7119
Characterization Of Physical And Chemical
Properties Of Mahua Oil Biodiesel For
Improvement In Its Cold Flow Properties
Navdeep Sharma Dugala, Gyanendra Singh Goindi, Keshav Raj
Abstract: In the current scenario, the energy sector of the developing world depends on fossil fuels. To reduce fossil fuel dependence, it is essential to develop new alternative energy source which can replace fossil fuels. Biodiesel is an effective approach that can replace conventional fossil fuels. Mahua biodiesel is produced from Raw Mahua oil by simple transesterification method. This experiment shows biodiesel production from raw Mahua oil with methanol instead of alcohol and KOH as a catalyst. Fuel properties of Mahua biodiesel are compiled in accordance with European (EN) and American (ASTM) standardization for testing of biodiesel and cold flow properties are further improved by blending Mahua biodiesel with ethanol. B20 blends of ethanol and Mahua biodiesel shows good pour properties.
Index Terms: Cloud Point, Ethanol, Mahua Biodiesel, Methanol, Physicochemical Properties, Pour Point, Transesterification. —————————— ——————————
1
INTRODUCTION
Biodiesel is receiving considerable attention in the world as an alternative fuel to fossil based fuels. It also provides a low-cost fuel to energy-dependent countries and reduces the global warming problems associated with fossil fuels. Biodiesel is produced from natural and renewable form of energy re-sources like edible, non-edible vegetable oils or animal fats [1], [2]. Biodiesel is defined as the mono alkyl esters of fatty acid chain of edible oils, non-edible oils, animal fats or waste oils. The transesterification process is the most suitable method to produce biodiesel. In the transesterification method, tri-glycerides are made to react with methanol, where a catalyst (acid or base) is used to get methyl esters as a product and glycerol as a by-product [3]–[6]. Mahua Biodiesel is also produced by the transesterification process from the Mahua seed oil, in which Mahua seed oil is reacted with methanol with a base catalyst at a specific temperature and time. To reduce the viscosity, heating, thermal cracking, pyrolysis, dilution and transesterification can be employed [7]. Mahua trees can produce Mahua seeds to the tune of 60 million tons annually. Mahua seeds oil has a yield of approximately 35% to 40% [8]. Mahua Oil has high acidic value so it needs a pretreatment process before its transesterification process. The yield of output depends upon the alcohol content, reaction temperature and time taken in reaction. The alcohol used in this process is methanol because it is more reactive than ethanol. The base catalyst used for biodiesel production is KOH (Potassium Hydroxide) since it is also more reactive than the NaOH (Sodium Hydroxide). Mahua Biodiesel has better emission characteristics than diesel according to other research finding but due to its high cloud and pour point temperatures, its cold flow characteristics are poor.
To enhance the low temprature properties of mahua biodiesel, we need to improve its cloud and pour point. This could be achieved by oil blending with diesel, kerosene, ethanol, etc. But diesel and kerosene fuel adversely affect emission characteristics so they cant be used. That leaves us to Ethanol which has a good effect on emission as well as cold flow characteristics according to another research finding. Peterson et al. [9] performed a comparison of four methyl and ethyl esters of biodiesel feedstocks. This comparison was mainly based on the engine performance & fuel characteristics tests. They found that the cloud point temperature of tallow biofuel is approximately 16℃ as compared to No. 2 diesel which has a cloud point of -12℃. Lee et al. [10] discovered that the cloud point of a normal soybean methyl ester can be decreased to -7.1℃ via the winterization method. Serrano et al. [11] experimented the low temperature flow properties of several vegetable oils biodiesel and discovered that cloud filter plugging point was -14℃ for rapeseed, -4℃ for soybean, -6℃ for high oleic soybean and 13℃ for palm oil methyl ester. Davis et al. [12] used urea and methanol for soybean methyl ester fractionation to produce improved biodiesel with a low cloud point of -45℃. In any case, most of the high cloud point biodiesel fraction is removed. Giraldo et al. [3] analyzed a variety of additives in palm biodiesel in the range from 1%– 10% to study cold flow properties and noted that better results could be achieved by addition of 5% 2-butyl. Cao et al. [13] identified that C.P., P.P., and C.F.P.P. of the B20 blend decreased by 8℃, 10℃ and 11℃, after the EVAC (ethylene vinyl acetate copolymer) treatment by 0.04 wt%. Rasimoglu and Temur [14] discovered that if oil to alcohol ratio was kept between 1:3.15 and 1:4.15, better cold flow properties could be achieved for biodiesel and they found best C.P., C.F.P.P., and P.P. of biodiesel produced from corn oil was found to be -4℃, -12℃, and -10℃. Boshui et al. [15] used a variety of additives in soybean biofuel to enhance its low temperature flow properties and observed its effects. By adding 0.03% of olefin-ester copolymers (OECP) in biodiesel the PP and CFPP of biodiesel reduced significantly. The pour point was found to be -9℃ and the cloud filter plugging point was -6℃. Lv et al. [16] noted pour point, cloud point, and cloud filter plugging point of palm oil methyl ester to be 18℃, 16℃ and 13℃. By using DEP: PGE: PA additive in 3:1:1 or 2:2:1 ratio, the cloud filter plugging point of palm oil methyl ester was reduced by ________________________________
• Navdeep Sharma Dugala is currently pursuing PhD degree program in mechanical engineering in Chandigarh University, Punjab, India E-mail: navdeepdugala@gmail.com
• Dr. Gyanendra Singh Goindi is a professor in Department of Mechanical Engineering in Chandigarh University, Punjab, India. E-mail: gyanendra.e4527@cumail.in
7120 7℃. Phung et al. [17] conducted an experiment
by triglycerides autooxidation and reported that the cloud point of canola and soybean autoxidized biodiesel was -13℃. Makareviciene et al. [18] analyzed the result of the blending of butanol with rapeseed oil butyl & methyl esters. Ali et al. [19] recommended that for palm biodiesel, the B30 blend is optimal for fuel properties as per requirements. They reduced the pour point of palm oil methyl ester from 14℃ to less than 0℃. Joshi et al. [20] considered Ethyl Levulinate for improving cottonseed oil cold flow properties. Cloud point reduced by 4℃ to 5℃, while pour point and cloud filter plugging point had 3℃ to 4℃ and 3℃ decrease respectively at 20 volume% ethyl levulinate. Nainwal et al. [21] reported that using kerosene (up to 20 vol%) in biodiesel can improve the low temperature flow properties. In this experimentation, we prepared two samples of mahua biodiesel having different parameters to them like alcohol to oil ratio, reaction timing, catalyst quality, reaction temperature, etc. Two more samples of mahua biodiesel were prepared in so as to improve its low temprature flow properties. Each has a different proportion of Ethanol added to it.
2
MATERIAL
AND
METHODS
Raw Mahua oil was purchased from Himani International, Delhi. Methanol (99%) pure was purchased from Oswal Scientific Stores (Chandigarh). The base catalyst used for the Transesterification process was Potassium Hydroxide (KOH) in pallet form and Sulphuric Acid (H2SO4) as an acid catalyst in the pretreatment process. The experimentation was done on a laboratory scale apparatus, which contained 1-liter glass flask, stirring rod and a water bath whose maximum temperature was 1000C. The flask was kept inside the water bath and solution was manually stirred after five minutes interval.
2.1 Preheating
The raw mahua oil during pre-heating process, was heated up to 100℃ temperature in a water bath for 30 min and stirred manually after 5 min of interval. This was done to remove water contained from seed oil because water’s boiling point is 100℃, and to remove unwanted dirt by filtering it.
2.2 Pretreatment
The pretreatment of oil was done so as to minimize the FFA content from 12% to approximately less than 1%. In simple words; the pretreatment process was done to decrease the acidic amount form raw mahua seed oil. Two sampled were made, each containing a different proportion of methanol to oil. Sample 1 contained preheated Mahua Oil, 50% vol./vol. methanol to oil and 1% vol./vol. Sulphuric Acid (H2SO4) as a base catalyst. The reaction was carried for two hours in a glass beaker inside a water bath whose temperature was kept from 60℃ to 80℃ and the solution was stirred manually after 5 min interval. The mixture was then allowed to slowly settle down for one hour inside a separating funnel. The pretreatment reaction is as given below:
R1 COOH + ROH = R1 COOR + H2O
Sample 2 contained preheated Mahua Oil, 75% vol. /vol. methanol to oil and 1.5% vol. /vol. Sulphuric Acid (H2SO4) as a base catalyst. The reaction was carried for two hours in a glass beaker inside a water bath whose temperature was kept from 65℃ to 90℃ and the solution was stirred manually after 5
min interval. The mixture was again allowed to slowly settle down for one hour inside a separating funnel.
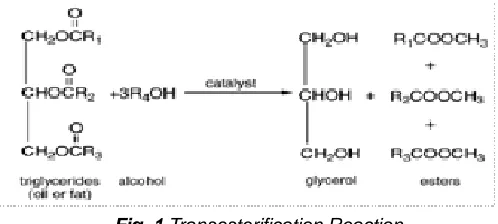
2.3 Transesterification
The transesterification process was done to remove the glycerol from seed oil in order to get methyl ester solution from it. The transesterification reaction is given in Fig.1.
Fig. 1 Transesterification Reaction
In this process, pretreated seed oil was used and heated to 60℃ temperature inside a water bath in a glass beaker.
For sample 1; Methoxide solution was created which had 40% vol. /vol. Methanol and 1% wt./vol. Potassium Hydroxide (KOH) in pallet form. Contents were mixed in a separate flask with cork on it and the flask for shaken for 10 min. Methoxide solution was added to pretreated mahua oil which was kept at 60℃ inside water bath and carried out this reaction for 30 min, and water bath temperature was varied from 60℃ to 70℃, manually stirred the solution after 5 min of interval. Allowed settling of solution for an overnight inside a separating funnel.
For sample 2; Methoxide solution was created which had 50% vol. /vol. Methanol and 1% wt./vol. Potassium Hydroxide (KOH) in pallet form. Mixed them in a separate flask with cork on it and shook this flask for at least 10 min. Methoxide solution was added to pretreated mahua oil which was kept at 60℃ inside water bath and carried out this reaction for 60 min, water bath temperature was varied from 60℃ to 70℃, manually stirred the solution after 5 min of interval. Allowed settling of solution for an overnight inside a separating funnel.
2.4 Separation
After overnight settling of the solution inside separating funnel, the solution made two different layers: the layer underneath was glycerin and on the top it was methyl ester. We removed the lower layer of glycerol carefully by opening the bottom end of separating funnel. Before doing this, we made sure the glass cork on the separating funnel was removed and finally we removed the upper layer of Methyl Ester of Mahua and placed it in a separate beaker or bottle.
2.5 Water Washing
7121 meant the upper layer of methyl ester was free from glycerin.
Now we heated the final solution of methyl ester 100℃ in order to remove any water contained present inside the methyl ester solution.
3 IMPROVING
COLD
FLOW
CHARACTERISTICS
Most of Biodiesel properties are similar to fossil fuel-based mineral diesel but improving the biodiesel cold temperature flowing characteristic is the major challenge in using it as a fuel for automobiles as in C.I. engines. Biodiesel fuel is produced from vegetable oils, animal fats, etc. which have a high amount of saturated fat content which shows high pour point and cloud point. The pour point occurs at a low temperature than the cloud point. It is a temperature at which oil takes a semi-solid shape and began to lose its flowability. And cloud point is a temperature at which biodiesel form a cloudy appearance.
3.1 Method
The experiments were conducted to obtain the cold temperature flow characteristics for biodiesel. These were tested for the various sample methyl-ester blends of mahua oil with ethanol (10% and 20% sample) i.e. MB90E10 (MME 90% and Ethanol 10%) and MB80E20 (MME 80% and Ethanol 20%).
3.2 Cloud and Pour Point Setup
The various sample blends of biodiesel were poured into a glass tube and kept inside the cloud point and pout point tester. We then placed the cork with a thermometer on it. We filled the surrounding of a glass tube with cold water or ice in order to achieve a desirable effect on biodiesel cold flow properties. We recorded the temperature where biodiesel started to form or give cloudy appearance (i.e. Cloud point) and temperature at which it formed a semi solid-state (i.e. Pour Point).
4 PROPERTY
TABLE
The Physico chemical properties of raw Mahua seed oil, Mahua biodiesel sample 1 and 2 are given in Table I along with values recommended by ASTM (6751-02) standards.
Table 1 Physico chemical properties of raw Mahua seed oil, Mahua biodiesel sample 1 and 2
Sr. No . Property Raw Mahu a Oil M.B. D Sampl e 1 M.B.D Sampl e 2 Miner al Diesel ASTM (6751-02) recommendat ions
1 Density
(kg/m3) 950 882.7 875.3 830 850-879
2 Specific
Gravity 0.95 0.883 0.875 .830 0.85-0.94
3
Kinemati c Viscosit y (cSt)
24.99 3.99 3.57 2.60 1.9-6.0
4
Flash Point (℃)
235 195 180 50 Min130
5 Fire Point (℃)
245 205 190 60 Min135
6
Cloud Point (℃)
12 8 6 -3 5-12
7
Pour Point (℃)
8 3 2 -8 3-5
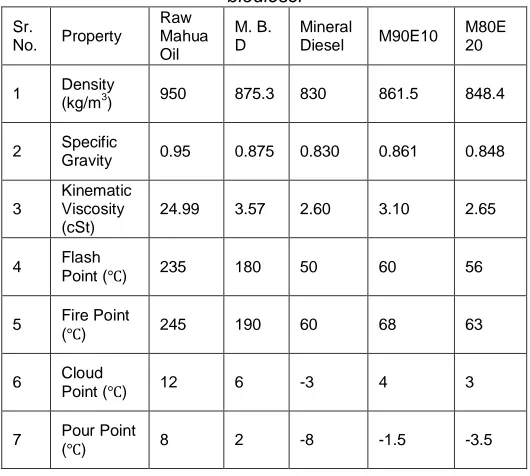
The Physico chemical properties of M90E10 and M80E20 biodiesel are given in Table 2 along with recommendations of ASTM (6751-02) standards.
Table 2 Physico chemical properties of M90E10 and M80E20 biodiesel
Sr.
No. Property
Raw Mahua Oil M. B. D Mineral
Diesel M90E10 M80E 20
1 Density
(kg/m3) 950 875.3 830 861.5 848.4
2 Specific
Gravity 0.95 0.875 0.830 0.861 0.848
3
Kinematic Viscosity (cSt)
24.99 3.57 2.60 3.10 2.65
4 Flash
Point (℃) 235 180 50 60 56
5 Fire Point
(℃) 245 190 60 68 63
6 Cloud
Point (℃) 12 6 -3 4 3
7 Pour Point
(℃) 8 2 -8 -1.5 -3.5
5 RESULT
AND
DISCUSSION
5.1 Density
Density is calculated at room temperature (20℃) and the density of Raw mahua oil is more than other fuel. Figure 2 shows the comparison of density between all biodiesel samples. In the figure, density of raw Mahua oil is maximum and minimum for diesel.
7122
5.2 Kinematic Viscosity
Kinematic viscosity is calculated at 40℃ temperature by using Redwood Viscometer. The comparison of kinematic viscosity between all biodiesel samples is shown in figure 3. Kinematic Viscosity of raw Mahua oil is very high compare to others.
Fig. 3 Comparison of kinematic viscosity between all biodiesel samples
5.3 Flash Point
Flash point is calculated with the help of the Pensky Martins Close Cup and figure 4 shows that the flash point of Raw Mahua Oil is more than other samples. Mahua Biodiesel also has flash point more than Diesel.
Fig. 4 Comparison of flash point between all biodiesel samples
5.4 Cloud Point
Cloud point is calculated in a Cloud Point and Pour Point Tester. And it is found that Mahua Biodiesel sample 2 has lower cloud Point than other samples of Biodiesel but not less than Diesel.
Fig. 5 Comparison of cloud point between all biodiesel samples
5.5 Pour Point
Fig. 6 Comparison of pour point between all biodiesel samples
Pour point is calculated in a Cloud Point and Pour Point Tester. And it is found that Mahua Biodiesel sample 2 has lower cloud Point than other samples of Biodiesel but not less than Diesel.
6 COLD
FLOW
PROPERTIES
RESULT
6.1 Cloud Point
7123 Fig. 7 Comparison of cloud point properties
6.2 Pour Point
It shows that the Pour Point of MB80E20 (Mahua Biodiesel 80% and Ethanol 20% ) is lower than other samples of biodiesel but higher than diesel. It means that the MB80E20 sample has better cold flowing characteristics than other biodiesel samples but better than Diesel.
Fig. 8 Comparison of pour point properties
7 CONCLUSIONS
In this experimentation, the various results were founded and some of them were as following:
Pretreatment was used to reduce the FFA value.
The transesterification process was used to separate biodiesel from glycerin.
MBD samples 2 have better properties than other biodiesel samples.
All the properties of Biodiesel were under the limits of ASTM standards.
To enhance the cold flow properties of biodiesel, ethanol was added to it.
MB80E20 had better Pour and Cloud Point than other samples of biodiesel.
This work can be further extended toward testing on C.I. Engine and we can find out the effect of these biodiesel samples on the engine’s performance and emission
characteristics.
REFERENCES
[1]. P. Verma and M. P. Sharma, ―Performance and emission characteristics of biodiesel fuelled diesel engines,‖ Int. J. Renew. Energy Res., vol. 5, no. 1, pp. 245–250, 2015.
[2]. G. Dwivedi and M. P. Sharma, ―Potential and limitation of straight vegetable oils as engine fuel - An Indian perspective,‖ Renew. Sustain. Energy Rev., vol. 33, pp. 316–322, 2014.
[3]. S. Y. Giraldo, L. A. Rios, and N. Suárez, ―Comparison of glycerol ketals, glycerol acetates and branched alcohol-derived fatty esters as cold-flow improvers for palm biodiesel,‖ Fuel, vol. 108, pp. 709–714, 2013. [4]. P. Verma, M. P. Sharma, and G. Dwivedi, ―Operational
and environmental impact of biodiesel on engine performance: A review of literature,‖ Int. J. Renew. Energy Res., vol. 5, no. 4, pp. 961–970, 2015.
[5]. J. Wang, L. Cao, and S. Han, ―Effect of polymeric cold flow improvers on flow properties of biodiesel from waste cooking oil,‖ Fuel, vol. 117, no. PART A, pp. 876–881, 2014.
[6]. M. A. H. Altaie, R. B. Janius, U. Rashid, Y. H. Taufiq Yap, R. Yunus, and R. Zakaria, ―Cold flow and fuel properties of methyl oleate and palm-oil methyl ester blends,‖ Fuel, vol. 160, pp. 238–244, 2015.
[7]. S. V. Ghadge and H. Raheman, ―Biodiesel production from mahua (Madhuca indica) oil having high free fatty acids,‖ Biomass and Bioenergy, vol. 28, no. 6, pp. 601–605, 2005.
[8]. L. Yang, M. Takase, M. Zhang, T. Zhao, and X. Wu, ―Potential non-edible oil feedstock for biodiesel production in Africa: A survey,‖ Renew. Sustain. Energy Rev., vol. 38, pp. 461–477, 2014.
[9]. C. L. Peterson, D. L. Reece, B. L. Hammond, J. Thompson, and S. M. Beck, ―Processing, Characterization, and Performance of Eight Fuels From Lipids,‖ Appl. Eng. Agric., vol. 13, no. 1, pp. 71– 79, 1997.
[10]. I. Lee, L. A. Johnson, and E. G. Hammond, ―Reducing the crystallization temperature of biodiesel by winterizing methyl soyate,‖ JAOCS, J. Am. Oil Chem. Soc., vol. 73, no. 5, pp. 631–636, 1996.
[11]. M. Serrano, R. Oliveros, M. Sánchez, A. Moraschini, M. Martínez, and J. Aracil, ―Influence of blending vegetable oil methyl esters on biodiesel fuel properties: Oxidative stability and cold flow properties,‖ Energy, vol. 65, pp. 109–115, 2014. [12]. R. Davis, S. Mohtar, and B. Tao, ―Production of
low-temp biodiesel through urea clathration,‖ 2007 ASABE Annu. Int. Meet. Tech. Pap., vol. 13 BOOK, 2007. [13]. L. Cao, J. Wang, C. Liu, Y. Chen, K. Liu, and S. Han,
―Ethylene vinyl acetate copolymer: A bio-based cold flow improver for waste cooking oil derived biodiesel blends,‖ Appl. Energy, vol. 132, pp. 163–167, 2014. [14]. N. Rasimoglu and H. Temur, ―Cold flow properties of
biodiesel obtained from corn oil,‖ Energy, vol. 68, pp. 57–60, 2014.
7124 [16]. P. Lv, Y. Cheng, L. Yang, Z. Yuan, H. Li, and W. Luo,
―Improving the low temperature flow properties of palm oil biodiesel: Addition of cold flow improver,‖ Fuel Process. Technol., vol. 110, pp. 61–64, 2013.
[17]. P. Phung, W. N. Rowlands, A. Thiyakesan, P. Benndorf, A. F. Masters, and T. Maschmeyer, ―Metal/bromide autoxidation of triglycerides for the preparation of FAMES to improve the cold-flow characteristics of biodiesel,‖ Catal. Today, vol. 233, pp. 162–168, 2014.
[18]. V. Makarevičiene, K. Kazancev, and I. Kazanceva, ―Possibilities for improving the cold flow properties of biodiesel fuel by blending with butanol,‖ Renew. Energy, vol. 75, pp. 805–807, 2015.
[19]. O. M. Ali, R. Mamat, N. R. Abdullah, and A. A. Abdullah, ―Analysis of blended fuel properties and engine performance with palm biodiesel-diesel blended fuel,‖ Renew. Energy, vol. 86, pp. 59–67, 2015.
[20]. H. Joshi, B. R. Moser, J. Toler, W. F. Smith, and T. Walker, ―Ethyl levulinate: A potential bio-based diluent for biodiesel which improves cold flow properties,‖ Biomass and Bioenergy, vol. 35, no. 7, pp. 3262– 3266, 2011.