The neural circuitry that controls movements of animal limbs generally contains both central driving elements and peripheral sensors providing feedback to the system. Insect legs are driven by motor neurones contained within each segmental ganglion. The actions of these motor neurones are coordinated by groups of local or intersegmental interneurones, some of which must comprise the rhythm-generating circuits that underlie repetitive movements such as walking. Each leg contains many proprioceptors that respond to different mechanical aspects of its activity and which feed back onto the motor neurones and interneurones controlling it. For example, the position, velocity and acceleration of the femoro-tibial joint are registered by the femoral chordotonal organ (locust, Zill, 1985; Matheson, 1990, 1992a,b; stick insect, Hofmann et al. 1985; Hofmann and Koch, 1985). A single strand receptor associated with the metathoracic femoral chordotonal organ responds to tibial extension (Bräunig, 1985). Campaniform sensilla on the proximal tibia monitor cuticular distortions that
result from resisted contractions of the extensor tibiae muscle (Burrows and Pflüger, 1988), and superficially placed multipolar receptors (Coillot and Boistel, 1968, 1969; Williamson and Burns, 1978; Bässler, 1977) respond to movements of the femoro-tibial joint. The prothoracic and mesothoracic legs of the locust contain another multipolar receptor, which is sensitive to tension generated by the main flexor tibiae muscle. It may act as a sensor in a load compensation circuit (Theophilidis and Burns, 1979).
The metathoracic leg of the locust is specialised for kicking and jumping (e.g. Heitler, 1974; Bennet-Clark, 1975), although it is also used for walking. Burns (1974) suggested that this specialisation of function was a reason why the metathoracic femoral chordotonal organ apparently contained only one of the two groups of homologous sensilla present in the other legs. Furthermore, the flexor tension receptor present in the prothoracic and mesothoracic legs has not been observed in the metathoracic leg (Theophilidis and Burns, 1979). We have The tibia of each leg of the locust is moved by two
antagonistic muscles, the extensor and flexor tibiae. A variety of sense organs on and in each leg provide feedback about this joint’s position and movement and about forces acting on the exoskeleton and muscles. One such organ is a muscle tension receptor found within the flexor tibiae muscle of the mesothoracic leg. We now show that an apparently homologous multipolar receptor is present in the hind leg, but that here it is associated with a specialised flexor muscle, the accessory flexor. This muscle comprises 13 fibres, innervated by five of the thirteen motor neurones that innervate the main flexor muscle and, since these are slow motor units, the response properties of the receptor are constrained. The multipolar receptor attaches to the muscle fibres near their proximal insertion onto the femoral cuticle. It generally has four primary dendrites, which do not branch extensively within the muscle.
We show that the receptor responds strongly to active, isometric contractions but only poorly to imposed changes
of accessory flexor muscle length (i.e. passive changes in tibial position). It does not respond to tension generated by the main flexor muscle or by the extensor muscle. The tension receptor causes short-latency (0.9–1.8 ms) excitatory inputs onto the three common inhibitory motor neurones and longer-latency (3.7–8.1 ms) inhibitory inputs onto the slow extensor tibiae motor neurone. In quiescent animals, it causes excitatory inputs onto flexor tibiae motor neurones (2.2–3.8 ms) but, in more active animals, its inputs onto these neurones are often inhibitory, with delays of 6–10 ms.
The slow nature of the accessory flexor muscle and the pattern of central connections of the receptor suggest that together they are involved in the control of slow movements or posture, potentially acting through a servomechanism.
Key words: locust, Schistocerca gregaria, Locusta migratoria, muscle tension receptor, accessory flexor tibiae, multipolar neurone, muscle spindle.
Summary
AN ELABORATE TENSION RECEPTOR SYSTEM HIGHLIGHTS SENSORY
COMPLEXITY IN THE HIND LEG OF THE LOCUST
THOMAS MATHESON1 ANDLAURENCE H. FIELD2
1Department of Zoology, University of Cambridge, Downing Street, Cambridge CB2 3EJ, England and
2Department of Zoology, University of Canterbury, Private Bag 4800, Christchurch, New Zealand
Accepted 5 April 1995
recently shown that the metathoracic femoral chordotonal organ does possess both groups of sensilla present in the other legs (Matheson and Field, 1990). In the present paper, we describe a multipolar receptor neurone that responds to tension generated by the accessory flexor tibiae muscle of the metathoracic leg. This receptor therefore appears to be homologous to that found in the flexor tibiae muscle of the other legs, providing further evidence that the functional specialisation of the metathoracic legs has not been accompanied by a marked reduction in their sensory supply.
We show that the accessory flexor tibiae muscle, which is absent in the prothoracic and mesothoracic legs (Heitler, 1974; Theophilidis and Burns, 1979), is innervated by only two of the nine excitatory flexor motor neurones, by two common inhibitory motor neurones and, probably, by one dorsal unpaired median (DUM) neurone. At least seven other flexor tibiae motor neurones do not innervate the accessory flexor muscle. This means that the animal has partially independent control over the amount of force generated in the two flexor muscles. The receptor responds only to the force generated by the few fibres of the accessory flexor and not to force generated by the ‘main’ flexor muscle. The tension receptor is somewhat similar to arthropod muscle receptor organs (MROs). For example, the MRO at the coxa–trochanter joint of the locust leg consists of a single multipolar receptor neurone associated with a group of three muscle fibres that lie parallel to, but are innervated independently of, the trochanteral levator muscle (Bräunig and Hustert, 1985a; Bräunig et al. 1986).
The partially independent control of the accessory flexor muscle suggests that, of the nine known excitatory flexor tibiae motor neurones, those that innervate the accessory flexor muscle could act somewhat similarly to b-motor neurones of vertebrate muscle spindles, which innervate both extrafusal and intrafusal muscle fibres. The muscle spindles of amphibians, like the locust accessory flexor muscle, receive only such shared (b)motor innervation.
Materials and methods
Adult locusts were taken from crowded cultures in Cambridge, England (Schistocerca gregaria Forskål), or Christchurch, New Zealand (Locusta migratoria L.). All electrophysiological experiments were performed on S. gregaria. Morphological observations were carried out on both species.
Morphological observations Cobalt backfills
The tension receptor was stained through its axon in n5B1 using conventional backfilling techniques (5 % cobalt chloride, 3–7 days at 4 ˚C). The preparation was fixed in Carnoy’s fixative, cleared in methyl salicylate and viewed using an interference contrast microscope (Leitz).
Electron microscopy
For electron microscopy, tissue was fixed in 2.5 %
glutaraldehyde buffered with 0.1 mol l21 sodium cacodylate (pH 7.4), for 3 h at 4 ˚C. It was then rinsed in the buffer for 1 h, post-fixed in 1 % osmium tetroxide (buffered as above) for 3 h, rinsed again (all at 4 ˚C), and then dehydrated in an ethanol series at room temperature. The dehydrated tissue was transferred to propylene oxide, embedded in Spurr’s epoxy resin and sectioned on an LKB 8800 ultrotome. Sections were stained in 3 % uranyl acetate followed by lead citrate, and viewed on a Jeol 1200EX transmission electron microscope (TEM).
Electrophysiology Extracellular recordings
Animals were fixed ventral side up in Plasticine. The left hind leg was held at right angles to the body and rotated posteriorly so that its anterior surface faced upwards. Nerves and muscles were exposed where necessary by cutting small windows in the anterior femoral cuticle. Sensory and motor activity was recorded from appropriate nerves using 50mm diameter silver wire hook electrodes. Muscle activity was recorded using 50mm diameter silver wires inserted through the cuticle into the relevant muscle. Nerves and muscles could be stimulated by passing current through the recording electrodes.
Force recordings
Forces produced by nearly isometric contractions of the tibial muscles were measured using pairs of foil strain gauges
mounted on stiff steel probes. Each probe was calibrated with a series of known masses spanning the range of forces to be measured. The relationship between voltage and force for each
B C
D A
n3B5c
n3B5c 136c ext apo
TR
TR
SR
fs n5B1
n5B1
n5B1e
n5B1
n5B1f flex
apo
vlig
Heitler’s lump
dlig FCO
FCO acc flex us
FCO
apo n5B1e2
n5B1e1
n5B1e2
Femur
1 mm
420µm
Tibia
probe was linear in the range used. To measure the overall force produced by all the muscles acting at the femoro-tibial joint, a probe was waxed directly onto the tibia of the leg. To measure the force produced by the accessory flexor muscle alone, a pair of custom-made forceps was attached to a probe so that it could grasp the flexor apodeme (on which the accessory flexor inserts). The apodeme was then cut proximal and distal to the forceps, leaving only fibres of the accessory flexor muscle attached. To produce controlled movements of the muscle apodeme, the probe was attached to a Ling vibrator controlled by a custom-built ramp generator (Matheson and Ditz, 1991). The relationship between the linear position of the apodeme and the femoro-tibial angle was determined in five animals (not shown, see also Field and Burrows, 1982). This relationship allowed us to express all apodeme movements as equivalent changes in the femoro-tibial angle.
Intracellular recordings and motor innervation
The mesothoracic and metathoracic ganglia were exposed by cutting a window in the ventral thoracic cuticle. Some overlying air sacs were removed, and the ganglia were supported on a wax-coated silver platform. To facilitate penetration by microelectrodes, the sheath of the metathoracic ganglion was treated with a 1 % (w/v) solution of protease (Sigma type XIV). Electrodes had resistances of approximately 60 MVwhen filled with 1 mol l21potassium acetate.
Recordings were made from the somata of identified tibial motor neurones and midline spiking interneurones in the metathoracic ganglion. The extensor muscle is innervated by two excitatory motor neurones, a common inhibitor motor neurone (CI1) and a dorsal unpaired medial neurone (DUMETi) (Hoyle, 1978). The flexor muscle is innervated by two common inhibitors (CI2, CI3; Hale and Burrows, 1985) and at least nine excitatory motor neurones (Phillips, 1981) divided into three groups: anterior, posterior and lateral. Each group contains a fast, a slow and an intermediate neurone (Hoyle and Burrows, 1973).
The motor neurones were identified by the positions of their somata and by the following additional features. The fast extensor tibiae motor neurone (FETi) was identified by its antidromic spike in response to electrical stimulation of the extensor muscle. The slow extensor (SETi) was identified by a 1:1 correlation of its spikes with potentials recorded in the extensor muscle. Flexor tibiae motor neurones were identified by their characteristic depolarisations following FETi spikes (Burrows et al. 1989). The type of flexor motor neurone (fast, intermediate or slow) was determined by simultaneously or sequentially recording from all three flexors in the particular group (see Parker, 1993). CI1 was identified by a 1:1 correlation of its spikes with potentials recorded in the extensor nerve. CI2and CI3were identified by 1:1 correlations between their spikes and potentials recorded in the flexor nerve (n5B2).
Patterns of motor innervation were investigated by (a) intracellular stimulation of flexor tibiae motor neurones in the metathoracic ganglion, and (b) stimulation of n5B2 with
gradually increasing voltage while recording intracellularly from accessory flexor muscle fibres.
To detect the central connections made by the tension receptor, its spikes recorded from n5B1 in mid femur triggered a signal averager that was used to detect postsynaptic potentials in central neurones. A simultaneous recording of the tension receptor spike in n5 near the ganglion was used to calculate both the afferent conduction velocity and, knowing the relevant distances between recording sites, the unexplained central delay (i.e. the period left unaccounted for by the estimated time taken for the afferent spike to travel from n5 to the intracellular recording site).
Throughout this paper, values are presented as mean ±S.E.M. unless stated otherwise.
Results
Morphology of the receptor
The tension receptor (TR) is a large multiterminal sensory neurone (type II; Zawarzin, 1912) with a mean soma diameter of 34mm (range 26–41mm, N=5). It attaches near the proximal origin of the anterior accessory flexor muscle (136c), with its dendrites oriented towards the origin of the muscle fibres on the cuticle (Fig. 1A). It has 1–4 primary dendrites, which range in length from 97 to 240mm (N=16 dendrites in seven neurones) and generally lack secondary branches (Fig. 1D). They intermingle with fibres of the accessory flexor muscle (Fig. 1C), but do not obviously extend as far as the origins of the muscle fibres on the cuticle. The soma gives rise to an axon which,
together with an axon from a hair sensillum on the anterior dorsal femur, forms a tiny short nerve extending directly towards the junction of n5B1 and n5B1e, with which it merges.
Ultrastructure and innervation of the accessory flexor muscle The accessory flexor muscle comprises two isolated distal bundles of fibres inserting onto the anterior and posterior
ext apo
n5B1 FCO n5B2
acc flex
acc flex n flex
flex apo 300µm
B
A
C
D
E
F
m
m
m
m
surfaces of the flexor tibiae muscle apodeme. Because these are slightly separated from the main flexor fibres (M136a) and have more dorsal origins on the anterior and posterior surfaces of the femur, they were designated by Snodgrass (1929) as separate muscles (M136b,c). Only the anterior accessory flexor (M136c, Fig. 2A) is associated with a multipolar receptor neurone. This muscle, which contains 13 fibres (N=5), is innervated at four points by a distal femoral branch of n5B2 (and not n5B1, which carries the tension receptor axon), which also has branches into several distal fibres of the main flexor (Fig. 2A,B). Electron micrographs of transverse sections of the accessory flexor motor branches showed a maximum of five axons (Fig. 2C), although most of the branches amongst (e.g. arrowheads in Fig. 2B) or apposed to the muscle fibres (Fig. 2E) had only three axons. The neuromuscular synapses (not shown) contained electron-lucent synaptic vesicles.
The muscle fibres are characterised by relatively sparse sarcoplasmic reticulum and a high thin:thick filament ratio of 7:1 (Fig. 2E,F; cf. Hoyle, 1978). The sarcoplasmic reticulum was usually associated with transverse tubules as dyads, but it did not segregate the contractile proteins into discrete bundles (‘fibrils’, Elder, 1975) typical of insect phasic muscle. Sarcomere length was 8.7±0.6mm (S.D., N=191), which is typical of insect slow tonic muscle (Elder, 1975; Hoyle, 1975).
Activity of the receptor neurone
Hook electrodes placed on sensory nerve 5B1 in mid-femur record the activity of many afferent neurones in the leg, including those of the femoral chordotonal organ. When all branches of n5B1 distal to the accessory flexor muscle, including that to the femoral chordotonal organ, were ablated, a single tonic spike remained. Cutting n5B1 proximal to the accessory flexor muscle or crushing the proximal ends of the muscle fibres (where the tension receptor cell body lies, see Fig. 1) abolished this tonic activity. We therefore conclude that the tonic spikes in a single axon come from the sensory neurone in the accessory flexor muscle.
With the femoro-tibial angle set at approximately 90 ˚, the spike frequency of the receptor in quiescent animals ranged from 6 to 25 Hz (12.5±1.6 Hz, N=14). The firing rate was uniform, with few bursts or doublets of spikes. The conduction velocity, measured between hook electrodes in mid-femur and near to the metathoracic ganglion, ranged from 0.9 to 1.5 m s21 (1.1±0.03 m s21, N=27).
Responses of the tension receptor to passive stretch and active tension
To determine whether the tension receptor responds primarily to isometric tension or to movement, the accessory flexor muscle was activated in two ways. First, it was induced to contract actively by tickling the animal with a soft paintbrush or by injecting current into particular flexor motor neurones so that they spiked. Second, it was passively stretched by extending the tibia or pulling on the flexor apodeme.
Voluntary isometric contractions caused the TR firing rate to increase (Fig. 3Ai,ii, Bi). More powerful flexions were
accompanied by larger changes in firing frequency (Fig. 3Ai,ii), up to a maximum recorded of 175 Hz. High frequencies of receptor firing were maintained throughout periods of isometric contraction lasting several seconds. When the motor nerves to the accessory flexor muscle were ablated, leaving the motor innervation to the main flexor muscle intact, active isometric flexions were not accompanied by increases in the TR firing rate (compare Fig. 3Bi and Bii), indicating that it does not respond to the tension generated by the main flexor muscle. Under the same conditions, active isometric extensions also had no effect on the TR firing rate (Fig. 3Biii).
In contrast to the above, passive extension and flexion movements had little or no effect on the tonic firing rate of the TR (Fig. 3Aiii,iv, C). The tonic firing frequency was lowest when the tibia was nearly fully flexed (10 ˚), increased by 53 % (from 11±2 Hz to 17±3 Hz, N=4) when the tibia was fully extended (160 ˚) and, on return to 10 ˚, showed little hysteresis. A similar increase was seen in two preparations in which the motor innervation to the accessory flexor muscle had been cut,
although the firing rates in these preparations were 12 % lower than those for the intact preparations. The maximum tonic firing rate measured during passive stretch was therefore only
11 % that of the maximum rate recorded during active isometric contractions.
The relationship between passive tension in the accessory
Bi Bii Biii
Ei Eii Eiii
C
Di Dii
Ai
Aii
Aiii
Aiv
25
20
15
10
5
0 0.2 g
70
50
30
100 80 60 40 20 0
Flexion
Extension
150 mg (B) 30 mg (D) 80 ° (D,E) Stretch
Force Receptor
Stretch Receptor
Firing frequency (Hz)
Firing frequency (Hz)
Receptor Active force
Active force
Passive extension
Passive flexion
40
30
20
0 40
1 s
70 90
50
30
Firing frequency (Hz)
1 s
80 120 160
0 80 160
10 10 Max
Femur–tibia angle (degrees)
Denervated
Intact Ablated
Innervated
flexor muscle and TR firing rate was examined in more detail by isolating the accessory flexor muscle from the main flexor muscle. During slow (66 ˚ s21) stretch and release movements from the equivalent of 80 ˚ to 160 ˚, the TR firing rate closely paralleled the changes in passive force (Fig. 3Di). During faster (267 ˚ s21) stretches, the TR responded with a marked, but very transient, increase in firing rate (Fig. 3Dii). During these faster movements, however, the forceps tended to vibrate on the springy strain gauge. Experiments without the strain gauge yielded similar results, indicating that vibrations did not contribute to the observed response (Fig. 3Ei–iii). In fact, the lack of response to the damped oscillation (e.g. Fig. 3Dii) shows that the tension receptor is relatively insensitive to imposed vibration. Instantaneous firing frequency reached 90 Hz during a movement at 533 ˚ s21.
Motor innervation of the accessory flexor muscle Two slow excitatory flexor motor neurones (those from the anterior and lateral groups) innervate the accessory flexor muscle. They also innervate the main flexor muscle. In some, but not all, recordings they were shown to receive input from the tension receptor (see below). The accessory flexor tibiae muscle is innervated by both of the common inhibitory motor neurones that also innervate the main flexor muscle (CI2, CI3). They receive excitatory inputs from the tension receptor (see below).
Stimulation of the motor nerve (not shown) suggests that different accessory flexor muscle fibres are innervated by different numbers of motor neurones. The following combinations were recorded: two excitors and one inhibitor; two excitors and two inhibitors; two excitors; one excitor. The most frequent pattern (two excitors and one inhibitor) corresponds with the commonest innervation pattern (three axons) observed in TEM sections of the accessory flexor muscle (Fig. 2D).
Responses of the accessory flexor muscle and tension receptor to stimulation of motor neurones
During supramaximal stimulation of the motor nerve (n5B2), tension recordings of the semi-isolated accessory flexor muscle showed contraction profiles characteristic of slow muscle. For stimulation frequencies in the range 10–50 Hz, tension slowly increased (e.g. 0.02 g per 2 s at 10 Hz) in proportion to stimulus frequency without reaching a plateau (Fig. 4A–D); the minute individual twitches showed marked facilitation (200 % after 20 twitches, Fig. 5A). Furthermore, single twitches had a slow relaxation phase. The same stimulus conditions delivered to the main flexor muscle gave rapid twitch contractions, which reached a plateau within five twitches (Fig. 4F,G) and showed barely detectable facilitation (18 % after 20 twitches, Fig. 4E). Single twitches relaxed to baseline in 33–50 % of the observed relaxation time of single accessory flexor twitches. These results corroborate the finding that slow motor neurones innervate the accessory flexor muscle.
The slow flexor motor neurone from the anterior group,
which innervated the accessory flexor muscle, fired rapidly during struggling, and the tension receptor firing rate increased (Fig. 5A). Leg movement was blocked. The change in TR firing was only apparent at the higher frequencies of motor neurone activity, suggesting that a threshold level of muscle activation had to be exceeded before the receptor was affected. Depolarising this flexor motor neurone to increase its firing frequency also caused an increase in the TR firing rate (Fig. 5B). Hyperpolarisation of the flexor motor neurone to suppress its tonic firing did not reduce the TR firing rate below the resting level.
The relationship between the firing frequency of another flexor motor neurone (controlled by injection of current) and the TR firing rate is shown in Fig. 5C. At normal resting potential this motor neurone fired at approximately 26 Hz. In
B C D
F G
E A
AF, 10 Hz AF,
10 Hz
AF, 20 Hz
Flexor, 2 Hz
Flexor, 10 Hz
Flexor, 20 Hz AF, 2 Hz
this case also, there was a threshold rate of motor neurone activity (between 10 and 16.5 Hz) below which no further alteration in TR firing rate occurred. The tension receptor did not generally respond markedly to individual motor spikes. In one case, however, each excitatory muscle potential elicited a marked transient increase in the TR firing rate (Fig. 5E).
Intracellular recordings from the accessory flexor muscle showed that, when the muscle received a barrage of hyperpolarising potentials from one of the common inhibitor motor neurones, the TR firing rate was reduced (Fig. 5D, arrow), although the overall flexor force did not change. Individual inhibitory inputs from CI2had little or no apparent effect on the TR firing rate (Fig. 5E). In Fig. 5D, the initial burst of large spikes recorded in the main flexor myogram was coincident with a marked increase in total force, but this was not reflected in either the muscle fibre recording or the tension receptor firing rate. The discrepancy between the pattern of tibial force and the pattern of tension receptor firing (which should reflect accessory flexor tension) indicates that the accessory flexor can, indeed, be activated at least partially independently of the main flexor. We cannot rule out the further possibility that the receptor responds to the force generated by only some of the 13 muscle fibres of the accessory flexor muscle. The wide spread of its dendrites suggests that it contacts most of the fibres.
Central connections of the tension receptor Inhibitory motor neurones
All three common inhibitory neurones received depolarising inputs from the tension receptor (Fig. 6A–C), with central latencies of 0.9–1.8 ms (mean 1.4±0.1 ms, N=8; see Fig. 8C). The input to CI1was clear in only two of six recordings from this cell.
Excitatory motor neurones
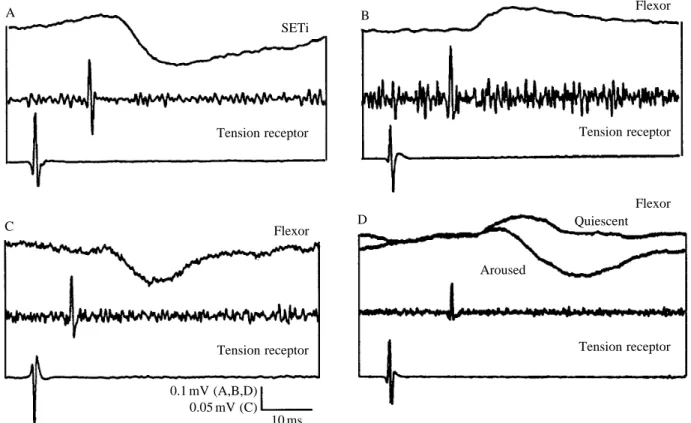
Extensor tibiae. The extensor tibiae muscle of the locust hind leg is innervated by one slow motor neurone (SETi) and one fast motor neurone (FETi). Tension receptor spikes had no effect on FETi, but elicited hyperpolarising potentials in SETi, with a central delay of 3.7–8.1 ms (Figs 7A, 8C). It is unlikely, therefore, that this effect was monosynaptic. The inhibitory nature of the PSP is confirmed in Fig. 9A, which shows that an increase in TR firing frequency was followed by a reduction in SETi firing frequency. Background synaptic inputs to SETi made it impossible to determine whether every TR spike was followed by an IPSP, but signal averaging always revealed the same inhibitory effect (N>12).
Flexor tibiae. In quiescent animals, the tension receptor had excitatory effects in 17 recordings of flexor motor neurones (Fig. 7B), inhibitory effects in 5 (Fig. 7C), no effects in 40, and unclear effects in 3 (N=65). A depolarising input from the TR was most commonly recorded in slow (42 %), less often in intermediate (23 %) and least often in fast (12 %) flexor motor neurones. There was no evidence for a different occurrence of TR depolarising input to flexor motor neurones in the three
different anatomical groups (anterior, 30 %; posterior, 25 %; lateral, 27 %). The central delay for the depolarising potentials ranged from 2.2 to 3.8 ms (mean 3.0±0.1 ms, N=16), suggesting a polysynaptic pathway.
In active animals, these depolarising inputs were sometimes replaced by longer-latency hyperpolarising or mixed inputs (6–10 ms; mean 8.0±0.6 ms, N=6; Figs 7D, 8C). This difference in timing between the initial inflections of depolarising and hyperpolarising potentials is evident in Fig. 7D.
Interneurones
Five recordings of spiking interneurones with somata near the ventral midline of the metathoracic ganglion revealed depolarising inputs from the tension receptor (Fig. 8A). Another neurone, which could not be induced to spike with depolarising current, and whose soma was near to, but clearly not that of, CI3, also received a depolarising input (Fig. 8B). The central latencies of these inputs ranged from 1.5 to 2.5 ms (mean 2.0±0.2 ms, N=6; see Fig. 8C).
Tension receptor activity enhances tibial flexion Electrically stimulating the accessory flexor muscle caused the muscle to contract and the tension receptor firing rate to increase (Fig. 9). This caused the tonic firing of SETi to slow down or stop (Fig. 9A). In a different preparation, similar electrical stimulation led to a burst of large depolarising potentials in an anterior flexor tibiae motor neurone (Fig. 9B). The potentials in the flexor motor neurone did not match the pattern of spikes in the tension receptor, indicating the contribution of an indirect pathway. These results suggest that, in quiescent animals, activation of the tension receptor enhances tibial flexion.
Discussion
Theophilidis and Burns, 1979). It differs in that (a) its primary dendrites generally do not branch, and (b) it is in a specialised muscle, the accessory flexor.
Structure and innervation of the accessory flexor muscle The ultrastructure and physiological characteristics of the
accessory flexor muscle strongly suggest that it is of the slowly contracting, tonic type. The corresponding antagonist (M135c, accessory extensor) that attaches to the extensor muscle apodeme has essentially the same ultrastructure and thin:thick filament ratio (7:1, Hoyle, 1978). This muscle was shown by Hoyle (1978) to consist only of slow muscle fibres which
B
C
D A
35
25
15 Tension receptor
Flexor
SETi
Extensor myogram
Tension receptor
Accessory flexor
Anterior flexor
Current
TR firing frequency (Hz)
Anterior flexor Tension receptor
Accessory flexor
Flexor myogram
Force
Tension receptor
0 10 20 30
Flexor firing frequency (Hz)
+2 nA
0nA
−2 nA
E
Accessory flexor CI2
Tension receptor firing frequency
(Hz)
20
15
10
display slowly rising contraction profiles with a slope proportional to stimulation frequency (e.g. 2.1 g per 10 s during 10 Hz stimulation of its single excitatory axon, SETi). Single twitches showed the same relaxation profiles as those observed here in single twitches of the accessory flexor muscle. The slow, tonic nature of the accessory flexor muscle must set constraints on its rate of tension development and hence on the range of afferent impulse frequencies developed in the tension receptor. The conclusion is that the system is operationally designed for the control of postural or slow to medium rates of limb movement, rather than fast phasic movements.
Although the commonest pattern of motor innervation recorded electrically from the accessory flexor muscle was three units (two slow excitors and one inhibitor), up to four motor axons were detected in this way, and up to five axons were seen in electron micrographs (Fig. 2C). It is probable that the full complement of efferent axons includes two slow flexor tibiae motor neurones (from the anterior and lateral groups), two common inhibitors (CI2 and CI3) and one dorsal unpaired median (DUM) neurone. The common inhibitory neurones in the locust are known to innervate slow muscle fibres exclusively. In tettigoniids, the accessory flexor muscle is innervated by 13 motor axons, but the presence of associated mechanoreceptors has not been reported (Theophilidis and Dimitriadis, 1990).
Physiological responses of the tension receptor The metathoracic tension receptor, like those in the prothoracic and mesothoracic legs of the locust (Theophilidis and Burns, 1979), and in the legs of the stick insect (Bässler,
1977), spikes tonically at rest. Passive stretch of the muscle into which the receptor inserts causes the receptor’s tonic firing rate to increase slightly. In the metathoracic leg of the locust, the relationship between passive accessory flexor muscle length and receptor firing rate is approximately linear, but in the prothoracic and mesothoracic legs it is not (Theophilidis and Burns, 1979). In the mesothoracic leg, it has been suggested that this non-linear relationship is caused by a corresponding non-linearity in the relationship between muscle length and tension (Theophilidis and Burns, 1979). The values of muscle length given by Theophilidis and Burns are for the entire flexor muscle, however, not for the fibres into which the
Fig. 5. (A) During struggling movements the TR firing rate (top trace) increased when an anterior flexor motor neurone that innervated the accessory flexor muscle was depolarised and fired strongly (second trace). When the slow extensor motor neurone (SETi) received depolarising inputs and fired (third and bottom traces) the TR firing rate remained unchanged. (B) When the firing rate of the flexor motor neurone (third trace), which innervated the accessory flexor muscle (second trace), was increased by injection of depolarising current (bottom trace), the muscle contracted (not shown) and the TR firing rate increased (top trace). During injection of depolarising current, the TR firing rate (0.2 s bins) closely followed the flexor motor neurone firing rate (inset). The relationship between flexor motor neurone firing rate and tension receptor firing rate is further illustrated for another flexor motor neurone in C. Values are means ±S.E.M. (N=5
trials for one neurone). (D) During struggling movements of another animal, monitored by recordings of the main flexor muscle (third trace) and of the force generated by the tibia (bottom trace), a barrage of hyperpolarising potentials in an accessory flexor muscle fibre (arrow, second trace) was accompanied by a reduction in the TR firing frequency (digital output, top trace). (E) In one animal, the instantaneous firing frequency of the tension receptor (bottom trace) was transiently increased following each excitatory potential recorded intracellularly from the accessory flexor muscle (middle trace), but was relatively unaffected by inhibitory inputs from common inhibitor 2 (CI2; top and middle traces). CI2was recorded intracellularly from
its soma in the metathoracic ganglion. Scale bars: 12.5 mV (A,D); 5 mV, 5 nA, 15 Hz (tension receptor), 7.5 Hz (anterior flexor) (B); 0.1 g (D); 5.4 mV (E); 1 s (A,B); 0.24 s (D); 1.4 s (E).
B
C A
Tension receptor
Tension receptor
Tension receptor
0.05 mV
CI2
CI3
CI1
10 ms
Fig. 6. (A–C) All three common inhibitors (CI1, CI2, CI3, top traces)
receptor inserts. These fibres attach to the apodeme at an angle, so a linear movement of the apodeme (i.e. linear stretch of the entire flexor) will cause a non-linear change in the length of the ‘receptor’ fibres, thus probably contributing to the apparently non-linear length–tension relationship. In any case, the percentage change in TR spike frequency as the tibia is set at different angles in the range 0–160 ˚ is only about 6 % of its maximum spike frequency during isometric contractions of the accessory flexor muscle. We conclude that the TR is minimally important in coding static joint position. Golgi tendon organs of vertebrates also respond primarily to active contraction of their associated muscle fibres, and not to passive stretch (Jami, 1992).
Rapid stretches of the accessory flexor muscle generated transient increases in receptor firing rate (Fig. 3D,E). These presumably reflect transient transmission of tension to the proximal end of the muscle (where the receptor lies) that would rapidly be damped by the viscoelasticity of the fibres. Faster stretches caused larger transient changes in firing rate. Only very rapid movements over relatively large arcs caused substantial changes in firing frequency, so it seems unlikely that in natural situations the phasic response is particularly important (see Fig. 3Aiii,iv for more natural movements).
The metathoracic TR, like those of the anterior legs,
responds much more strongly to active isometric tension than to imposed stretch of the muscle, indicating that tension, rather than length, is the primary stimulus. Artificial activation of a single excitatory flexor motor neurone is sufficient to affect the firing rate of the receptor. The relationship between the firing rate of the motor neurone and that of the receptor is linear, at least for the (lower) frequencies that could be tested. The accessory flexor/tension receptor system generally acts like a low-pass filter, smoothing out the more rapid changes in motor neurone firing frequency (see Fig. 5B). This is consistent with the conclusion reached above that the accessory flexor is primarily a slow, tonic muscle. Rarely, however, individual action potentials of an excitatory motor neurone caused clear changes in the TR firing rate (Fig. 5E), suggesting, perhaps, that the gain of the system can increase. Tension generated by the main part of the flexor muscle, or by the extensor muscle, is not registered by the TR. In other words, it responds only to tension generated by the accessory flexor fibres.
Reflex effects of the tension receptor
By combining our knowledge of motor neuronal connections (see references in Materials and methods) with the synaptic connections revealed in the present study, it is possible to propose a circuit diagram of the reflex effects of
B
C D
A
Tension receptor
Tension receptor
Tension receptor
Tension receptor
0.1 mV (A,B,D) 0.05 mV (C)
SETi
Flexor
Flexor Flexor
Quiescent
Aroused
10 ms
the TR (Fig. 10). Three salient points emerge. First, in quiescent animals, the TR gives primarily positive feedback onto the flexor tibiae motor neurones (Fig. 9), including those that innervate the accessory flexor. Second, negative feedback is also provided to the accessory flexor muscle (through the CIs), presumably to prevent instability of the positive feedback loop. Third, the extensor tibiae muscle receives reflex inhibition from the TR (through the actions of the receptor on SETi and CI1), complementing the flexor excitation. The predominant effect of TR activity in quiescent animals is, as demonstrated by electrically induced contraction of the receptor muscle, to support tibial flexion.
In active animals (defined here as those attempting to struggle), the excitatory effects on flexor motor neurones were sometimes, but not always, replaced by inhibitory effects. Role of the common inhibitors
The common inhibitors exclusively innervate muscle units that receive slow motor innervation. They act to gate out this excitatory input and enhance the rate of muscle fibre relaxation. During rhythmic movements, the effect of this is to disable tonic fibres that might otherwise cause a build up of residual tension and thus increase joint stiffness. This permits fast motor units to dominate, and increases the velocity of the movement (Wolf, 1990). In the TR system, at least two mechanisms could operate under different conditions. First, and simplest, depending upon the relative synaptic efficacy of the various pathways, the common inhibitors could provide a negative feedback component to the positive feedback reflex loop represented by the excitatory connections between the TR and accessory flexor excitors. This could act to gate out the positive feedback. Second, the inhibitors could be driven centrally to relax both the accessory and main flexor muscles during rapid tibial extensions. Inhibition of the accessory flexor muscle during tibial extension should prevent it from building up tension and thus prevent an increase in TR firing rate. This mechanism may be particularly important during kicking and jumping (see below).
The tension receptor could participate in a servomechanism The accessory flexor/tension receptor system could act as a servomechanism if the slow motor neurones that innervate the main and accessory flexor muscles were driven centrally to produce a set posture. If the system were perturbed by passive tibial extension or an increased load, the TR would respond with increased firing. This would excite the flexor motor neurones and inhibit SETi. As long as the load or imposed extension remained, the positive feedback would continue (being limited by the maximal frequency of receptor firing). Once the muscle contraction overcame the load and the tibia re-flexed, then the receptor firing rate would decrease and the positive feedback subside. The responses illustrated in Fig. 9 demonstrate that such reflex circuitry is available to the locust, although we do not know whether it is used in this way. A similar proposal has been made for the presumably homologous receptors of the prothoracic and mesothoracic legs (Theophilidis and Burns, 1979).
A
Tension receptor Midline spiker
C
10
8
6
4
2
0
PSP
central delay (ms)
hFl dFl hSETi dCI dOther PSP type
B
Tension receptor
0.06 mV
Unidentified
10 ms
Fig. 8. (A) A spiking neurone recorded near the ventral midline of the ganglion received a depolarising input with a latency of 1.5 ms. (B) An unidentified neurone recorded from near the cell body of CI3
B A
0.25 mV
0.13 s (A) 0.5 s (B) SETi
Flexor
n5B1 Myogram
n5B1
Fig. 9. Electrical stimulation of the accessory flexor muscle (largest deflections in lower traces of A,B) caused an increase of the tension receptor firing rate (small deflections in lower traces), which led to a reduction in the firing rate of the slow extensor tibiae motor neurone (top two traces in A) and a burst of depolarising potentials in an anterior flexor tibiae motor neurone (top trace in B).
Fig. 10
Inhibitory
Excitatory 1.5–2.5 ms
2.2–3.8 msa 3.7–8.1 ms
6–10 ms
0.9–1.8 msb
TR
Flex Flex CICI2
3 CI1 SETi FETi
Int MS
Int
Accessory flexor muscle
Walking
During walking, the swing phase for the metathoracic leg is driven by tibial flexion (Burns, 1973). Because unopposed active tibial flexions do not cause marked increases of TR firing rate (not shown; see Theophilidis and Burns, 1979), the receptor must have only a minimal influence on normal swing. Obstruction of swing, however, would cause an increase of flexor tension and, thus, a pronounced increase in TR firing rate. A similar increase of receptor firing rate would occur if the flexor motor neurones began firing while SETi was still active at the end of stance or while the tarsus was still in contact with the substratum. The overall positive feedback reflex
elicited by the tension receptor would enhance flexor firing and inhibit SETi, thus reinforcing the swing phase in the face of such perturbations.
Jumping
The accessory flexor muscle, like the main tibial flexor muscle, should be strongly activated during the co-contraction phase of these motor programmes. Because the tibia is fully flexed, there should be a build up of isometric tension in the accessory flexor muscle and a large resulting increase in tension receptor firing rate. The accessory flexor tension receptor makes connections that are appropriate for it to contribute to continued flexor firing during co-contraction (i.e. it excites tibial flexor motor neurones) and to promote the final rapid extension (through excitation of the common inhibitors). The common inhibitors are depolarised throughout co-contraction, but particularly near the end (Burrows, 1995). It is not known what proportion of this depolarisation is derived from sensory inputs, although the rhythmic drive seen in CI1 during walking is predominantly central in origin (Wolf, 1990). In any case, the role of sensory feedback in driving the jump and kick circuitry is still unclear (see Heitler and Bräunig,
Fig. 10. Summary of the connections made by the metathoracic flexor tension receptor (TR). Values near synapses are the observed ranges of synaptic latencies (arange for all flexors, brange for all common
inhibitors; see text for details). Flex, flexor motor neurones; Int, unidentified interneurone(s); CI, common inhibitor motor neurones (1–3); SETi, slow extensor tibiae motor neurone; FETi, fast extensor tibiae motor neurone; MS, midline spiking interneurones. Two slow flexor motor neurones innervated the accessory and main flexor muscles. Seven other flexor motor neurones innervated only the main flexor muscle. No inputs from the TR were seen in FETi.
Table 1. Summary of the physiological characteristics of multipolar sensilla (MS) of locust and stick insect legs
Independent motor
Receptor Receptor site Adequate stimulus Stimulus modality innervation? Reflexes evoked
cxMROa Receptor muscle Trochanteral depression; Passive muscle stretch; Yes Inhibition of agonist and
(locust) receptor muscle receptor muscle excitation of antagonist
contraction tension power muscles (resistance
reflex)
Pro/meso TRb Main flexor Tibial extension; flexor Passive muscle stretch; No Positive feedback onto
(locust) muscle contraction power muscle tension homonymous power muscle; antagonist inhibition
Meta TRc Accessory flexor Tibial extension; Passive muscle stretch; No Positive feedback onto
(locust) receptor muscle receptor muscle parallel power muscle and
contraction tension receptor muscle;
antagonist inhibition. Negative feedback onto
same muscles
Joint MSd Arthrodial membrane Tibial extension and Arthrodial membrane − Weak positive feedback onto
(locust) and lateral cuticle static joint position distortion, joint strain power muscle; negative feedback onto same muscle
Apodeme Extensor apodeme Tibial movement away Connective tissue − Unknown receptore from mid-position distortion? (Not power
(stick insect) muscle tension)
Meso/meta TRe Main flexor Tibial flexion; receptor Passive power muscle ? Weak excitatory feedback
(stick insect) muscle contraction stretch; power muscle onto antagonist power
tension muscle
aBräunig and Hustert (1985a); bTheophilidis and Burns (1979); cpresent report; dRDPL, RDAL, RVPL; Coillot and Boistel (1969), Bässler
(1977), Heitler and Burrows (1977), Williamson and Burns (1978); eBässler (1977).
1988). Ablation of the accessory flexor muscle does not prevent jumping (A. P. Norman and T. Matheson, unpublished results) so, as for other sense organs, the animal must be able to compensate to some extent for the loss of any such feedback. There are three clear questions that should guide further study of this system. First, what is the pattern of motor activity in the accessory flexor muscle during voluntary behaviours, particularly walking and the co-contraction phase of kicking and jumping? Second, what are the relative efficacies of excitor and inhibitor motor axons in regulating tension in the accessory flexor muscle? Third, how are TR reflexes gated centrally during voluntary behaviour?
Comparative aspects of multipolar sensillum reflex circuits How does this reflex organisation compare with that known for other leg multipolar sensilla? Are there common principles of operation unique to the type II sensilla, inasmuch as they are usually associated with muscles? Table 1 summarises the available information for stick insect and locust receptors. Two kinds of reflexes are seen in multipolar receptor systems. The first is a negative feedback reflex onto the main power muscle that stimulated the receptor through joint movement or tension (cxMRO, stick insect TR). This is usually accompanied by excitation of the antagonistic power muscle, and constitutes a resistance reflex. The second is a positive feedback reflex onto the homonymous or parallel power muscle (locust tibial flexor TRs, joint multipolar sensilla). There is generally inhibition of antagonistic power muscles, thus forming a load-compensating reflex circuit. Such circuits have also been found in muscle tension receptors of other arthropods (e.g. Macmillan, 1976). In addition, the positive feedback pathway of the locust metathoracic TR has a negative feedback component (Fig. 10). The negative component is mediated by the common inhibitors CI2and CI3, as well as a long-latency inhibitory pathway onto the flexor motor neurones. It appears to be a specialisation of the locust metathoracic TR reflex circuitry, because it has not been found in other multipolar sensillum reflexes. Its function will remain unclear until we learn more about the conditions under which it becomes predominant. In principle, it could function as a high-threshold mechanism for gating out the positive feedback to the flexor muscle.
In summary, it appears that the multipolar receptors in insect legs have been adapted for a variety of reflex systems, some of which are devoted to feedback control of muscle tension in the face of variable load, while others are devoted to feedback maintenance of joint position (Bässler, 1977; Theophilidis and Burns, 1979; Bräunig and Hustert, 1985b; present study).
We thank Jan McKenzie for her help with the electron microscopy, and Jacques Hervé for his help with some of the initial experiments. Peter Bräunig, Malcolm Burrows, Fred Kuenzi and two anonymous referees made valuable comments on the manuscript. T.M. was supported by a research fellowship at Girton College, Cambridge, and by an SERC grant to Malcolm Burrows. L.H.F. was the recipient of an Erskine Fellowship from the University of Canterbury.
References
BÄSSLER, U. (1977). Sense organs in the femur of the stick insect and their relevance to the control of position of the femur–tibia joint. J. comp. Physiol. 121, 99–113.
BENNET-CLARK, H. C. (1975). The energetics of the jump of the locust
Schistocerca gregaria. J. exp. Biol. 63, 53–83.
BRÄUNIG, P. (1982). The peripheral and central nervous organisation of the locust coxo-trochanteral joint. J. Neurobiol. 13, 413–433. BRÄUNIG, P. (1985). Strand receptors associated with the femoral
chordotonal organs of stick insect legs. J. exp. Biol. 116, 331–341. BRÄUNIG, P. ANDHUSTERT, R. (1985a). Actions and interactions of proprioceptors of the locust hind-leg coxo-trochanteral joint. I. Afferent responses in relation to joint position and movement. J. comp. Physiol. 157A, 73–82.
BRÄUNIG, P. ANDHUSTERT, R. (1985b). Actions and interactions of proprioceptors of the locust hind-leg coxo-trochanteral joint. II. Influence on the motor system. J. comp. Physiol. 157A, 83–89. BRÄUNIG, P., PFLÜGER, H.-J. ANDHUSTERT, R. (1986). The specificity
of central nervous projections of locust mechanoreceptors. J. comp. Neurol. 218, 197–207.
BURNS, M. D. (1973). The control of walking in Orthoptera. I. Leg
movements in normal walking. J. exp. Biol. 58, 45–58.
BURNS, M. D. (1974). Structure and physiology of the locust femoral chordotonal organ. J. Insect Physiol. 20, 1319–1339.
BURROWS, M. (1995). Motor patterns during kicking movements in
the locust. J. comp. Physiol. 176, 289–305.
BURROWS, M. ANDPFLÜGER, H.-J. (1988). Positive feedback loops from proprioceptors involved in leg movements of the locust. J. comp. Physiol. 163, 425–440.
BURROWS, M., WATSON, A. H. D. AND BRUNN, D. E. (1989). Physiological and ultrastructural characterisation of a central synaptic connection between identified motor neurones in the locust. Eur. J. Neurosci. 1, 111–126.
COILLOT, J. P. ANDBOISTEL, J. (1968). Localisation et déscription des
récepteurs à l’étirement au niveau de l’articulation tibio-femorale de la patte sauteuse du criquet, Schistocerca gregaria. J. Insect Physiol. 14, 1661–1667.
COILLOT, J. P. ANDBOISTEL, J. (1969). Étude de l’activité électrique
propagée des récepteurs à l’étirement de la patte métathoracique du criquet, Schistocerca gregaria. J. Insect Physiol. 15, 1449–1470. ELDER, H. Y. (1975). Muscle structure. In Insect Muscle (ed. P. N. R.
Usherwood), pp. 1–7. London, New York, San Francisco: Academic Press.
FIELD, L. H. ANDBURROWS, M. (1982). Reflex effects of the femoral chordotonal organ upon leg motor neurones of the locust. J. exp. Biol. 101, 265–285.
HALE, J. P. AND BURROWS, M. (1985). Innervation patterns of inhibitory motor neurones in the thorax of the locust. J. exp. Biol.
117, 401–413.
HEITLER, W. J. (1974). The locust jump. Specialisations of the
metathoracic femur–tibia joint. J. comp. Physiol.89A, 93–104. HEITLER, W. J. AND BRÄUNIG, P. (1988). The role of fast extensor
motor activity in locust kick reconsidered. J. exp. Biol. 136, 289–309.
HEITLER, W. J. ANDBURROWS, M. (1977). The locust jump. II. Neural circuits of the motor programme. J. exp. Biol. 66, 221–241. HOFMANN, T. ANDKOCH, U. (1985). Acceleration receptors in the
femoral chordotonal organ in the stick insect Cuniculina impigra. J. exp. Biol. 114, 225–237.
femoral chordotonal organ in the stick insect Cuniculina impigra. J. exp. Biol. 114, 207–223.
HOYLE, G. (1975). The neural control of skeletal muscles. In Insect
Muscle (ed. P. N. R. Usherwood), pp. 501–543. London: Academic Press.
HOYLE, G. (1978). Distributions of nerve and muscle fibre types in locust jumping muscle. J. exp. Biol. 73, 205–233.
HOYLE, G. ANDBURROWS, M. (1973). Neural mechanisms underlying behaviour in the locust Schistocerca gregaria. I. Physiology of identified motor neurones in the metathoracic ganglion. J. Neurobiol. 4, 3–41.
JAMI, L. (1992). Golgi tendon organs in mammalian skeletal muscle: functional properties and central actions. Physiol. Rev. 72, 623–666. KUSTER, J. E. ANDFRENCH, A. S. (1983). Sensory transduction in a locust multipolar joint receptor: the dynamic behaviour under a variety of stimulus conditions. J. comp. Physiol.150A, 207–215. MACMILLAN, D. (1976). Arthropod tension receptors. In Structure and
Function of Proprioceptors in the Invertebrates (ed. P. J. Mill), pp. 427–439. London: Chapman and Hall.
MATHESON, T. (1990). Responses and location of neurones in the locust metathoracic femoral chordotonal organ. J. comp. Physiol.
166A, 915–927.
MATHESON, T. (1992a). Morphology of the central projections of
physiologically characterised neurones from the locust metathoracic femoral chordotonal organ. J. comp. Physiol. 170A, 101–120.
MATHESON, T. (1992b). Range fractionation in the locust metathoracic
femoral chordotonal organ. J. comp. Physiol. 170A, 509–520. MATHESON, T. ANDDITZ, F. (1991). A simple computer-controlled
analogue ramp generator for producing multiple ramp-and-hold stimuli. J. Neurosci. Methods 39, 45–52.
MATHESON, T. AND FIELD, L. H. (1990). Innervation of the
metathoracic femoral chordotonal organ of Locusta migratoria. Cell Tissue Res. 259, 551–560.
MÜCKE, A. (1991). Innervation pattern and sensory supply of the midleg of the locust Schistocerca gregaria (Insecta, Orthoptera). Zoomorphologie 110, 175–187.
PARKER, D. J. (1993). The physiology and pharmacology of direct
central connections between the fast extensor and flexor tibiae motor neurones in the locust. PhD thesis. University of Cambridge, UK.
PHILLIPS, C. E. (1981). Organisation of motor neurones to a multiply
innervated insect muscle. J. Neurobiol. 12, 269–280.
SNODGRASS, R. E. (1929). The thoracic mechanisms of a grasshopper
and its antecedents. Smithson. misc. Collns 82, 1–111.
THEOPHILIDIS, G. ANDBURNS, M. D. (1979). A muscle tension receptor
in the locust leg. J. comp. Physiol. 131, 247–254.
THEOPHILIDIS, G. ANDDIMITRIADIS, V. K. (1990). The structure and
innervation of the metathoracic flexor tibiae muscle of two species of Orthoptera (Insecta). Comp. Biochem. Physiol. 97A, 583–594. WILLIAMSON, R. ANDBURNS, M. D. (1978). Multiterminal receptors
in the locust leg. J. Insect Physiol. 24, 661–666.
WOLF, H. (1990). Activity patterns of inhibitory motor neurones and their impact on leg movements in tethered walking locusts. J. exp. Biol. 152, 281–304.
ZAWARZIN, A. R. (1912). Histologische Studien über Insekten. II. Das
sensible Nervensystem der Aeschna larven. Z. wiss. Zool. 100, 245–286.

![Fig. 6. (A–C) All three common inhibitors (CI 1 , CI 2 , CI 3 , top traces) received excitatory inputs from the tension receptor [recorded in both N5 (middle trace) and n5B1 (lower trace) to allow calculation of spike conduction velocity]](https://thumb-us.123doks.com/thumbv2/123dok_us/1159981.637558/11.913.493.829.93.788/inhibitors-received-excitatory-receptor-recorded-calculation-conduction-velocity.webp)