Materials Science and Metallurgy Engineering
Full text
Figure
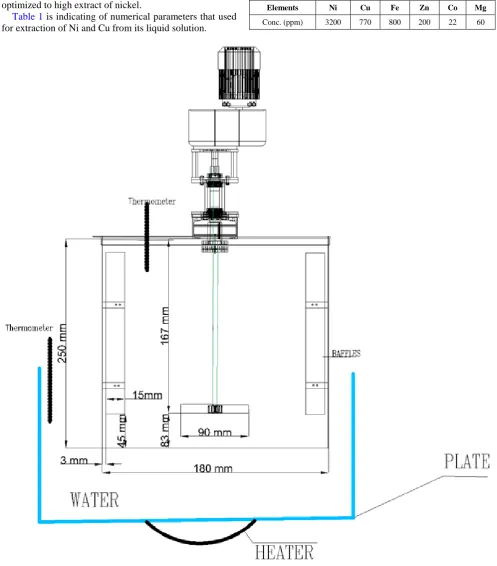
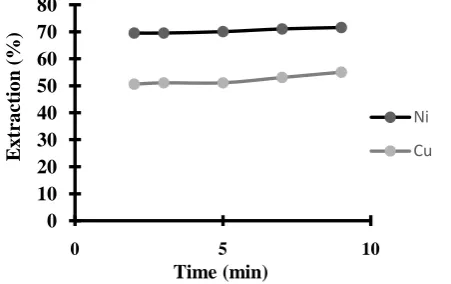
Related documents
Transplantation - Criteria for acceptance – The committee discussed this area and agreed that whilst the criteria for acceptance could have a large impact on an
This essay asserts that to effectively degrade and ultimately destroy the Islamic State of Iraq and Syria (ISIS), and to topple the Bashar al-Assad’s regime, the international
Most of the investigated possible sources of stress showed significant correlations with burnout. The most interesting findings were that women seems to experience more “Demands”
Knowledge and use of insecticide treated nets as a malaria preventive tool among pregnant women in a local government area of Lagos state,
In this task, repeated pairing of unhealthy food types to no-go cues took place in the intervention condition while both unhealthy as well as healthy food types were paired
Statistically significant variances at level p 0.05 were found between peanut species for the major unsaturated fatty acids, such as oleic (C18:1) and linoleic acid (C18:2)
• The pain anticipation scores according to the items expected pain, expected unpleasantness and expected fear are negative correlated with pain experience,