Low Expression of Human Epithelial Sodium Channel in Airway
Epithelium of Preterm Infants With Respiratory Distress
Otto Helve, MD*; Olli M. Pitka¨nen, MD, PhD‡; Sture Andersson, MD, PhD*; Hugh O’Brodovich, MD§㛳; Turkka Kirjavainen, MD, PhD*; and Gail Otulakowski, PhD㛳
ABSTRACT. Objective. Active ion transport is critical to postnatal clearance of lung fluid. The importance of epithelial sodium channel (ENaC) in this clearance has been demonstrated in animal studies in which␣-ENaC knockout mice died postnatally as a result of respiratory insufficiency. In animals, the expression of ␣-ENaC in respiratory epithelium is dependent on gestational age, but when assessed by in situ hybridization in the human (h), the mRNA is present from the earliest stages of pulmonary development. Therefore, the purpose of the present investigation was to quantify mRNA of the␣-,
-, and␥-hENaC subunits of newborn preterm infants with respiratory distress and compare the gene expres-sion data against those detected in healthy term infants. In addition, the effect of systemic dexamethasone ther-apy on the 3 hENaC subunits was studied in 4 preterm infants who received prolonged assisted ventilation.
Methods. The expression of subunits of hENaC was determined in samples taken from nasal respiratory ep-ithelium of 7 healthy term infants (gestation age: 39.3ⴞ 0.9 weeks [mean ⴞ standard deviation) and 5 preterm infants (gestational age: 27.2ⴞ 0.9 weeks) with respira-tory distress syndrome within 5 hours of birth. Beta-methasone had been given to all mothers of preterm infants. In 4 additional preterm infants who still required assisted ventilation at 43 ⴞ 6 days postnatal age, the expression of␣-hENaC was determined in samples taken before and during treatment with dexamethasone.
Results. Preterm infants with respiratory distress syndrome had low expression of all hENaC subunits relative to healthy term infants (␣-hENaC: 5.38 ⴞ 2.01 [amol/fmol cytokeratin 18] vs 9.13ⴞ2.26;-hENaC: 2.44
ⴞ1.43 vs 4.25ⴞ1.10;␥-hENaC: 2.43ⴞ0.11 vs 6.81ⴞ3.24). Each of the 4 preterm infants who were treated with dexamethasone at⬃1 month of age showed an increase in expression of␣-hENaC and-hENaC subunit normal-ized to cytokeratin 18.
Conclusion. All 3 subunits of the hENaC are low in preterm relative to full-term infants.␣-hENaC mRNA in respiratory epithelium is increased by therapeutic doses of glucocorticosteroid. Low expression of ␣-hENaC in human respiratory epithelium may play a role in the pathogenesis of respiratory distress in preterm infants.
Pediatrics2004;113:1267–1272;epithelium, glucocorticoid, ion transport, lung liquid, respiratory distress syndrome.
ABBREVIATIONS. ENaC, epithelial sodium channel; PD, poten-tial difference; RDS, respiratory distress syndrome; hENaC, hu-man epithelial sodium channel; QRT-PCR, quantitative reverse transcriptase–polymerase chain reaction; CK, cytokeratin.
A
ctive ion transport plays a critical role in the liquid movement across the fetal and perina-tal lung epithelium.1–3The fetal lung activelysecretes Cl⫺ and fluid throughout gestation and develops the ability to actively reabsorb Na⫹ only during late gestation.4 At birth, the mature lung
switches from active Cl⫺and fluid secretion to active Na⫹and fluid absorption in response to circulating catecholamines and changes in oxygen tension.2,5–7
The active absorption of fluid is coupled with vecto-rial Na⫹transport from the apical to the basolateral side of the epithelium. Inability to switch from secre-tion of fluid to absorpsecre-tion may contribute to the respiratory distress of the preterm infant.2,3,8
The amiloride-sensitive epithelial sodium channel (ENaC) is important for transepithelial lung liquid movement. The channel consists of 3 subunits, of which the ␣-subunit is prerequisite for appreciable channel function.9 Experiments in several animal
models indicate that the function of ENaC is vital for lung fluid clearance at birth. Instillation of amiloride, a Na⫹ channel blocker, into the trachea of newborn guinea pigs results in impediment of lung liquid absorption, hypoxia, and respiratory distress.10
Loss-of-function mutations of the murine ␣-ENaC gene results in early death of the newborn animals as a result of respiratory distress. Microscopy of the lungs of these homozygous knock-out test animals re-veals normal morphologic development; however, gravimetric measurements showed excess lung fluid, and electrophysiologic measurements of the trachea showed impaired response to amiloride, suggesting reduced Na⫹ channel activity.11In humans, studies
in premature infants have shown that the amiloride-sensitive drop in electrical potential difference (PD) between the nasal epithelium and the subcutaneous space, a surrogate measure for Na⫹ transport and ENaC activity, is decreased in infants who develop neonatal respiratory distress (RDS).8
Administration of corticosteroids for the treatment of neonatal RDS is an established intervention to
From the Departments of *Neonatology and ‡Cardiology, Hospital for Children and Adolescents, Helsinki University Central Hospital, Helsinki, Finland; §Departments of Paediatrics and Physiology, University of To-ronto, ToTo-ronto, Ontario, Canada; and㛳Programme in Lung Biology Re-search, Research Institute of the Hospital for Sick Children, Toronto, On-tario, Canada.
Received for publication Jan 17, 2003; accepted Jul 14, 2003.
Reprint requests to (O.H.) Hospital for Children and Adolescents, Univer-sity of Helsinki, Biomedicum B429b, PO Box 700, FIN-00029 HUS, Finland. E-mail: otto.helve@hus.fi
reduce inflammation and promote lung maturity and surfactant production. Corticosteroids have been shown to increase lung␣-ENaC expression and func-tion in fetal rat lung; however, data on the effects of glucocorticoid on human ENaC (hENaC) expression in humans in vivo have not previously been reported in the literature. In cultures of human fetal lung explants, a high dose of dexamethasone induced not only the␣-subunit but all 3 hENaC-subunit mRNAs by 2- to 3-fold.12Dexamethasone treatment induced
␣-hENaC13and␥-hENaC14mRNAs of human lung–
derived epithelial H441 cells (results for -hENaC have not been reported in the literature), but in the A549 cell line, dexamethasone induced significant increases in - and ␥-hENaC mRNA and protein, with no significant effect on␣-hENaC.15These
stud-ies indicate that lung explants and human cell lines maintained under different culture conditions gener-ate a variety of hENaC-relgener-ated responses to glucocor-ticoid treatment, any or all of which may not accu-rately mimic the response of the intact individual, and emphasize the need to complete in vivo experi-ments to determine the effects of glucocorticoid treatment on hENaC expression in human respira-tory epithelium.
The expression of hENaC mRNA along the respi-ratory tract of adult humans has recently been de-scribed using both in situ hybridization and quanti-tative reverse transcriptase–polymerase chain reaction (QRT-PCR).16However, no quantitative data exist on
hENaC expression in respiratory epithelium of term and preterm infants. The nasal epithelium has been used as a representative of pulmonary epithelium because it displays electrophysiologic characteristics comparable to the more distal airway epitheli-um.17–19
The aim of the present work was to study whether the epithelial expression of ␣-, -, and ␥-hENaC mRNA immediately after birth is different in pre-term infants with RDS when compared with pre-term infants without lung disease. In addition, we studied whether dexamethasone therapy had an effect on the hENaC subunits’ mRNA expression in 4 continu-ously ventilator-dependent preterm infants at 35 to 50 days of age.
METHODS Patients
Patient data are given in Tables 1 and 2. Newborn infants were studied within 5 hours of birth. All 7 control term infants were
healthy, whereas the 5 preterm infants had RDS; none of them received surfactant before sampling. The mothers had received ante partum betamethasone treatment as a total of 1 to 4 doses of 12 mg intramuscularly (from 18 days to 24 hours before delivery). Two mothers had received ante partum terbutaline: 1 received an infusion of 1 mg/hour for 6 hours, which was discontinued 5 hours before delivery; the second received 5 mg/hour for 48 hours until delivery. None of the mothers had preeclampsia, and none had diabetes or any other chronic conditions that would impose additional risks to pregnancy. The initial arterial to alveolar oxy-gen tension ratio of the 5 preterm infants was 0.05 to 0.8, and they received an average of 2 doses of surfactant (Curosurf; Chiesi, Parma, Italy). Four additional preterm infants with prolonged respiratory distress were studied. Treatment was started at 43⫾6 postnatal days (gestational age: 24.9 ⫾ 0.2 weeks; gestational weight: 764⫾178 g). The infants received dexamethasone treat-ment at a dose of 0.2 mg/kg/day as 2 daily doses for 3 days and thereafter at 0.1 and 0.05 mg/kg/day for 3 and 5 days, respec-tively. All 4 infants were weaned successfully from the ventilator. Initial samples were gathered before the commencement of dexa-methasone treatment. Subsequently, samples were gathered in these patients 7 to 20 hours after the initial dose.
The study protocol was approved by the Ethics Committee of the Hospital for Children and Adolescents of the Helsinki Univer-sity Hospital and by the Research Ethics Board of the Hospital for Sick Children, Toronto. The parents gave informed consent.
Sample Collection and Storage
The samples were prepared and quantified as described previ-ously.16,20The nasal samples were gathered under direct vision by
scraping the nasal epithelium using a Rhino-Probe (Arlington Scientific, Springville, UT). In the patients who were treated with dexamethasone, the samples were gathered from the nare con-tralateral to the nare with the nasotracheal intubation tube. A commercially available purification kit was used for total RNA preparation (RNeasy Kit; Qiagen, Valencia, CA). The samples were collected on ice and immediately dispersed with insulin syringe into the lysis buffer containing 10L of -mercaptoetha-nol per 1 mL of buffer. The samples were stored at⫺80°C, and the succeeding purification steps were performed as sets of 5 to 8 samples as described by the manufacturer. The purified total RNA preparations were quantified by slot blot analysis, diluted to 2.4 ng/L, and stored at⫺80°C in single-use aliquots as described previously.20
In the preterm infants who received dexamethasone treatment, the total RNA quantification of the turbinate specimens was per-formed using a commercially available kit including preweighed standard RNA preparation and RiboGreen quantification reagent (RiboGreen RNA Quantitation Kit; Molecular Probes, Eugene, OR). The emission at 520 nm of the adducts was measured after excitation at 480 nm using a spectrofluorometer (LS50B; Perkin Elmer, Shelton, CT), and the sample RNA contents were deduced from the standard plot.
Quantification of mRNA
Samples for competitive QRT-PCR were thawed on ice and diluted to give a dilution series that extended from 24 to 0.375 ng of total RNA. Each reaction mixture contained a constant amount
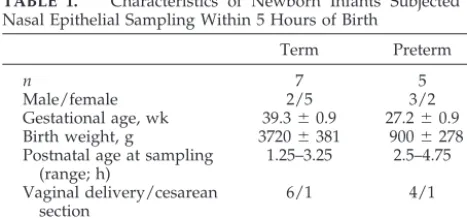
TABLE 1. Characteristics of Newborn Infants Subjected to Nasal Epithelial Sampling Within 5 Hours of Birth
Term Preterm
n 7 5
Male/female 2/5 3/2
Gestational age, wk 39.3⫾0.9 27.2⫾0.9 Birth weight, g 3720⫾381 900⫾278 Postnatal age at sampling
(range; h)
1.25–3.25 2.5–4.75
Vaginal delivery/cesarean section
6/1 4/1
1-Min Apgar score 9 6⫾2
Umbilical artery pH 7.29⫾0.1 7.26⫾0.1
TABLE 2. Characteristics of Preterm Infants Who Received Dexamethasone as a Result of Severe Lung Disease
n 4
Male/female 3/1
Gestational age, wk 24.9⫾0.2
Birth weight, g 764⫾178
Vaginal delivery/cesarean section 3/1
Postnatal age, d 43⫾6
Postnatal weight, g 1205⫾264
Fio2 0.60⫾0.18
First day on dex 0.51⫾0.10 Second day on dex 0.41⫾0.08 Third day on dex 0.34⫾0.05 Days to extubation after commencement
of dex (range: d;n⫽4)
2–5
of the truncated␣-,-,␥-ENaC or cytokeratin 18 (CK18) cRNA as a competitive internal standard. The primers used and the reaction conditions for the RT and PCR reactions were as described previ-ously.16,20 PCR products were separated by electrophoresis,
stained with ethidium bromide, and quantified using a charged coupled device camera and SCION Image software. ENaC and CK18 mRNA concentrations were calculated (in attomoles per microgram of total RNA) as described.20The ENaC expression of
each sample was normalized against that of CK18, which was used as an epithelial marker (ENaC: CK18, attomole per femto-mole).
Statistical Analysis
Patients’ clinical data are presented as mean⫾standard devi-ation. QRT-PCR was done in duplicate for each target mRNA in each sample; assay results were averaged and the mean was used as a single datum. The normalized assay data (ENaC/CK18) are presented as mean⫾standard deviation. Comparisons were per-formed with the Mann-WhitneyUtest.P⬍.05 was considered statistically significant.
RESULTS
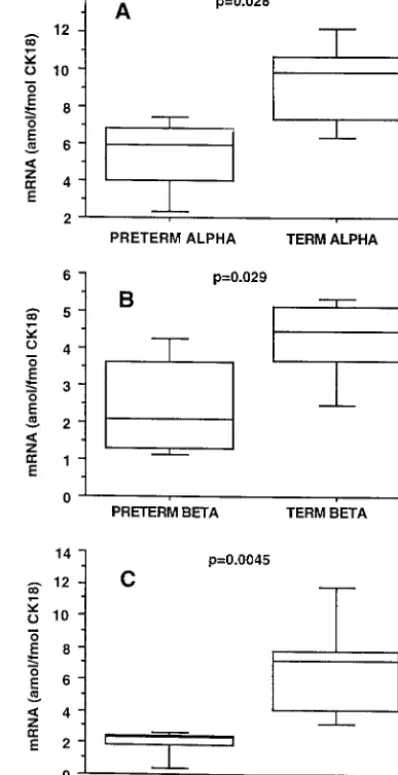
The subunits ␣-, -, and ␥-hENaC and CK18
mRNA were found in all samples. We found that expression of all 3 hENaC subunits in the nasal epi-thelium were significantly lower in preterm infants relative to healthy term infants. In healthy term
in-fants, ␣-hENaC mRNA was 9.1 ⫾ 2.3, -hENaC
mRNA was 4.3⫾1.1, and␥-hENaC mRNA was 6.8
⫾3.2 amol/fmol CK18 (Fig 1). In the preterm infants,
␣-hENaC mRNA was 5.4 ⫾ 2.0, -hENaC mRNA
was 2.4 ⫾ 1.4, and ␥-hENaC mRNA was 2.4 ⫾ 0.1
amol/fmol CK18 (Fig 1). The expression of each sub-unit was significantly higher in term than in preterm infants (P⬍ .05)
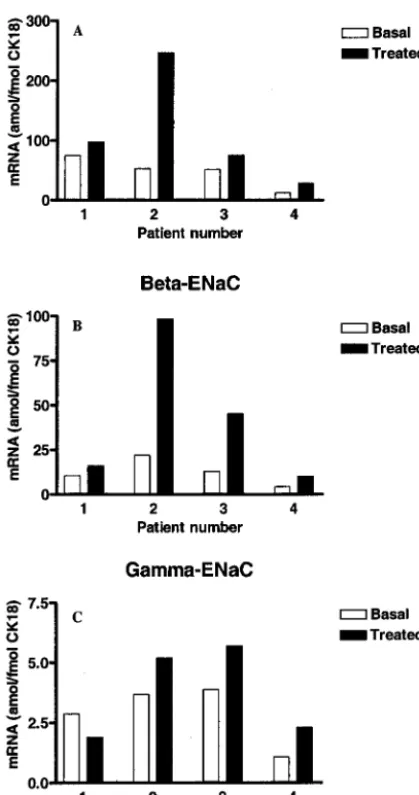
The␣- and-hENaC/CK18 mRNA ratios in the 4 premature infants who were sampled after several weeks of assisted ventilation increased markedly af-ter commencement of dexamethasone treatment. Ini-tial ␣-hENaC (amol/fmol CK18) was 47.9 ⫾ 26.1,
-hENaC (amol/fmol CK18) was 12.4 ⫾ 7.3, and
␥-hENaC (amol/fmol CK18) was 2.9 ⫾ 1.3. Twenty hours after treatment with dexamethasone, there was an increase of 143 ⫾ 155% from basal in the
expression of ␣-hENaC/CK18 mRNA and an
in-crease of 195⫾130% in-hENaC/CK18 mRNA. In 3
patients, ␥-hENaC mRNA relative to CK18 mRNA
increased slightly, whereas in 1 patient, the ratio decreased (Fig 2).
DISCUSSION
Neonatal RDS results from a relative surfactant deficiency and excess lung liquid.2,10,11,21–23 The
presence of excess lung liquid has been documented in infants; those who die of respiratory distress dur-ing the first few hours of life have significantly higher lung water content relative to infants who survive beyond 48 hours.24Our present quantitative
data demonstrate that all hENaC subunit mRNAs are significantly lower in preterm infants with RDS than in healthy term infants and suggest that hENaC expression is regulated quantitatively during human fetal airway development.
The role of hENaC function in neonatal RDS has not been elucidated fully. In rodents, ENaC mRNA expression in the lung has been correlated to gesta-tional age.25–27 Mouse models indicate that the
␣-ENaC subunit is essential for lung fluid clearance at birth, whereas - and ␥-ENaC knockout mice, which die as a result of renal insufficiency, display only a mild lung phenotype.11,28In contrast to
labo-ratory animals, patients with the recessive systemic pseudohypoaldosteronism type I, who carry a loss-of-function mutation in hENaC ␣- or -subunit or their regulatory regions, do not develop neonatal respiratory disease.25,29–32 However, the later lung
phenotype of these patients includes recurrent respi-ratory infections, increased volume of airway surface liquid, and absence of amiloride-sensitive sodium transport in the nasal epithelium, indicating an im-pact of the hENaC mutations on lung fluid balance. Despite lower basal readings within their nasal tur-binates, these patients demonstrate inhibition of transepithelial PD after topical amiloride that is com-patible with reduced but existing hENaC activity.31
Fig 1. Expression of␣- (A),- (B), and␥- (C) ENaC subunits in nasal epithelium in newborn preterm (n⫽5) and term (n⫽7) infants. ENaC subunit and CK18 mRNA was quantified in each sample as described in the text and previously.20Data were
Although ␣-ENaC has been considered as a vital subunit for channel function, recent experimental data have demonstrated that defective␣-ENaC sub-units when coexpressed with the other subsub-units are able to produce significant channel activity in Xeno-pusoocytes.30 It thus is possible that mutant ENaC
subunits or other compensatory mechanisms such as increased mucociliary function are sufficient to sup-port lung fluid clearance, explaining the lack of neo-natal RDS in patients with systemic pseudohypoal-dosteronism type I.30,31
Although expression of all 3 subunits is required for maximal channel activity, inXenopusoocytes, the coexpression of only 2 subunits of the ENaC channel as␣:␥- or␣:-ENaC results in appreciable transmem-brane sodium conductance; the biophysical proper-ties of such␣␥versus␣channels differ significantly in terms of single-channel conductance, Na⫹
selec-tivity, and amiloride sensitivity.33 Additional
evi-dence exists suggesting unique roles for theand␥ subunits, including a recent publication that sug-gested that the␥subunit is more important for chan-nel surface expression than the subunit.34
ENaC subunit mRNA expression varies quantita-tively along the respiratory tract in adult humans.16
Only qualitative data are available on human fetal and neonatal airway, demonstrating constitutive ex-pression of␣-hENaC mRNA throughout lung devel-opment.35 In utero, the entire airway epithelium is
likely to be under a homogeneous humoral and local ontogenic regulation, and in fact previous data have demonstrated that nasal epithelial function shows a correlation with neonatal lung disease, supporting the idea that it can be used as a surrogate for lung epithelium.8,36 Therefore, we think that the present
observation of low expression of all hENaC subunits in the nasal epithelia of preterm infants with RDS is important, and its comparison with previous inves-tigations highlights interesting points. Both full-term and preterm infants express considerably less␣- and
-hENaC mRNA than adults.16 In contrast, the
ex-pression of␥-hENaC seems to peak at term gestation, with much lower levels in both preterm infants and
adults. ␣-ENaC thus is expressed at markedly
greater levels than - and ␥-ENaC mRNA in adult respiratory tract epithelia: we observed mRNA ratios of 20:4:1 in human20 and 50:1:5 in mouse nasal
tur-binate37by competitive QRT-PCR. The ratio of␣::␥
mRNA observed within the infants included in this study was 2:1:1 and resembles the predicted protein stoichiometry (␣2␥) presented previously.38 This
suggests that posttranscriptional steps may be regu-lated differently in the newborn compared with the adult respiratory epithelium.
Preterm infants whose mother has received beta-methasone ante partum show improved postnatal pulmonary adaptation compared with infants of un-treated mothers.39The mothers of all preterm infants
in our study had received betamethasone, yet these infants expressed lower levels of hENaC mRNA than full-term infants, suggesting that the “basal” level of hENaC in untreated preterm infants may be even lower. Dexamethasone treatment of the 4 infants with prolonged ventilator dependence in our study was efficacious in improving fraction of inspired oxygen dependence, as illustrated in Table 2, and all 4 were weaned from the ventilator within 5 days of initiating steroid treatment.
Previous studies have shown that glucocorticoid treatment in newborn infants increases dynamic lung compliance and expiratory time constant and decreases total pulmonary resistance after 12 hours of treatment.40 Glucocorticoid treatment improves
pulmonary mechanics and gas exchange in a signif-icant number of preterm infants with prolonged de-pendence on assisted ventilation.41 There are many
potential mechanisms whereby postnatal glucocorti-coid treatment could improve lung function. These may include anti-inflammatory effects, increases in surfactant production, and induction of ENaC (for review, see ref 41). In test conditions, glucocorticoid treatment increases clearance of pulmonary edema
Fig 2. Expression of␣- (A),- (B), and␥- (C) ENaC subunits in nasal epithelium in 4 preterm infants before (basal) and 7 to 20 hours after the commencement of dexamethasone treatment (treated). ENaC subunit and CK18 mRNA was quantified in each sample as described in the text and previously.20Data were
in adult rats and␣-ENaC expression in the fetal rat lung.25,42 Significant differences seem to exist,
how-ever, in the ontogeny and glucocorticoid regulation of ENaC expression in lung between rodent and human. Both Northern and in situ hybridization analysis of developing rat and mouse lung indicate that ENaC subunit mRNAs are expressed only late in gestation, whereas in humans, hENaC mRNA has been detected in all airway epithelia from the earliest fetal lung bud stage onward.25–27,43In this study, we
focused on the effects of dexamethasone on ENaC mRNAs in premature infants with prolonged RDS. This report represents the first assessment of glu-cocorticoid effects on ENaC synthesis in human in-fants and indicates that both the ␣ and  subunits seem to be upregulated by dexamethasone.␥-hENaC mRNA was increased in 3 of the 4 infants. Our findings suggest that glucocorticoid induces transep-ithelial sodium movement through pathways such as ENaC, which may represent 1 mechanism whereby glucocorticoids improve pulmonary function of the preterm infants. Accordingly, it would be important, indeed, to find out by direct in situ measurements in a larger group of clinical patients whether the in-creased expression of hENaC subunits after glu-cocorticoid therapy correlates with an increased amiloride-sensitive transepithelial PD in the nasal turbinate.
Our study included newborn premature infants whose gestational age ranged from 26 to 28 weeks. At this stage of maturity, the infants are regularly born with respiratory distress, and finding proper nonaffected infants of similar antenatal history is difficult. In addition, other factors than mere mRNA content may participate in the regulation of the func-tional Na⫹ channels. These include increased trans-lational efficiency by mRNA stabilization and differ-ences in posttranslational assembly of the subunits in the membrane. Nevertheless, the present data sup-port the hypothesis that neonatal RDS is associated with reduced ENaC expression. Surprising is that in preterm infants with prolonged ventilatory depen-dence, the␣-hENaC mRNA (amol/fmol CK18) ratio was higher and showed greater interindividual vari-ation in comparison with preterm infants’ samples obtained within 5 hours of birth. The increased ex-pression of␣-hENaC may reflect postnatal matura-tion of the airway epithelium. These infants had been exposed to nasotracheal intubation with the opposite side serving as the route for suction of the nasopha-ryngeal secretions. Also, the side of sample collection had to accommodate the nasogastric feeding tube. In addition to postnatal age, factors including inhaled oxygen concentration, circulating catecholamines in-duced by prolonged stress, and cell turnover as a result of local trauma during intubation may have affected hENaC and CK18 mRNA expression.6,44,45
These factors preclude drawing firm conclusions when comparing hENaC mRNA expression in in-fants sampled soon after birth with inin-fants under prolonged ventilation but do not invalidate our ob-servation of increased hENaC mRNA expression in vivo in response to dexamethasone treatment. Of note is that the hENaC subunit values measured in
these chronically ventilator-dependent infants were in the range of those reported in adults in an earlier paper by our research group.16
In conclusion, in newborn preterm infants with RDS, the expression of all subunits of hENaC in respiratory epithelium is significantly lower than in healthy term infants. Our findings suggest that the human upper airway epithelial hENaC expression and hence Na⫹transport are developmentally regu-lated and can be augmented by treatment with the glucocorticoid dexamethasone. The present findings call for additional studies to elucidate whether in the preterm infant respiratory distress can be alleviated by therapeutic induction of hENaC.
ACKNOWLEDGMENTS
This work was supported by Wilhelm och Else Stockmanns Stiftelse, the Foundation for Pediatric Research, Finska La¨karesa¨ll-skapet, Sigrid Juse´lius Foundation, the Special Governmental Sub-sidy for Health Sciences, the Ontario Thoracic Society, the Cana-dian Institutes in Health Research group Grant in Lung Biology, and the Hospital for Sick Children (Toronto).
We thank the personnel of the neonatal unit of the Hospital for Children and Adolescents for kind cooperation; Marita Suni, Mar-jatta Vallas, and Yanxia Wen for excellent technical assistance; and the Pediatric Graduate School of the University of Helsinki for support.
REFERENCES
1. Strang LB. Fetal lung liquid: secretion and reabsorption.Physiol Rev. 1991;71:991–1016
2. Pitkanen OM, O’Brodovich HM. Significance of ion transport during lung development and in respiratory disease of the newborn.Ann Med. 1998;30:134 –142
3. O’Brodovich H. Epithelial ion transport in the fetal and perinatal lung. Am J Physiol.1991;261(suppl):C555–C564
4. Olver RE, Strang LB. Ion fluxes across the pulmonary epithelium and the secretion of lung liquid in the foetal lamb.J Physiol.1974;241:327–357 5. Brown MJ, Olver RE, Ramsden CA, Strang LB, Walters DV. Effects of adrenaline and of spontaneous labour on the secretion and absorption of lung liquid in the fetal lamb.J Physiol.1983;344:137–152
6. Pitkanen O, Transwell AK, Downey G, O’Brodovich H. Increased Po2 alters the bioelectric properties of fetal distal lung epithelium.Am J Physiol.1996;270(suppl):L1060 –L1066
7. Bland RD, Nielson DW. Developmental changes in lung epithelial ion transport and liquid movement.Annu Rev Physiol.1992;54:373–394 8. Barker PM, Gowen CW, Lawson EE, Knowles MR. Decreased sodium
ion absorption across nasal epithelium of very premature infants with respiratory distress syndrome.J Pediatr.1997;130:373–377
9. Canessa CM, Schild L, Buell G, et al. Amiloride-sensitive epithelial Na⫹ channel is made of three homologous subunits. Nature. 1994;367: 463– 467
10. O’Brodovich H, Hannam V, Seear M, Mullen JB. Amiloride impairs lung water clearance in newborn guinea pigs.J Appl Physiol.1990;68: 1758 –1762
11. Hummler E, Barker P, Gatzy J, et al. Early death due to defective neonatal lung liquid clearance in alpha-ENaC-deficient mice.Nat Genet. 1996;12:325–328
12. Venkatesh VC, Katzberg HD. Glucocorticoid regulation of epithelial sodium channel genes in human fetal lung. Am J Physiol. 1997; 273(suppl):L227–L233
13. Sayegh R, Auerbach SD, Li X, et al. Glucocorticoid induction of epithe-lial sodium channel expression in lung and renal epithelia occurs via trans-activation of a hormone response element in the 5⬘-flanking re-gion of the human epithelial sodium channel alpha subunit gene.J Biol Chem.1999;274:12431–12437
14. Thomas CP, Auerbach SD, Zhang C, Stokes JB. The structure of the rat amiloride-sensitive epithelial sodium channel gamma subunit gene and functional analysis of its promoter.Gene.1999;228:111–122
16. Pitkanen OM, Smith D, O’Brodovich H, Otulakowski G. Expression of alpha-, beta-, and gamma-hENaC mRNA in the human nasal, bronchial, and distal lung epithelium.Am J Respir Crit Care Med.2001;163:273–276 17. Knowles M, Gatzy J, Boucher R. Increased bioelectric potential differ-ence across respiratory epithelia in cystic fibrosis.N Engl J Med.1981; 305:1489 –1495
18. Jorissen M, Bertrand B, Eloy P. Ciliary dyskinesia in the nose and paranasal sinuses.Acta Otorhinolaryngol Belg.1997;51:353–366 19. Knowles MR, Carson JL, Collier AM, Gatzy JT, Boucher RC.
Measure-ments of nasal transepithelial electric potential differences in normal human subjects in vivo.Am Rev Respir Dis.1981;124:484 – 490 20. Otulakowski G, Flueckiger-Staub S, Ellis L, et al. Relation between
alpha, beta, and gamma human amiloride-sensitive epithelial Na⫹
channel mRNA levels and nasal epithelial potential difference in healthy men.Am J Respir Crit Care Med.1998;158:1213–1220
21. Avery M, Mead J. Surface properties in relation to atelectasis and hyaline membrane disease.Am J Dis Child.1959;97:517–523
22. Jobe AH. Pulmonary surfactant therapy.N Engl J Med.1993;328:861– 868 23. O’Brodovich HM. Immature epithelial Na⫹channel expression is one of the pathogenetic mechanisms leading to human neonatal respiratory distress syndrome.Proc Assoc Am Physicians.1996;108:345–355 24. DeSa DJ. Pulmonary fluid content in infants with respiratory distress.
J Pathol.1969;97:469 – 478
25. Tchepichev S, Ueda J, Canessa C, Rossier BC, O’Brodovich H. Lung epithelial Na channel subunits are differentially regulated during de-velopment and by steroids.Am J Physiol.1995;269(suppl):C805–C812 26. O’Brodovich H, Canessa C, Ueda J, Rafii B, Rossier BC, Edelson J.
Expression of the epithelial Na⫹channel in the developing rat lung. Am J Physiol.1993;265(suppl):C491–C496
27. Dagenais A, Kothary R, Berthiaume Y. The alpha subunit of the epithe-lial sodium channel in the mouse: developmental regulation of its expression.Pediatr Res.1997;42:327–334
28. McDonald FJ, Yang B, Hrstka RF, et al. Disruption of the beta subunit of the epithelial Na⫹channel in mice: hyperkalemia and neonatal death associated with a pseudohypoaldosteronism phenotype.Proc Natl Acad Sci U S A.1999;96:1727–1731
29. Bonny O, Rossier BC. Disturbances of Na/K balance: pseudohypoaldo-steronism revisited.J Am Soc Nephrol.2002;13:2399 –2414
30. Bonny O, Chraibi A, Loffing J, et al. Functional expression of a pseudohypoaldosteronism type I mutated epithelial Na⫹channel lack-ing the pore-formlack-ing region of its alpha subunit.J Clin Invest.1999;104: 967–974
31. Kerem E, Bistritzer T, Hanukoglu A, et al. Pulmonary epithelial sodium-channel dysfunction and excess airway liquid in pseudohypoaldoste-ronism.N Engl J Med.1999;341:156 –162
32. Thomas CP, Zhou J, Liu KZ, Mick VE, MacLaughlin E, Knowles M. Systemic pseudohypoaldosteronism from deletion of the promoter
re-gion of the human beta epithelial na(⫹) channel subunit.Am J Respir Cell Mol Biol.2002;27:314 –319
33. McNicholas CM, Canessa CM. Diversity of channels generated by dif-ferent combinations of epithelial sodium channel subunits.J Gen Physiol. 1997;109:681– 692
34. Konstas AA, Korbmacher C. The gamma-subunit of ENaC is more important for channel surface expression than the beta-subunit.Am J Physiol Cell Physiol.2003;284:C447–C456
35. Smith DE, Otulakowski G, Yeger H, Post M, Cutz E, O’Brodovich HM. Epithelial Na(⫹) channel (ENaC) expression in the developing normal and abnormal human perinatal lung.Am J Respir Crit Care Med.2000; 161(suppl):1322–1331
36. Gowen CW Jr, Lawson EE, Gingras J, Boucher RC, Gatzy JT, Knowles MR. Electrical potential difference and ion transport across nasal epi-thelium of term neonates: correlation with mode of delivery, transient tachypnea of the newborn, and respiratory rate. J Pediatr. 1988; 113(suppl):121–127
37. Kassovska-Bratinova S, Vannam V, Freywald T, Otulakowski G, O’Brodovich H. Absence of detectable amiloride-sensitive ion transport in murine tracheal epithelia may result from lack of beta-ENaC mRNA expression.Am J Respir Crit Care Med.2000;161:A234
38. Firsov D, Gautschi I, Merillat AM, Rossier BC, Schild L. The heterotet-rameric architecture of the epithelial sodium channel (ENaC).EMBO J. 1998;17:344 –352
39. Wright LL, Horbar JD, Gunkel H, et al. Evidence from multicenter networks on the current use and effectiveness of antenatal corticoste-roids in low birth weight infants.Am J Obstet Gynecol.1995;173:263–269 40. Gladstone IM, Ehrenkranz RA, Jacobs HC. Pulmonary function tests and fluid balance in neonates with chronic lung disease during dexa-methasone treatment.Pediatrics.1989;84:1072–1076
41. Bancalari E. Corticosteroids and neonatal chronic lung disease.Eur J Pediatr.1998;157(suppl 1):S31–S37
42. Folkesson HG, Norlin A, Wang Y, Abedinpour P, Matthay MA. Dexa-methasone and thyroid hormone pretreatment upregulate alveolar ep-ithelial fluid clearance in adult rats.J Appl Physiol.2000;88:416 – 424 43. Talbot CL, Bosworth DG, Briley EL, et al. Quantitation and localization
of ENaC subunit expression in fetal, newborn, and adult mouse lung. Am J Respir Cell Mol Biol.1999;20:398 – 406
44. Rafii B, Tanswell AK, Pitkanen O, O’Brodovich H. Induction of epithe-lial sodium channel (ENaC) expression and sodium transport in distal lung epithelia by oxygen.Curr Top Membr.1999;47:239 –244
45. Minakata Y, Suzuki S, Grygorczyk C, Dagenais A, Berthiaume Y. Impact of beta-adrenergic agonist on Na⫹channel and Na⫹-K⫹-ATPase ex-pression in alveolar type II cells. Am J Physiol. 1998;275(suppl): L414 –L422
SETBACK FOR HOSPITALS
“The medical education establishment suffered a setback when a federal judge refused to dismiss a lawsuit contending that medical residents are forced to participate in a system that ensures that they work long hours for low pay. The judge . . . rejected pleas by medical schools and teaching hospitals that he dismiss the suit. It argues that a system known as the match, which pairs medical school graduates with hospitals for training, is a violation of antitrust laws.”
Lewis NA.New York Times. February 12, 2004
DOI: 10.1542/peds.113.5.1267
2004;113;1267
Pediatrics
Kirjavainen and Gail Otulakowski
Otto Helve, Olli M. Pitkänen, Sture Andersson, Hugh O'Brodovich, Turkka
Preterm Infants With Respiratory Distress
Low Expression of Human Epithelial Sodium Channel in Airway Epithelium of
Services
Updated Information &
http://pediatrics.aappublications.org/content/113/5/1267
including high resolution figures, can be found at:
References
http://pediatrics.aappublications.org/content/113/5/1267#BIBL
This article cites 45 articles, 6 of which you can access for free at:
Subspecialty Collections
http://www.aappublications.org/cgi/collection/pulmonology_sub
Pulmonology
sub
http://www.aappublications.org/cgi/collection/fetus:newborn_infant_
Fetus/Newborn Infant
following collection(s):
This article, along with others on similar topics, appears in the
Permissions & Licensing
http://www.aappublications.org/site/misc/Permissions.xhtml
in its entirety can be found online at:
Information about reproducing this article in parts (figures, tables) or
Reprints
http://www.aappublications.org/site/misc/reprints.xhtml
DOI: 10.1542/peds.113.5.1267
2004;113;1267
Pediatrics
Kirjavainen and Gail Otulakowski
Otto Helve, Olli M. Pitkänen, Sture Andersson, Hugh O'Brodovich, Turkka
Preterm Infants With Respiratory Distress
Low Expression of Human Epithelial Sodium Channel in Airway Epithelium of
http://pediatrics.aappublications.org/content/113/5/1267
located on the World Wide Web at:
The online version of this article, along with updated information and services, is
by the American Academy of Pediatrics. All rights reserved. Print ISSN: 1073-0397.