Successful Treatment of Juvenile
Polyposis of Infancy With Sirolimus
Veronica B. Busoni, MD,aMarina Orsi, MD,aPablo A. Lobos, MD,aDaniel D’Agostino, MD,aMarta Wagener, MD,b Paola De la Iglesia, MD,aVictor L. Fox, MDcabstract
Juvenile polyposis syndrome is a rare autosomal dominant condition characterized by multiple hamartomatous polyps throughout the
gastrointestinal tract. Juvenile polyposis of infancy is a generalized severe form of juvenile polyposis syndrome associated with a poor prognosis. A 47-month-old female infant presented initially with gastrointestinal bleeding and protein-losing enteropathy at 4 months of age. At the age of 12 months, the condition worsened, requiring albumin infusions every 24 to 48 hours and red blood cell transfusions every 15 days. Upper gastrointestinal endoscopy, colonoscopy, and small-bowel enteroscopy revealed diffuse polyposis that was treated with multiple endoscopic polypectomies. Despite subtotal colectomy with ileorectal anastomosis, protein-losing enteropathy and bleeding persisted, requiring continued blood transfusions and albumin infusions. A chromosomal microarray revealed a single allele deletion in chromosome 10q23, involving both thePTENandBMPR1A genes. Loss of
PTENfunction is associated with an increased activation of the protein kinase B (AKT)/mammalian target of rapamycin (mTOR) pathway involved in cell proliferation. Treatment with sirolimus, an mTOR inhibitor, was initiated with the aim of inhibiting polyp growth. Soon after initiation of treatment with sirolimus, blood and albumin infusions were no longer needed and resulted in improved patient growth and quality of life. This case represents thefirst detailed report of successful drug therapy for life-threatening juvenile polyposis of infancy.
Juvenile polyposis syndrome (JPS) is a rare autosomal dominant condition characterized by the presence of multiple distinct juvenile polyps in the gastrointestinal tract. It has been clinically defined by the presence of
$5 juvenile polyps in the colon, juvenile polyps throughout the gastrointestinal tract, or any number of juvenile polyps and a family history of juvenile polyposis.1In 1975, Sachatello and Griffen2defined 3 types: juvenile polyposis coli, generalized juvenile polyposis, and juvenile polyposis of infancy (JPI). Patients with JPS are at an increased risk of gastric and colorectal cancer
(cumulative lifetime risk: 39%; relative risk: 34%).1,3,4
JPI is a generalized form of JPS that develops during thefirst months of life with polyps in stomach, small bowel, and colon. JPI is the most severe form of JPS and is associated with a poor prognosis. This syndrome is characterized by gastrointestinal bleeding, diarrhea, protein-losing enteropathy, anemia, rectal prolapse, and intestinal intussusception in infants and young children.5Severe protein-losing enteropathy can lead to growth retardation, emaciation, cachexia, and death. JPI has rarely been reported in the medical literature, and
aHospital Italiano de Buenos Aires, Buenos Aires, Argentina; bHospital de Niños Dr Alassia, Santa Fe, Argentina; and cBoston Children’s Hospital, Boston, Massachusetts
Dr Busoni conceptualized and designed the study, collected, analyzed, and interpreted data, and drafted the initial manuscript; Dr Fox conceptualized and designed the study and analyzed and interpreted data; Drs Orsi, Lobos, D’Agostino, De la Iglesia, and Wagener analyzed and interpreted data; and all authors revised and critically reviewed the manuscript for important intellectual content, approved thefinal version to be published as submitted, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
DOI:https://doi.org/10.1542/peds.2018-2922 Accepted for publication May 1, 2019
Address correspondence to Veronica B. Busoni, MD, Pediatric Gastroenterology, Hepatology and Liver Intestinal Transplantation Division, Department of Pediatrics, Hospital Italiano de Buenos Aires, Potosi 4135, Buenos Aires CP 1199, Argentina. E-mail: veronica.busoni@hospitalitaliano.org.ar
PEDIATRICS (ISSN Numbers: Print, 0031-4005; Online, 1098-4275).
Copyright © 2019 by the American Academy of Pediatrics
FINANCIAL DISCLOSURE:The authors have indicated they have nofinancial relationships relevant to this article to disclose.
FUNDING:No external funding.
POTENTIAL CONFLICT OF INTEREST:The authors have indicated they have no potential conflicts of interest to disclose.
the scarcity of this condition and its high rate of mortality have limited the study of its molecular basis in the past.6
Mutation of theBMPR1AandPTEN genes are responsible for JPS and Cowden syndrome, respectively. The development of polyposis is a typical feature of both disorders.
Microdeletions in chromosome 10q23 involving both contiguous genes are associated with aggressive polyposis in childhood.6PTEN mutations are associated with several
hamartomatous syndromes, including juvenile polyposis, Cowden
syndrome, and Bannayan-Riley-Ruvalcaba syndrome, collectively named thePTENhamartoma tumor syndrome.7Associated extraintestinal manifestations include congenital heart disease, developmental delay, and macrocephaly.8
Morbidity associated with polyposis in JPI is significant. Standard treatment includes repeated endoscopic surveillance and polypectomy, with frequent need of red blood cell (RBC) transfusions and albumin infusions. Advanced
nutritional support may be necessary in cases of severe growth impairment. In the most severe cases, colectomy may be necessary to reduce blood and protein losses.
CASE DESCRIPTION
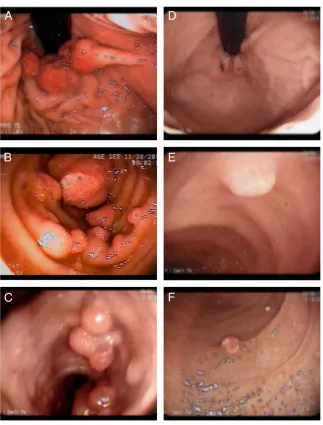
A term (38 weeks’gestation) girl delivered by cesarean delivery (weight 3300 g, length 49.5 cm, cephalic circumference 35.5 cm) presented at age 5 months with gastrointestinal bleeding and protein-losing enteropathy. Endoscopic examination, including upper gastrointestinal endoscopy, colonoscopy, and small-bowel enteroscopy, revealed diffuse polyposis (Fig 1 A–C). Repeated RBC transfusions and intravenous infusions of albumin were required despite multiple polypectomies performed during a series of
endoscopic procedures (Table 1). Histopathology revealed
characteristic features of juvenile polyps with no dysplasia in all resected polyps. Biopsies of grossly normal gastrointestinal mucosa showed normal histology (Fig 2). During laparotomy-assisted
enteroscopy performed at 8 months of age, a major laceration of the mesentery was produced by traction during manipulation of the small bowel. A 15-cm jejunal resection with end-to-end anastomosis was
performed with no later complications.
At the age of 12 months, the condition worsened, requiring albumin
infusions every 24 to 48 hours to maintain an average albumin level of 1.4 g/dL. RBC transfusions were required every 15 days.
At 17 months of age, a subtotal colectomy with ileorectal
anastomosis was performed during an emergent laparotomy for an ileocolonic intussusception. After this FIGURE 1
procedure, protein-losing enteropathy persisted and
requirements of blood transfusions and albumin infusion remained unchanged, presumably as a result of persisting protein exudation and bleeding from the gastric, small-bowel, and rectal polyps. Placement of a central venous catheter was required because of frequent blood sampling and albumin infusions.
The patient also had congenital abnormalities, including macrocephaly (head circumference.97th percentile) and complex congenital heart disease consisting of pulmonary stenosis and a ventricular septal defect.
A chromosomal microarray was performed, which revealed a 3078-Mb deletion in chromosome 10q23.2q23.31 involving both the
PTENgene andBMPR1Agene.
Given the difficulty to stabilize our patient and discharge her from the hospital, at 20 months of age, we
decided to initiate treatment with sirolimus, an inhibitor of mTOR and the molecular pathway known to be activated in patients withPTEN mutations. An informed consent with detailed information about sirolimus and its uses in organ transplant, vascular anomalies, andPTEN disorders; the goal of therapy in JPI; and monitoring, risks, side effects, alternatives, and potential benefits was signed by the parents and physician. Our goal was to slow gastrointestinal polyp proliferation or, at least, favor their involution. Before starting treatment with sirolimus, the patient had an average albumin level of 1.5 g/ dL, requiring albumin infusions every 24 to 48 hours. The clearance ofa-1 antitrypsin measuring protein exudation by fecal material was 176 mL/24 hours at initiation of treatment (normal value:
,12.5 mL/day).
The initial dose was 0.8 mg/m2per day taken twice daily. After 1 month
of treatment with sirolimus with a target serum level of 6 to 8 ng/mL, albumin infusions and RBC
transfusions were no longer required. Albumin levels remained steadily
.3.8 to 4 g/dL and hemoglobin levels over 12 g/dL.a-1 antitrypsin clearance also improved to near-normal levels at 13.3 mL/day (Fig 3). Endoscopic evaluations showed a definite change in polyp growth, with scarce distribution and smaller size (Fig 1 D–F). At 47 months of age, after 27 months of treatment with sirolimus, the patient remains stable with no transfusion requirements.
Congenital heart disease was treated through cardiac catheterization after gastrointestinal disease was under control at 22 months of age. Valvuloplasty was performed for pulmonary stenosis, with a residual gradient of 30 mm Hg. The
interventricular communication was closed with a device with minimum residual shunt.
FIGURE 2
Histology: A, Colonic juvenile polyp with dilated hyperplastic colonic glands, inflammatory infiltrate, and surface erosion. B, Gastric juvenile polyp composed of hyperplastic foveolar epithelium with cyst formation and edematous stroma. C, Colonic mucosa with preserved histoarchitecture.
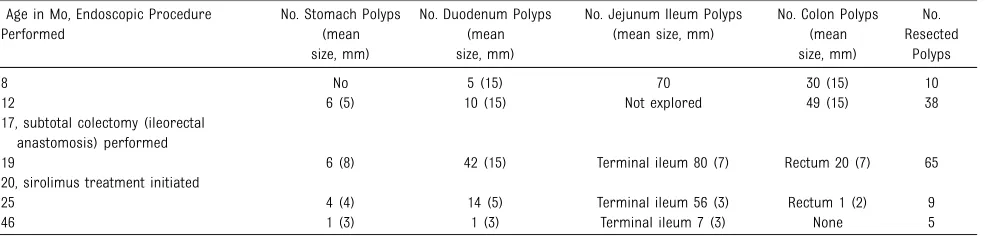
TABLE 1Endoscopic Procedures Before and After Sirolimus
Age in Mo, Endoscopic Procedure Performed
No. Stomach Polyps (mean size, mm)
No. Duodenum Polyps (mean size, mm)
No. Jejunum Ileum Polyps (mean size, mm)
No. Colon Polyps (mean size, mm)
No. Resected
Polyps
8 No 5 (15) 70 30 (15) 10
12 6 (5) 10 (15) Not explored 49 (15) 38
17, subtotal colectomy (ileorectal anastomosis) performed
19 6 (8) 42 (15) Terminal ileum 80 (7) Rectum 20 (7) 65 20, sirolimus treatment initiated
25 4 (4) 14 (5) Terminal ileum 56 (3) Rectum 1 (2) 9
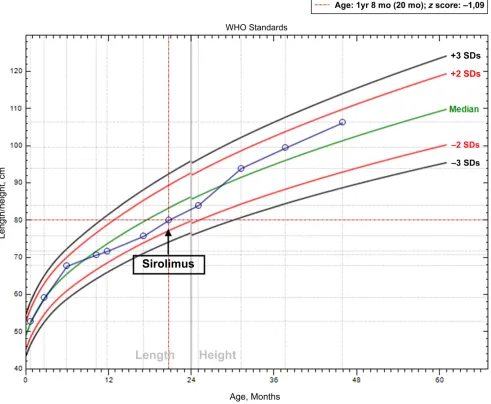
Since initiation of treatment with sirolimus, the magnitude of the enteropathy was reduced, and the patient could be discharged from the hospital without the need for central venous access 1 month post initiation of treatment. Edema resolved, and no albumin infusion or RBC transfusion was needed in the last 27-month follow-up interval. Post-treatment growth improvement was also evident (Fig 4). The patient and family’s quality of life improved by allowing the child to return home (300 miles from the hospital).
DISCUSSION
We believe that this is thefirst detailed report of successful drug therapy of this devastating and life-threatening condition. The child’s pretreatment and improved post-treatment clinicalfindings (laboratory values, growth
parameters, and endoscopicfindings) demonstrate a dramatic change in the natural evolution of this disease. The genetic defect underlying our patient’s condition is well defined by a deletion in chromosome 10q23 that includes thePTENgene. The potential mechanism by which sirolimus works can be explained by its known inhibitory effect on the
phosphatidylinositol 3-kinase/AKT/ mTOR pathway involved in cell proliferation and regulated byPTEN.
As reported by Oliveira et al,9patients with JPI are offered subtotal
colectomy combined with
intraoperative endoscopic removal of polyps as a palliative disease control. It is remarkable that despite multiple polyp resection or even subtotal colectomy, we were unable to obtain a decrease in the magnitude of protein loss. Sirolimus treatment improved severe protein-losing enteropathy in our patient and reduced the need for frequent infusions of albumin, with an evident improvement in her quality of life.
Sirolimus (originally named rapamycin) is an approved drug for suppression of the immune system after organ transplant and evidence suggests that use of mTOR inhibitors in children undergoing solid organ transplant is efficacious and safe.10Its effect is produced by the inhibition of a protein called mTOR, a Ser/Thr protein kinase, which regulates cell growth and proliferation and is involved in the regulation of the immune system and the development of blood vessels and tissue growth. Sirolimus (and other mTOR inhibitors) are currently being used for the treatment of complex vascular
malformations11and different types of cancer, hoping that it will act on the abnormal development of blood vessels (angiogenesis). As an inhibitor of the mTOR pathway, an important regulator of cell proliferation and survival, sirolimus and its analogs have an expanding role in the treatment of tumors associated with tuberous sclerosis complex,12,13 lymphoproliferative disease, and autoimmune cytopenias.14,15
The etiologic basis for the subset of juvenile polyposis termed JPI is a contiguousPTENandBMPR1Agene deletion.16PTENis a tumor
suppressor gene whose function is frequently lost through genetic and epigenetic mechanisms. Loss ofPTEN increases the activation of the AKT/ mTOR pathway, which increases cell proliferation and survival and is associated with subsequent development of various tumors.
There is some preliminary evidence on the effect of sirolimus on cell
proliferation in conditions with generalized polyposis. Hardiman et al17reported experimental evidence of an inhibitory effect of rapamycin in a transgenic adenomatous polyposis coli mutation–dependent colon polyposis mouse model. The effect on polyps included a decrease in proliferation, an increase in
differentiation, and prolonged time to development of dysplasia, resulting in increased animal survival.
Authors of several case reports have suggested a potential role in the treatment of patients withPTEN hamartoma tumor syndrome,18–21 and there is emerging experimental and clinical evidence for role in the treatment of adenomatous
polyposis17,22and Peutz-Jeghers syndrome.23–25
Although we are encouraged by the dramatic beneficial effect of sirolimus in our patient over a period of time exceeding 2 years, we remain cautious about predicting the long-term effects. The development of drug resistance FIGURE 3
remains a possibility, although this is not well described when used to treat other benign conditions. We also acknowledge that we are reporting a clinical response to sirolimus without direct evidence of its molecular effect on cells or tissue in our patient. Such evidence is beyond the scope of this report.
CONCLUSIONS
We report successful treatment of a child suffering from complications of severe infant juvenile polyposis using sirolimus. This treatment should be considered for other children who are
similarly affected by this condition. Given the rareness of the disease, multicenter studies should gather experiences with multiple patients to explore the safety and long-term effectiveness of this drug therapy. This report contributes to the growing knowledge and experience of using inhibitors of the mTOR pathway for various conditions associated with tumor formation and cellular proliferation.
ACKNOWLEDGMENTS
We acknowledge a recent brief report in the form of a letter to the
editor that was published after our manuscript was submitted for review. The authors reported a 5-year sustained positive response to sirolimus in a child with JPI.26
ABBREVIATIONS AKT: protein kinase B
JPI: juvenile polyposis of infancy JPS: juvenile polyposis syndrome mTOR: mammalian target of
rapamycin RBC: red blood cell FIGURE 4
REFERENCES
1. Brosens LA, Langeveld D, van Hattem WA, Giardiello FM, Offerhaus GJ. Juvenile polyposis syndrome.World J Gastroenterol. 2011;17(44):4839–4844
2. Sachatello CR, Griffen WO Jr. Hereditary polypoid diseases of the
gastrointestinal tract: a working classification.Am J Surg. 1975;129(2): 198–203
3. Jass JR, Williams CB, Bussey HJ, Morson BC. Juvenile polyposis–a precancerous condition.
Histopathology. 1988;13(6):619–630
4. Giardiello FM, Hamilton SR, Kern SE, et al. Colorectal neoplasia in juvenile polyposis or juvenile polyps.Arch Dis Child. 1991;66(8):971–975
5. Sachatello CR, Hahn IS, Carrington CB. Juvenile gastrointestinal polyposis in a female infant: report of a case and review of the literature of a recently recognized syndrome.Surgery. 1974; 75(1):107–114
6. Dahdaleh FS, Carr JC, Calva D, Howe JR. Juvenile polyposis and other intestinal polyposis syndromes with
microdeletions of chromosome 10q22-23.Clin Genet. 2012;81(2):110–116
7. Marsh DJ, Kum JB, Lunetta KL, et al. PTEN mutation spectrum and genotype-phenotype correlations in Bannayan-Riley-Ruvalcaba syndrome suggest a single entity with Cowden syndrome.
Hum Mol Genet. 1999;8(8):1461–1472
8. Septer S, Zhang L, Lawson CE, Cocjin J, Attard T, Ardinger HH. Aggressive juvenile polyposis in children with chromosome 10q23 deletion.World J Gastroenterol. 2013;19(14):2286–2292
9. Oliveira PH, Cunha C, Almeida S, et al. Juvenile polyposis of infancy in a child with deletion of BMPR1A and PTEN
genes: surgical approach.J Pediatr Surg. 2013;48(1):e33–e37
10. Ganschow R, Pape L, Sturm E, et al. Growing experience with mTOR inhibitors in pediatric solid organ transplantation.Pediatr Transplant. 2013;17(7):694–706
11. Adams DM, Trenor CC III, Hammill AM, et al. Efficacy and safety of sirolimus in the treatment of complicated vascular anomalies.Pediatrics. 2016;137(2): e20153257
12. Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus long-term use in patients with tuberous sclerosis complex: four-year update of the EXIST-2 study.PLoS One. 2017;12(8):e0180939
13. Jeong A, Wong M. mTOR inhibitors in children: current indications and future directions in neurology.Curr Neurol Neurosci Rep. 2016;16(12):102
14. Bride KL, Vincent T, Smith-Whitley K, et al. Sirolimus is effective in relapsed/ refractory autoimmune cytopenias: results of a prospective multi-institutional trial.Blood. 2016;127(1): 17–28
15. Bride K, Teachey D. Autoimmune lymphoproliferative syndrome: more than a FAScinating disease.F1000 Res. 2017;6:1928
16. Delnatte C, Sanlaville D, Mougenot JF, et al. Contiguous gene deletion within chromosome arm 10q is associated with juvenile polyposis of infancy, reflecting cooperation between the BMPR1A and PTEN tumor-suppressor genes.Am J Hum Genet. 2006;78(6): 1066–1074
17. Hardiman KM, Liu J, Feng Y, Greenson JK, Fearon ER. Rapamycin inhibition of polyposis and progression to dysplasia in a mouse model.PLoS One. 2014;9(4): e96023
18. Marsh DJ, Trahair TN, Martin JL, et al. Rapamycin treatment for a child with germline PTEN mutation.Nat Clin Pract Oncol. 2008;5(6):357–361
19. Iacobas I, Burrows PE, Adams DM, Sutton VR, Hollier LH, Chintagumpala MM. Oral rapamycin in the treatment of patients with hamartoma syndromes and PTEN mutation.Pediatr Blood Cancer. 2011;57(2):321–323
20. Schmid GL, Kässner F, Uhlig HH, et al. Sirolimus treatment of severe PTEN hamartoma tumor syndrome: case report and in vitro studies.Pediatr Res. 2014;75(4):527–534
21. Zak M, Ledbetter M, Maertens P. Infantile Lhermitte-Duclos disease treated successfully with rapamycin.
J Child Neurol. 2017;32(3):322–326
22. Yuksekkaya H, Yucel A, Gumus M, Esen H, Toy H. Familial adenomatous polyposis; succesful use of sirolimus.
Am J Gastroenterol. 2016;111(7): 1040–1041
23. Wei C, Amos CI, Zhang N, et al. Suppression of Peutz-Jeghers polyposis by targeting mammalian target of rapamycin signaling.Clin Cancer Res. 2008;14(4):1167–1171
24. Klümpen HJ, Queiroz KC, Spek CA, et al. mTOR inhibitor treatment of pancreatic cancer in a patient with Peutz-Jeghers syndrome.J Clin Oncol. 2011;29(6): e150–e153
25. Kuwada SK, Burt R. A rationale for mTOR inhibitors as chemoprevention agents in Peutz-Jeghers syndrome.Fam Cancer. 2011;10(3):469–472
DOI: 10.1542/peds.2018-2922 originally published online July 31, 2019;
2019;144;
Pediatrics
Wagener, Paola De la Iglesia and Victor L. Fox
Veronica B. Busoni, Marina Orsi, Pablo A. Lobos, Daniel D'Agostino, Marta
Successful Treatment of Juvenile Polyposis of Infancy With Sirolimus
Services
Updated Information &
http://pediatrics.aappublications.org/content/144/2/e20182922 including high resolution figures, can be found at:
References
http://pediatrics.aappublications.org/content/144/2/e20182922#BIBL This article cites 26 articles, 5 of which you can access for free at:
Subspecialty Collections
http://www.aappublications.org/cgi/collection/gastroenterology_sub Gastroenterology
following collection(s):
This article, along with others on similar topics, appears in the
Permissions & Licensing
http://www.aappublications.org/site/misc/Permissions.xhtml in its entirety can be found online at:
Information about reproducing this article in parts (figures, tables) or
Reprints
DOI: 10.1542/peds.2018-2922 originally published online July 31, 2019;
2019;144;
Pediatrics
Wagener, Paola De la Iglesia and Victor L. Fox
Veronica B. Busoni, Marina Orsi, Pablo A. Lobos, Daniel D'Agostino, Marta
Successful Treatment of Juvenile Polyposis of Infancy With Sirolimus
http://pediatrics.aappublications.org/content/144/2/e20182922
located on the World Wide Web at:
The online version of this article, along with updated information and services, is
by the American Academy of Pediatrics. All rights reserved. Print ISSN: 1073-0397.