An analysis of bimanual actions in natural feeding of semi wild chimpanzees
Full text
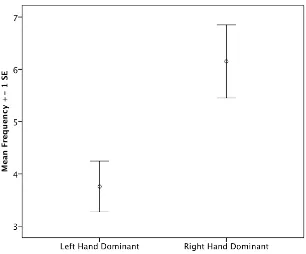
Figure

Related documents
tnia end, tissue explant.a from the bulb scale leaves of... behaviour
The EC-US negotiations on agricultural trade liberalization in the Uruguay Round of GATT (General Agreement on Tariffs and Trade) provide a good illustration of the impact
The increase in the amount of light seed harvested from inoculated pods is evidence for a direct effect of siliqua infection on the seed, but the yield
This paper is an extension of a conference paper (Stability analysis of single-phase grid-feeding inverters with PLL using Harmonic Linearisation and Linear Time Periodic (LTP)
jaatao.p`%yaokanao sadacarNaacaa maaga- AvalaMbalaa paihjao.baaOd\Qa Qamaa-cyaa p`saar –p`caaracyaa ]WoSaanao maz ivaharatUna iSaxaNaacaI vyavasqaa kolaI.saMGasqaapnaa k$na
The disabilities for which manpower development programme are conducted in different categories ranging from certificate to Master Degree in Regular, Distance and
Our analyses of nucleolar domi- mosome 3 QTL is inherited from Cvi, differences in the nance in Cvi/Ler RI lines favor the latter hypothesis in relative expression of NOR4 and NOR2
It is remarkable that the expression of GFP has no obvious negative influence on cell growth or function which allows monitoring of gene expression and protein localization in