Evaluation of Oxidative Stress and Stress Induced Serum
Protein Changes in Psoriasis
*Ashok Vanangamudi
1, Divya Ramanathan
21Assistant Professor, Department of Biochemistry, Karpagam Faculty of Medical Sciences and Research, Coimbatore,
India-641032
2Assistant Professor, Department of Physiology, Karpagam Faculty of Medical Sciences and Research, Coimbatore.
Psoriasis is a non-infectious, chronic inflammatory disease of the skin. This study aimed to explore the oxidative stress and stress induced protein changes in psoriasis and to analyze the possible interrelationship between these factors among psoriasis patients. This was a hospital-based case control study which included 50 cases of psoriasis and 50 healthy controls. Serum malondialdehyde was measured by thiobarbituric acid reactivity assay method. Protein carbonyl (PC) was measured by Levine’s method. Protein bound sialic acid (PBSA) was measured using modified Aminoff’s method using a spectrophotometer. Data analysis was done using SPSS software version 16.0. Our results showed significant elevation in the oxidant status (MDA, PC and PBSA) among the psoriasis cases compared to the controls. Multiple linear regression analysis showed a linear relationship between MDA, PBSA and duration of psoriasis. Our study provides evidence to increased ROS production, indicated by increased lipid peroxidation in serum and stress induced protein changes in psoriasis. The findings of our study also suggest that as the duration of psoriasis increases the level of oxidative stress also increases which is indicated by the increase in MDA and PBSA levels.
Keywords: oxidative stress, malondialdehyde, Protein carbonyl, PBSA
INTRODUCTION
Psoriasis is a disorder of the skin characterized by inflammation of the skin and hyperproliferation of keratinocytes. The most commonly affected are young or middle-aged adults (Camp RDR, 1998).Around 2% of the general population worldwide are affected with psoriasis (Wolters M et al, 2005). The prevalence of psoriasis in Indian population varies from 0.4% - 2.4% (Dogra M et al, 2010).
The causes for psoriasis are genetic factor, trauma, skin infection, drugs, emotional stress, alcohol and smoking. The generation of reactive oxygen species (ROS) from neutrophils, keratinocytes and fibroblasts can lead to neutrophil activation and they have major role in the development of psoriasis (Vivian S 2013).
Oxidative stress is due to excessive production of reactive oxygen species (ROS) or defect in its removal mechanisms. The free radicals generated by oxidative stress causes oxidation of polyunsaturated fatty acids in cell membrane and results in the generation of lipid peroxidation products like malondialdehyde (MDA). In
psoriatic patients, the cell membrane of skin cells has increased arachidonic acid, which is the substrate for production of malondialdehyde. MDA is used as a biomarker of oxidative stress and lipid peroxidation (Vijaykumar M et al 2014, Sikar A. 2012, Trouba K. 2002). The ROS can damage the proteins or induce chemical modification of amino acids in the proteins during the process of oxidative stress. Carbonyl groups are introduced into proteins by reaction with ROS produced during lipid peroxidation. The carbonyl content of serum proteins is used as a biomarker of protein oxidation (Stadtman E,1998).
*Corresponding Author: Ashok Vanangamudi, Assistant Professor, Department of Biochemistry, Karpagam faculty of medical sciences and research, Coimbatore, India-641032. E-mail: dr.ashokmbbs1986@yahoo.com, Tel: +919159572499, Co-Author Tel: +919074202158 Email:
divyaashok47@yahoo.in
Research Article
Sialic acid is the terminal carbohydrate in the glycan moiety of glycoproteins. Sialic acid is a N-acetylated derivative of neuraminic acid which increases during injury and inflammation (Devatdarova M, 1968). According to a study by Rajapppa M et al (2016) serum levels of oxidative stress and inflammatory markers like highly sensitive C-reactive protein, total sialic acid, protein bound sialic acid were significantly higher in psoriasis vulgaris patients as compared to the normal controls. This shows that oxidative stress and inflammatory markers like serum total sialic acid and protein bound sialic acid are associated with the pathogenesis of psoriasis.
The objectives of the study were
1. To analyze oxidative stress in patients with psoriasis and compare it with normal controls.
2. To estimate oxidative stress induced protein carbonyl content (PC) and protein bound sialic acid (PBSA) in psoriasis and compare it with controls.
3. To find the correlation between oxidative stress, protein carbonyls and protein bound sialic acid in patients with psoriasis.
MATERIALS AND METHODS
This was a hospital-based case control study. This study was conducted after getting approval by the Institutional Ethics Committee Sri Manakula Vinayagar Medical College and Hospital, Pondicherry. The duration of study was two years 2013-2015.The sample size was 100 which included 50 cases of psoriasis and 50 healthy controls.
Inclusion criteria
Cases: Patients diagnosed to have psoriasis in the Department of Dermatology.
Controls: Normal healthy controls, non-psoriatric patients attending dermatology outpatient department.
Exclusion criteria
1. Psoriasis patients with systemic illness like diabetes mellitus, renal failure, coronary artery disease and history of recent drug intake for treatment.
2. Any subjects with systemic infections and chronic inflammatory conditions.
3. Any subjects who were not willing to give informed consent.
Biochemical analysis
Serum total protein was estimated by Biuret method (Doumas BT. 1981). Serum MDA was estimated by thiobarbituric acid reactivity assay method (Satok K 1978).
12 Serum protein carbonyl was estimated by modified
Levine’s method (Chakraborthy H .2000). Serum protein bound sialic acid was estimated by modified Aminoff’s method (Aminoff D 1961).
Statistical Analysis
The data was presented as mean ± standard deviation (mean ± SD). Student t test was used to compare the various parameters between psoriasis subjects and controls. Multiple linear regression analysis was done to analyze the relationship between various parameters of
oxidative stress. P value of less than 0.05 was statistically
significant.
RESULTS
The comparison of various baseline characteristics of the study groups was shown in Table1. All data were represented as mean ± standard deviation. In our study it was observed that the parameter of oxidative stress, MDA was elevated (p<0.001) in psoriatic patients as compared to controls (Table2). Also, the stress induced serum protein changes, PC and PBSA concentrations were high (p<0.001) in the patients with psoriasis as compared to controls (Table2).
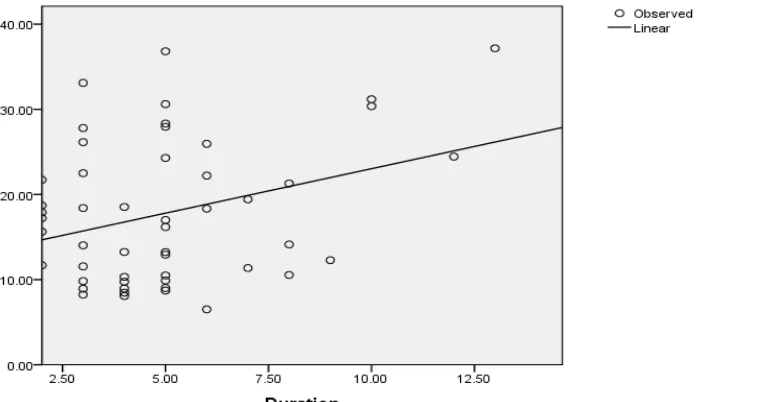
Regression analysis showed a linear relationship between MDA and the duration of psoriasis (Table 3 and Figure 1). Linear regression analysis showed a linear relationship between PBSA and the duration of psoriasis (Table 3 and Figure 2).
DISCUSSION
The skin is a vulnerable target for oxidative injury by free radicals, as it is continuously exposed to UV radiation and other environmental stresses which generate reactive oxygen species (Dipali P et al 2009). In normal cells, there will be a balance between oxidative damage and protection by antioxidant protections. However inadequate antioxidant protection mechanisms or excess free radical production generates a condition known as oxidative stress (Trouba K et al 2002).
Psoriasis is a chronic inflammatory disorder of the skin which is characterized by increased proliferation of keratinocytes and inflammation in the epidermis and dermis (Young CN et al 2008). Various environmental and genetic factors, as well as intracellular and intercellular mediators, are found to play an important role in the pathogenesis of psoriasis (Myers WA et al 2006).
Our study showed significant difference in the oxidant status between the psoriasis cases and controls. Oxidant status in psoriasis was assessed by the estimation of serum malondialdehyde, protein carbonyl and protein bound sialic acid. In the present study, serum MDA levels were increased in psoriasis patients when compared to normal controls (p<0.001). These findings are in correlation with the findings of Dipali P et al (2009) and Jyothi RS et al (2011).
Protein carbonyl content is the biomarker of oxidative protein damage. A study by VV Barygina et al (2013) showed that protein carbonyl was increased in patients with psoriasis compared to the control group. In our study the serum PC were elevated in patients with psoriasis as compared to the controls.
Several studies have shown the relation between oxidative stress and the alteration in sialylation of proteins. Increased sialic acid concentrations have been reported during inflammatory processes in plasma and serum (Nanetti L et al 2008, Ajit Varki 2008).In our study serum protein-bound sialic acid was increased in psoriasis patients as compared to the controls.
Multiple linear regression analysis showed a correlation between MDA and duration of psoriasis. Similarly, PBSA was found to be correlated with the duration of psoriasis. This indicates that the level of oxidative stress increases with the duration of psoriasis.
The processes involved in the etiopathogenesis of psoriasis are complex which involves ROS production and free radical damage. Our observation supports the concept that oxidative damage plays an important role in the inflammatory response and etiopathogenesis of psoriasis.
CONCLUSION
The present study provides evidence to increased ROS production, which is indicated by increased production of malondialdehyde in serum and stress induced protein changes in psoriasis. The findings of our study also suggest that the level of oxidative stress increases with the duration of psoriasis. Moreover, dietary supplementation of natural antioxidant vitamins may be considered as a therapeutic approach to prevent the cell damage by free radicals.
Declaration of conflict of interest: None.
REFERENCES
Ajit Varki. (2008). Sialic acids in human health and disease. Trends Mol Med. 14(8):351–360.
Aminoff D. (1961). Methods for the quantitative estimation of neuraminic acid and their application to hydrosylates of sialomucoids. Biochem J. 82:384-92.
Camp RDR: Psoriasis In. Rook A, Wilkinson DS, Ebling FJG (eds) (1998). Rook’s Text Book of Dermatology. 6th ed. New Jersy: Blackwell publishers; p.1589-1592.
Chakraborty H, Ray N. (2000) Lipid peroxidation associated protein damage in rat brain crude synaptosomal fraction mediated by iron and ascorbate. Neurochem Int J. 39:311-17.
Dalle-Donne I, Giustarini D, Colombo R, Rossi R, Milzani A. (2003) Protein carbonylation in human diseases. Trends Mol Med. 9(4):169-176.
Devatdarova M. (1968). Sialic acid levels in the serum of healthy persons and patients with psoriasis. Vestn Dermatol Venerol. 42(8):43-46.
Dipali P Kadam, Adinath N Suryakar, Rajesh D Ankush, Charushila Y Kadam, Kishor H Deshpande. (2010). Role of Oxidative Stress in Various Stages of Psoriasis. Ind J Clin Biochem. 25(4):388–392.
Dogra S, Yadav S. (2010). Psoriasis in India: prevalence and pattern. Ind J Dermatol Venerol Leprol. 76(6):595-601.
Doumas BT. (1981). A candidate reference method for determination of total protein in serum. Clin Chem Acta. 27:1642-1650.
Jyothi RS, Govindswamy KS, Gurupadappa K. (2011) Psoriasis: an oxidative stress condition. J Clin Diagn Res. 5(2):252-253.
Mahmoud Y, Eman N, Najlaa K, Rowida A, Mohamad N. (2013). Role of Oxidative Stress in Psoriasis: An Evaluation Study. J Am Sci. 9(8):151-155.
Myers WA, Gottlieb AB, Mease P. (2006). Psoriasis and psoriatic arthritis: clinical features and disease mechanisms. Clin Dermatol. 24:438–447.
Nanetti L, Vignini A, Raffaelli F. (2008). Sialic acid and sialidase activity in acute stroke. Disease markers. 28:167-173.
Rajappa M, Shanmugam R, Munisamy M, Chandrashekar L, Rajendiran KS, Thappa DM. (2016). Effect of antipsoriatic therapy on oxidative stress index and sialic acid levels in patients with psoriasis. Int J Dermatol. 55(8):422-430.
Satok K. (1978). Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta. 90(1):37-43.
Sikar Akturk A, Ozdogan HK, Bayramgurler D, Çekmen MB, Bilen N, Kıran R. (2012). Nitric oxide and malondialdehyde levels in plasma and tissue of psoriasis patients. J Eur Acad Dermatol Venereol. 26(7):833-837.
Stadtman E, Berlett B. Reactive oxygen mediated protein oxidation in ageing and disease. (1998). Drug Metab Rev. 30:225-43.
Trouba K, Hamadeh H, Amin R, Germolec D. (2002). Oxidative stress and psoriasis. Antioxid Redox Signal. 4(4):665-73.
Vivian S, Krishna M. (2013). Potential role of oxidative stress and antioxidant deficiency in pathogenesis of psoriasis. Int J Pharm Bio Sci. 4(3):1039-1044. VV Barygina, Becatti M, Soldi G, Prignano F, Lotti T, Nassi
P , Wright D , Taddei N , Fiorillo C. (2013). Altered redox status in the blood of psoriatic patients: involvement of NADPH oxidase and role of anti-TNF-α therapy. Redox Rep. 18(3):100-106.
Wolters M. (2005). Diet and psoriasis: Experimental data and clinical evidence. Br J Dermatol. 153:706-14.
Young CN, Koepke JI, Terlecky LJ, Borkin M, Boyd S, Terlecky SR. (2008). Reactive oxygen species in tumor necrosis factor α activated primary human keratinocytes: implications for psoriasis and inflammatory skin disease. J Invest Dermatol. 128(11): 2606–2614.
Zhou Q, Mrowietz U, Rostami-Yazdi M. (2009). Oxidative stress in the pathogenesis of psoriasis. Free Radic Biol Med. 47:891–905.
APPENDIX
Table 1: Comparison of baseline characteristics (mean ± standard deviation) in patients with psoriasis and controls Parameters Cases(n=50) Controls(n=50) P value
Age (years) 41.20 ±9.21 41.56 ±8.99 0.82 Height(m) 1.48 ±0.08 1.49 ±0.07 0.60 Weight (kg) 60.22 ±7.96 59.80 ±8.83 0.83 BMI (kg/m2) 27.51 ±3.39 27.19 ±4.23 0.68
Systolic BP (mmHg) 116.96 ± 6.54 114.88 ±4.14 0.66 Diastolic BP (mmHg) 71.04 ±7.01 69.52 ±6.86 0.27 Pulse rate (beats/min) 76.16 ±7.01 75.70 ±6.58 0.72 Serum Total protein (g/dl) 7.18 ± 0.51 7.02 ±0.51 0.11 Duration of psoriasis (years) 5.02 ±2.55 - -
Data are presented as Mean ±SD.*P value ≤0.05 is statistically significant. Independent student t test was used to analyze the data. BMI=Body mass index, BP=Blood pressure.
Table 2: Comparison of oxidative stress parameters (mean ± standard deviation) in patients with psoriasis and controls Parameters Cases(n=50) Controls(n=50) P value
Serum MDA (µmoles/L) 6.31±2.68 3.65±1.48 <0.001* Serum PC (µg/mg of protein) 2.88 ±0.75 2.43 ±0.35 <0.001* Serum PBSA (nmol/mg of protein) 17.82 ±8.33 10.41±3.55 <0.001*
Data are presented as Mean ±SD.*P value ≤0.05 is statistically significant. Independent student t test was used to analyze the data. MDA=Malondialdehyde, PC=Protein carbonyl, PBSA=Protein bound sialic acid.
Table 3: Linear regression analysis to find the effect of duration of psoriasis on parameters of oxidative stress
Independent Nonstandardized coefficients Standardized coefficients
P value Variables B value β
Figure 1: Linear regression analysis between duration of psoriasis and MDA levels among the cases
Figure 2: Linear regression analysis between duration of psoriasis and Protein bound sialic acid (PBSA) levels among the cases
Accepted 10 May 2019
Citation: Vanangamudi A, Ramanathan D (2019). Evaluation of Oxidative Stress and Stress Induced Serum Protein Changes in Psoriasis. International Research Journal of Biochemistry and Biotechnology, 5(1): 075-079.