FABRICATION, DEVELOPMENT AND CHARACTERIZATION OF CARVEDILOL TRANSDERMAL PATCHES: AN EMPIRICAL STUDY
Full text
Figure


Related documents
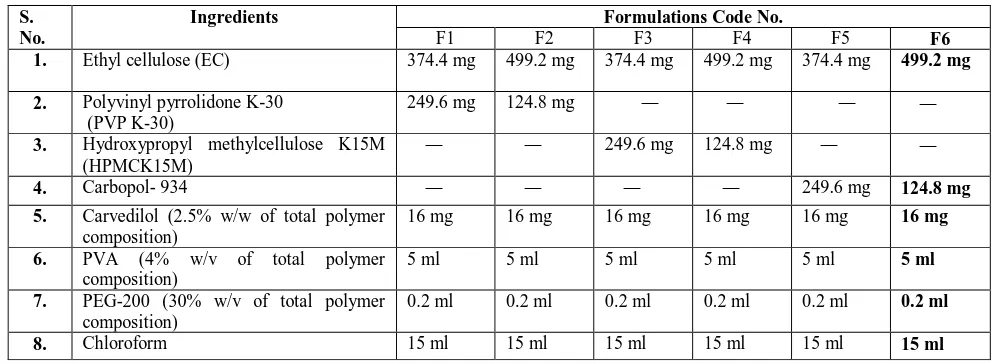
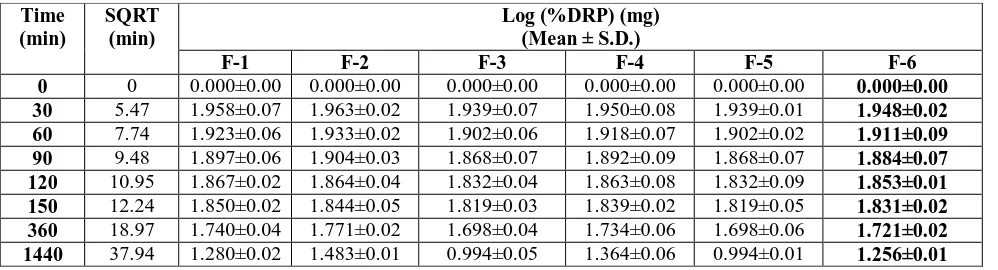
The prepared formulations were evaluated for thickness, weight variation, drug content, folding endurance, moisture content, in vitro permeation studies.. The
All the prepared formulation were subjected to evaluation studies i.e., weight variation, thickness, drug content, moisture content, moisture uptake, flatness and in-vitro
The physicochemical parameters such as flexibility, thickness, smoothness, weight variation, moisture content, hardness, folding endurance and tensile strength were
The formulated patches will evaluated for physical appearance, thickness, folding endurance, weight uniformity, percentage moisture uptake, percentage moisture loss,
The prepared transdermal films were evaluated for thickness, folding endurance, weight variation, flatness, moisture absorption, moisture loss, moisture content,
The films prepared by general procedure were evaluated for the following properties such as weight variation, thickness, folding endurance, estimation of
The prepared transdermal patches of nebivolol were evaluated for thickness, mass variation, drug content, moisture content, moisture vapor transmission, folding endurance,
The prepared patches were subjected to folding endurance, thickness of the film, weight uniformity, drug content, percentage moisture uptake, percentage moisture content, water

