Contents lists available at ScienceDirect
Journal of the Formosan Medical Association
Journal homepage: http://www.jfma-online.comJournal of the
Formosan Medical Association
ISSN 0929 6646
Formosan Medical Association Taipei, Taiwan
Volume 110 Number 8 August 2011
Resveratrol for prophylaxis of ischemic stroke Microglia and chronic pain ART in HIV-1-discordant couples in Taiwan Prognostic evaluation of patients with multicentric papillary thyroid microcarcinoma
Original Article
Prognostic Evaluation of Patients With Multicentric
Papillary Thyroid Microcarcinoma
Sheng-Fong Kuo,1Tzu-Chieh Chao,2Hung-Yu Chang,3Chuen Hsueh,4Chung-Han Yang,5
Jen-Der Lin3*
Background/Purpose:Patients with papillary thyroid microcarcinoma (PTMC) often have an excellent
prognosis. We hypothesize that patients with multicentric PTMC are associated with good clinical out-come, although multicentricity in papillary thyroid carcinoma may be associated with poor prognosis.
Methods:Retrospective analysis of multicentric PTMC cases in one medical center enrolled from 1987 to
2008 was conducted. At the end of follow-up, patients were classified as “recurrence-free” or “recurrence or persistent disease”. The tumor–node–metastasis (TNM) staging system was used, and (T1, N0, M0) was regarded as “early clinical stage”, whereas (T3–4, any N, any M) or (any T, N1, or M1) was regarded as “advanced clinical stage”.
Results:There were 61 patients with a median age of 45 years. After a median follow-up period of 7.3 years
(range: 2.1–22.1 years), the overall cause-specific survival rate was 98.36%. The patients with tumor diam-eters <0.5 cm were all recurrence-free. Advanced clinical stage, especially distant metastasis, was highly associated with recurrence or persistent disease.
Conclusion:Our results demonstrate excellent prognosis in multicentric PTMC patients. No patients with
tumor diameter <0.5 cm had recurrence or persistent disease. Tumor size is an important risk factor in patients with multicentric PTMC.
Key Words:microcarcinoma, multicentric tumor, papillary thyroid cancer, prognosis, tumor size
Although multicentricity is not a classical risk
factor of papillary thyroid carcinoma (PTC),1–3
multicentricity is frequently associated with an increased risk of recurrence, distant metastasis and
mortality in PTC patients.4–7To date, there are no
reports specifically on the clinical features and prognosis in patients with multicentric papillary thyroid microcarcinoma (PTMC). Accordingly,
©2011 Elsevier & Formosan Medical Association . . . . 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Chang Gung Memorial Hospital, Keelung, Chang Gung University, 2Department of General Surgery, 3Division of Endocrinology and Metabolism, Department of Internal Medicine, 4Department of Pathology, Chang Gung Memorial Hospital, Chang Gung University, and 5Department of Internal Medicine, Landseed Hospital, Taoyuan, Taiwan.
Received:December 8, 2009
Revised:May 4, 2010
Accepted:July 17, 2010
*Correspondence to:Dr Jen-Der Lin, Division of Endocrinology and Metabolism, Department
of Internal Medicine, Chang Gung Memorial Hospital, 5, Fu-Shin Street, Kweishan County, Taoyuan Hsien, Taiwan.
we retrospectively evaluated 61 patients with mul-ticentric PTMC from 325 PTMC patients treated at Chang Gung Medical Center in Taiwan, to show
their clinical manifestations and prognosis.8
Patients and Methods
PatientsBetween January 1987 and December 2008, 325 patients were diagnosed with PTMC and received
follow-up treatment for >2 years after surgery
at Chang Gung Medical Center, Taiwan. Among these, 61 had multicentric PTMC based on the final histopathological analyses of 325 PTMC patients. Multicentric PTMC was defined as the occurrence of at least two papillary cancer lesions in the thyroid, either within one lobe or in both.
The diameter of each lesion was ≤1.0 cm based
on the final pathologic records.
Initial survey and primary therapy
Most of the 61 patients underwent preoperative thyroid ultrasonography and fine needle aspi-ration cytology (FNAC) due to palpable thyroid nodules. All 61 patients received surgery, and frozen sections were obtained from most of them during surgery. In this paper, the term total thyroidectomy refers to near total or total thyroidectomy with or without central compartment and selective bilat-eral neck lymph node dissection; non-total thy-roidectomy refers to subtotal thythy-roidectomy or lobectomy.
Follow-up
After surgery, most patients received long-term thyroid hormone suppressive therapy. Postop-erative investigation usually consisted of thyroid ultrasonography, 74–185 MBq (2–5 mCi)
radioac-tive131I whole-body diagnostic scanning,
serum-stimulated thyroglobulin (Tg), anti-Tg antibody test, and thyroid function. Postoperative investi-gation was performed 1 month after surgery and at 6–12-month intervals thereafter. Serum Tg lev-els were measured with an immunoradiometric assay kit (CIS Bio International, Gif-sur-Yvette,
France). Large-dose 131I therapy was performed
when recurrent lesions were suspected. Surgery was performed whenever recurrent localized le-sions were confirmed based on thyroid ultrasonog-raphy with FNAC.
Outcome assessment
At the end of follow-up, patients were classified as “recurrence-free” or “recurrence or persistent dis-ease” based on the treatment outcome. Recurrence-free was defined as absence of distant metastasis and no local recurrence based on physical and noninvasive imaging (e.g. thyroid
ultrasonogra-phy, chest X-ray, 131I scan, computed tomography,
magnetic resonance imaging, and positron emis-sion tomography) at clinical follow-up. Recurrence was defined as distant metastasis or local tumor recurrence during clinical follow-up, and persis-tent disease as distant metastasis at diagnosis and throughout follow-up.
Tumor staging and risk stratification
Tumors were staged according to the International Union against Cancer (UICC) tumor–node–
metastasis (TNM) criteria (6th edition).3 In this
study, patients with confined intrathyroid PTMC (T1, N0, M0) were regarded as “early clinical stage”, and extra-thyroid extension PTMC (T3–4, any N, any M) or those with lymph node metastasis, soft tissue involvement, or distant metastasis (any T, N1, or M1) were regarded as “advanced clinical stage”.
Statistical analysis
Metric data were presented as median and range, and were analyzed by Mann–Whitney test. The
χ2or Fisher’s exact test was used for categorical
data. The association between tumor size and recurrence or persistent disease was explored by a receiver-operating-characteristic (ROC) curve. Besides, tumor size was dichotomized based on the best cut-off level chosen by ROC curve and the Yuden Index. Recurrence-free survival was an-alyzed by the Cox proportional-hazards model. Multivariate logistic regression was used to exam-ine factors associated with recurrence or persistent
disease of multicentric PTMC. Potential con-founders, collinearity and interaction between fac-tors were evaluated for building the final model.
Statistical significance was set at a two-sided p
value of ≤0.05. All analyses were conducted with
the use of statistical software (Stata, version 10.1; StataCorp, College Station, TX, USA).
Results
Clinical presentations
Based on the final histopathology from 325 PTMC patients, there were 61 multicentric and 264 uni-focal PTMC patients. Of the 61 multicentric PTMC patients, there were 46 women and 15 men with a median age of 45 years (range: 18–72 years). After a median follow-up period of 7.3 years (range: 2.1–21.1 years), one of the 61 patients died of metastatic thyroid cancer 5.33 years after
diagnosis.Twenty-seven patients presented with
asymptomatic thyroid nodules and underwent surgery because malignancy or suspected malig-nancy was detected by thyroid ultrasonography
and FNAC; 21 patients underwent surgery due to the presence of a large goiter; nine patients underwent surgery as a result of hyperthyroidism; and four patients underwent thyroid surgery be-cause metastatic lesions were suspected to origi-nate from the thyroid gland. Table 1 shows a comparison of the clinical characteristics between
multicentric (n=61) and unifocal (n=264) PTMC
patients.
Forty of the 61 multicentric PTMC patients presented with tumor in both lobes (65.6%), whereas the other 21 patients had PTMC con-fined to only one lobe (34.4%). Accordingly, mul-ticentricity was observed in 61 of 325 PTMC patients (18.8%), and the involvement of both lobes was detected in 40 of 325 PTMC patients (12.3%). Of the 61 multicentric PTMC patients, 50 underwent preoperative thyroid ultrasonogra-phy and FNAC, and PTC was detected in 17 of these (34%); 49 were examined by frozen sec-tions during surgery, and PTC was detected in 43 of 49 patients (87.8%). Sixteen of 61 patients were diagnosed with PTC incidentally based on final pathology (26.2%).
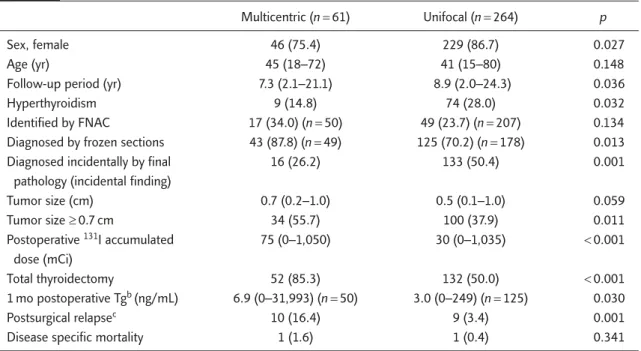
Table 1. Comparison of the clinical characteristics of multicentric and unifocal papillary thyroid microcarcinoma patientsa
Multicentric (n=61) Unifocal (n=264) p
Sex, female 46 (75.4) 229 (86.7) 0.027
Age (yr) 45 (18–72) 41 (15–80) 0.148
Follow-up period (yr) 7.3 (2.1–21.1) 8.9 (2.0–24.3) 0.036
Hyperthyroidism 9 (14.8) 74 (28.0) 0.032
Identified by FNAC 17 (34.0) (n=50) 49 (23.7) (n=207) 0.134 Diagnosed by frozen sections 43 (87.8) (n=49) 125 (70.2) (n=178) 0.013 Diagnosed incidentally by final 16 (26.2) 133 (50.4) 0.001
pathology (incidental finding)
Tumor size (cm) 0.7 (0.2–1.0) 0.5 (0.1–1.0) 0.059 Tumor size ≥0.7 cm 34 (55.7) 100 (37.9) 0.011 Postoperative 131I accumulated 75 (0–1,050) 30 (0–1,035) <0.001 dose (mCi) Total thyroidectomy 52 (85.3) 132 (50.0) <0.001 1 mo postoperative Tgb (ng/mL) 6.9 (0–31,993) (n=50) 3.0 (0–249) (n=125) 0.030 Postsurgical relapsec 10 (16.4) 9 (3.4) 0.001
Disease specific mortality 1 (1.6) 1 (0.4) 0.341
aData are presented as n (%) for categorical variables and median (range) for continuous variables; bPost total-thyroidectomy stimulated
Outcome and prognosis
Fifty-two of the 61 multicentric PTMC patients received total thyroidectomy (85.2%), including nine that underwent secondary operation for complete thyroidectomy; the other nine patients underwent non-total thyroidectomy (14.8%), in-cluding seven undergoing subtotal thyroidectomy and two undergoing lobectomy. The diameter of
each thyroid lesion in the 61 patients was ≤1.0 cm.
Fifty-three of the 61 patients (86.9%) were
ad-ministered at least 1.1 GBq (30 mCi) 131I during
postoperative follow-up. Two patients had per-manent hypoparathyroidism after total thyroi-dectomy; however, there were no other serious
complications related to surgery and 131I therapy.
Of the 61 multicentric PTMC patients, 51 (83.6%) were recurrence-free and 10 (16.4%) had recurrence or persistent disease at clinical follow-up, including six with recurrence and four with persistent disease. Table 2 shows a comparison of the clinical characteristics of 51 recurrence-free
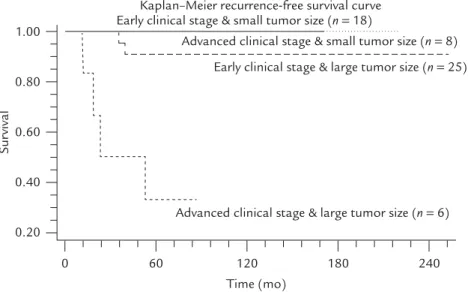
and 10 recurrence or persistent disease patients. The data shows that larger tumor diameter, higher postoperative serum stimulated Tg level and ad-vanced clinical stage were associated with recur-rence or persistent disease. The area under the ROC curve of tumor size predicting recurrence or persistent disease was 0.72 (95% confidence interval: 0.58–0.87). There was no recurrence or
persistent disease for tumor diameter <0.5 cm
(sensitivity 100%, specificity 35.3%), and the best cut-off level was 0.7 cm (sensitivity 90.0%,
speci-ficity 51.0%). Patients with tumor size <0.7 cm
were designated as the small tumor group and the remainder as the large tumor group. The Figure shows the Kaplan–Meier recurrence-free survival curves between patients with different tumor size and clinical stages (excluding 4 patients with per-sistent disease). Patients with advanced clinical stage and large tumor size had higher recurrent rate. Table 3, which summarizes the results of the multivariate logistic regression, reveals the factors
Table 2. Comparison of the clinical data of recurrence-free and recurrence or persistent disease multicentric papillary thyroid microcarcinoma patientsa
Recurrence-free Recurrence or persistent
(n=51) disease (n=10) p
Sex, female 42 (82) 4 (40) 0.010
Age (yr) 45 (18–72) 48 (24–72) 0.489
Follow-up period (yr) 7.3 (2.3–21.1) 8.1 (2.1–13.2) 0.907
Both lobes involved 34 (67) 6 (60) 0.725
Hyperthyroidism 7 (14) 2 (20) 0.633 TNM stage <0.001 T1, N0, M0 41 (80) 2 (20) T3–4, N0, M0 1 (2) 0 T1, N1, M0 6 (12) 3 (30) T3–4, N1, M0 3 (6) 1 (10) T1, N0, M1 0 3 (30) T3–4, N1, M1 0 1 (10)
Advanced clinical stage 10 (20) 8 (80) <0.001
Tumor size (cm) 0.6 (0.2–1.0) 0.95 (0.5–1.0) 0.025
Tumor size ≥0.7 cm 25 (49) 9 (90) 0.033
Postoperative 131I accumulated dose (mCi) 60 (0–445) 435 (90–1,050) <0.001
Total thyroidectomy 42 (82) 10 (100) 0.332
1 mo postoperative Tgb(ng/mL) 4.95 (0–35.4) (n=40) 186.1(3.05–31993) (n=10) 0.000
aData are presented as n (%) for categorical variables and medians (ranges) for continuous variables; bPost-total thyroidectomy
associated with recurrence or persistent disease. The results indicated that advanced clinical stage and large tumor size were significantly and inde-pendently associated with recurrence or persistent disease of multicentric PTMC.
Discussion
Although PTMC patients usually have an excellent
prognosis,9–13the clinical behavior and prognosis
of multicentric PTMC patients is not well known. This study revealed an excellent cause-specific sur-vival rate of 98.4% for multicentric PTMC patients after a median follow-up period of 7.3 years.
Based on the results of this study, patients with multicentric PTMC have larger tumor diameter and higher rate of recurrence or persistent disease than
those with unifocal PTMC, although their prog-nosis is excellent. The frequency of
multicentric-ity in PTMC is reported to be 15–32%.10–12,14,15
Both thyroid lobes are involved in 10–19% of
PTMC patients.11,12Our results are similar to
pre-vious studies, in which multicentricity was ob-served in 18.8% of patients, and both lobes were involved in 12.3% of cases. Neck ultrasonogra-phy is a sensitive and cost-effective method for the detection of thyroid cancer, and it increases the frequency of preoperative diagnosis of thyroid
microcarcinomas.16–19Both ultrasonography with
FNAC and frozen sections during surgery are helpful in identifying multicentricity of thyroid cancer in PTMC patients, which might avoid sec-ondary operation for these patients, although the diagnostic rate of FNAC is low (23.7–34%) for PTMC patients, because it is not easy to perform a successful FNAC on thyroid microcarcinoma.
Although there have been no reports specif-ically on the prognostic factors in patients with multicentric PTMC, previous studies on PTMC patients have indicated that tumor size, postop-erative Tg level, presence of lymph node metas-tases at diagnosis, sex, and extent of surgery are
prognostic factors.10,12,14,15,20,21Based on the ROC
curve in the present study, multicentric PTMC
patients with tumor diameter <0.5 cm tended to
Sur
vival
Kaplan–Meier recurrence-free survival curve Early clinical stage & small tumor size (n= 18)
Early clinical stage & large tumor size (n= 25) Advanced clinical stage & small tumor size (n= 8)
Advanced clinical stage & large tumor size (n= 6) 1.00 0.80 0.60 0.40 0.20 0 60 120 Time (mo) 180 240
Figure.Kaplan–Meier recurrence-free survival curves between patients with different tumor size and clinical stages. Solid line: early clinical stage and small tumor size; dotted line: advanced clinical stage and small tumor size; long dashed line: early clinical stage and large tumor size; short dashed line: advanced clinical stage and large tumor size.
Table 3. Factors predicting recurrence or persistent disease in patients with multicentric papillary thyroid carcinoma
Variables OR (95% CI)a p
Clinical stage 45.2 (5.1–397.2) 0.001 (early vs. advanced)
Tumor size 234.5 (3.5–15530.6) 0.011
aLogistic regression. Early clinical stage: (T1, N0, M0); advanced
clinical stage: (T3–4, any N, any M) or (any T, N1, or M1).
be recurrence-free. From the results of multivari-ate logistic regression, advanced clinical stage and larger tumor size were independently associated with recurrence or persistent disease. Tumor size was a more important risk factor than clinical stage in the present study. As the tumor size increased, the prognosis became poorer. Additionally, ad-vanced clinical stage in the present study included lymph node metastasis. Although lymph node metastasis increased risks of tumor recurrence, it did not influence survival rate in previous
stud-ies.22,23Four patients had persistent disease in the
present study, and all of them underwent thyroid surgery because metastatic lesions originated from the thyroid; these patients actually had the poor-est clinical outcome, and one of them died of metastatic thyroid cancer.
Whether multicentric tumors originate from different unrelated clones or from one focus with intrathyroid spreading is unclear. Results from studies such as those involving polymerase chain reactions with the human androgen receptor gene or BRAF gene imply that noncontiguous tumor
foci originate from independent precursors,24,25
whereas another study has shown that multicentric
papillary lesions arise from the same clone.26To
date, it is unknown whether the clinical presen-tations and prognosis differ between these two disease subgroups in multicentric PTC or PTMC patients.
There were some limitations in this retrospec-tive study. Because 14.8% of multicentric PTMC cases had non-total thyroidectomy, it might have been difficult to identify recurrence by serum Tg
levels and 131I scans. We determined recurrence
based mainly on physical and imaging studies
(e.g. thyroid ultrasonography, chest X-ray, 131I scan,
computed tomography, magnetic resonance im-aging, and positron emission tomography) other than serum Tg levels, and thus recurrence might have been under-reported as a result.
In conclusion, multicentric PTMC patients might have an excellent prognosis. Patients with
tumor diameter <0.5 cm tend to be
recurrence-free. Tumor size is an important independent prognostic factor in multicentric PTMC patients.
References
1. Cady B, Rossi R. An expanded view of risk-group definition in differentiated thyroid carcinoma. Surgery 1988;104: 947–53.
2. Hay ID, Bergstralh EJ, Goellner JR, et al. Predicting outcome in papillary thyroid carcinoma: development of a reliable prognostic scoring system in a cohort of 1779 patients surgically treated at one institution during 1940 through 1989. Surgery1993;114:1050–7.
3. Sobin LH, Wittekind CH. UICC: TNM Classification of
Malignant Tumors, 6th edition. New York: Wiley–Liss,
2002:52–6.
4. Katoh R, Sasaki J, Kurihara H, et al. Multiple thyroid in-volvement (intraglandular metastasis) in papillary thyroid carcinoma. A clinicopathologic study of 105 consecutive patients. Cancer1992;70:1585–90.
5. Loh KC, Greenspan FS, Gee L, et al. Pathological tumor-node-metastasis (pTNM) staging for papillary and follicular thyroid carcinomas: a retrospective analysis of 700 patients.
J Clin Endocrinol Metab1997;82:3553–62.
6. Mazzaferri EL, Jhiang SM. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med1994;97:418–28.
7. Ringel MD, Ladenson PW. Controversies in the follow-up and management of well-differentiated thyroid cancer.
Endocr Relat Cancer2004;11:97–116.
8. Lin JD, Kuo SF, Chao TC, et al. Incidental and noninciden-tal papillary thyroid microcarcinoma. Ann Surg Oncol
2008;15:2287–92.
9. Baudin E, Travagli JP, Ropers J, et al. Microcarcinoma of the thyroid gland. The Gustave Roussy Institute experience.
Cancer1998;83:553–9.
10. Chow SM, Law SC, Chan JK, et al. Papillary microcarci-noma of the thyroid-prognostic significance of lymph node metastasis and multifocality. Cancer2003;98:31–40. 11. Hay ID, Grant CS, Van Heerden JA, et al. Papillary thyroid
microcarcinoma: a study of 535 cases observed in a 50-years period. Surgery1992;112:1139–47.
12. Roti E, Rossi R, Trasforini G, et al. Clinical and histological characteristics of papillary thyroid microcarcinoma: results of a retrospective study in 243 patients. J Clin Endocrinol
Metab2006;91:2171–8.
13. Sakorafas GH, Giotakis J, Stafyla V. Papillary thyroid microcarcinoma: a surgical perspective.Cancer treat rev
2005;31:423–38.
14. Pelizzo MR, Boschin IM, Toniato A, et al. Papillary thyroid microcarcinoma (PTMC): prognostic factors, management and outcome in 403 patients. Eur J Surg Oncol2006;32: 1144–8.
15. Küçük NÖ, Tari P, Tokmak E, et al. Treatment for micro-carcinoma of the thyroid–clinical experience. Clin Nucl Med2007;32:279–81.
16. Lin JD, Huang BY, Chao TC, et al. Diagnosis of occult thyroid carcinoma by thyroid ultrasonography with fine needle aspiration cytology. Acta Cytol1997;41:1751–6.
17. Nam-Goong IS, Kim HY, Gong G, et al. Ultrasonography-guided fine-needle aspiration of thyroid incidentaloma: correlation with pathological findings. Clin Endocrinol2004; 60:21–8.
18. Lin JD, Chao TC, Huang BY, et al. Thyroid cancer in the thyroid nodules evaluated by ultrasonography and fine needle aspiration cytology. Thyroid2005;15:702–11. 19. Berker D, Aydin Y, Ustun I, et al. The value of fine-needle
aspiration biopsy in subcentimeter thyroid nodules.
Thyroid2008;18:603–8.
20. Lin JD, Chen ST, Chao TC, et al. Diagnosis and therapeutic strategy for papillary thyroid microcarcinoma. Arch Surg
2005;140:940–5.
21. Pellegriti G, Scollo C, Lumera G, et al. Clinical behavior and outcome of papillary thyroid cancers smaller than 1.5 cm in diameter: study of 299 cases. J Clin Endocrinol
Metab2004;89:3713–20.
22. Lin JD, Liou MJ, Chao TC, et al. Prognostic variables of papillary and follicular thyroid carcinoma patients with lymph node metastases and without distant metastases.
Endocr Relat Cancer1999;6:109–15.
23. Shaha AR. Implications of prognostic factors and risk groups in the management of differentiated thyroid cancer.
Laryngoscope2004;114:393–402.
24. Shattuck TM, Westra WH, Ladenson PW, et al. Independent clonal origins of distinct tumor foci in multifocal papillary thyroid carcinoma. N Engl J Med2005;352:2406–12. 25. Giannini R, Ugolini C, Lupi C, et al. The heterogeneous
dis-tribution of BRAF mutation supports the independent clonal origin of distinct tumor foci in multifocal papillary thyroid carcinoma. J Clin Endocrinol Metab2007;92:3511–6. 26. McCarthy RP, Wang M, Jones TD, et al. Molecular
evidence for the same clonal origin of multifocal papillary thyroid carcinomas. Clin Cancer Res2006;12:2414–8.