Formulation and Evaluation of Sustained Release Bilayer Tablets of Glimepiride and Metformin HCl.
Full text
Figure
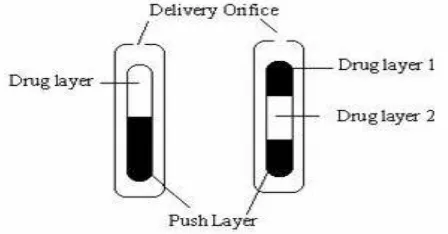
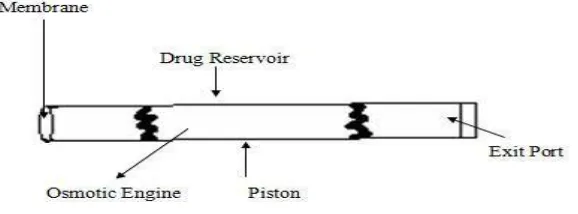
Related documents
In the present work, the sustained release matrix tablets of gliclazide were prepared by direct compression technique using synthetic polymers like
prepared totally and all the batches showed the release profile for a period of 24 hours. 49 formulated and evaluated of sustained release matrix tablets of Losartan
To study the kinetics of drug release from the sustained release matrix, the in vitro dissolution. study data of the optimized batch (F 5 ) was fitted into various kinetic
Hadi MA, Lokeswara Babu V, Pal N, Formulation & Evaluation of Sustained Release Matrix Tablets of Glimepiride Based on Combination of Hydrophilic &
Evaluation tests for prepared sustained release tablets: The formulated tablets were evaluated for the following Pre, post compression quality control studies
Based on the preformulation studies, 10 optimized formulations were prepared by direct compression using different controlled release polymers such as HPMC K4M, HPMC K100 LV
Immediate release layer delivers the initial dose, it contains superdisintegrant which increase drug release rate whereas sustained release layer gastro retentive by
The tablets were prepared by direct compression technique using HPMC-K4M, HPMC-K100M, Eudragit RSPO, Ethyl Cellulose as Sustained release polymer.. These SR tablets were