3- and MKK6-regulated gene expression is mediated by the p38 mito-gen-activated protein kinase signal transduc-tion pathway. Mol Cell Biol 1996; 16: 1247-55. [40] Chen YR, Meyer CF and Tan TH. Persistent acti -vation of c-Jun N-terminal kinase 1 (JNK1) in gamma radiation-induced apoptosis. J Biol Chem 1996; 271: 631-4.
[41] Das KC and Das CK. Curcumin (diferuloylmeth-ane), a singlet oxygen ((1)O(2)) quencher. Biochem Biophys Res Commun 2002; 295: 62-6.
[42] Mohammadi F, Bordbar AK, Divsalar A, Mohammadi K and Saboury AA. Analysis of binding interaction of curcumin and diacetyl-curcumin with human and bovine serum albu-min using fluorescence and circular dichroism spectroscopy. Protein J 2009; 28: 189-96. [43] Qin XY, Cheng Y, Cui J, Zhang Y and Yu LC.