Evidence Based Report
Biological Mesh for Abdominal Wall
Hernia Repair
Reviewer
Mark Ayson
Date Report Completed
July 2013
Important Note:
The purpose of this brief report is to summarise the best evidence for the use of biologic mesh
for abdominal wall hernia repair with a focus on recurrence and complications.
It is not intended to replace clinical judgement, or be used as a clinical protocol.
A reasonable attempt has been made to find and review papers relevant to the focus of this
report, however it does not claim to be exhaustive.
The document has been prepared by the staff of the Research Team, ACC. The content does
not necessarily represent the official view of ACC or represent ACC policy.
This report is based upon information supplied up to July 2013
1.
Background
An abdominal wall hernia is the protrusion of intra-abdominal organs or tissue through a defect in the
abdominal wall. They can be classified into primary ventral and incisional hernia. There are four main
types of primary ventral hernias: umbilical, para-umbilical, epigastric, and spigelian. Incisional hernias
develop at the site of a prior surgery; depending on the technique of the original surgery (laparoscopic
or open surgery) up to 15% of all patients develop incisional hernia
1. Most of these enlarge over time
and can become uncomfortable or painful and give rise to serious complications like bowel
strangulation
1.
Different surgical techniques are used to repair these hernia and a mesh prosthesis may be used to
assist in closing the defect in the abdominal wall by reinforcing the tissue or bridging the defect
1. A
mesh prosthesis can be made of synthetic materials or be biologic in origin.
Surgical repair of incisional hernias using synthetic material like polypropylene or expanded
polytetrafluorethylene mesh have shown a 50% reduction in abdominal hernia recurrence compared to
suture-only repairs at both 3 and 10 years of follow-up
2. Synthetic meshes have high tensile strength
2but can be associated with serious complications, including surgical site infections, delayed wound
healing, skin breakdown, and seroma and fistula formation
3, as well as, bowel obstruction or erosion
into viscera
4.
Biologic meshes have been produced as an alternative to synthetic mesh. They consist of an
extracellular collagen matrix and the commercially available ones are derived from human and porcine
dermis, porcine small intestinal submucosa, and bovine pericardium. After harvesting, the tissue
undergoes a process which removes all the cells from the tissue and some undergo additional
cross-linking between the collagen matrix
4.
The aim of this review is to summarise the best evidence for the use of biologic mesh for abdominal
wall hernia
*repair with a focus on recurrence and complications.
2.
Investigation
A search was conducted in 28 August 2012 in the following database: Ovid MEDLINE(R) In-Process &
Other Non-Indexed Citations, Ovid MEDLINE(R) 1946 to Present, and EMBASE 1988 to Present. The
references of review articles and included studies were also investigated, as was the Worldwide Web.
Only articles published in English were included. A repeat search was conducted in July 2013.
Search terms used included: biological mesh/graft/implant, bioprosthesis, biosynthetic
mesh/graft/implant, ventral/incisional hernia, and hernioplasty/herniorrhaphy.
Inclusion criteria: systematic reviews of biological mesh for ventral/incisional herniae reporting
outcomes such as hernia recurrence and complications.
Exclusion criteria: Animal or laboratory study, narrative review, or editorial; RCT, case-control, cohort,
cross-sectional or case series/reports; non-English studies.
This resulted in identifying 299 articles of which 6 systematic reviews were used in this report. Two
additional systematic reviews were included to give information on recurrence/complication rates in
synthetic mesh and two narrative reviews were also included for their opinions regarding the place
biological mesh plays in ventral/incisional hernia repairs.
Evidence tables were created for each included systematic review (see Appendix 1). Appendix 2
contains a summary of all the relevant primary studies included in the systematic reviews. A table of
the excluded studies can be found in Appendix 3.
Any relevant papers were assessed for their methodological quality using the following SIGN
†criteria:
LEVELS OF EVIDENCE
1++
High quality meta-analyses, systematic reviews of RCTs, or RCTs with a very low risk of bias
1+
Well-conducted meta-analyses, systematic reviews, or RCTs with a low risk of bias
1–
Meta-analyses, systematic reviews, or RCTs with a high risk of bias
2++
High quality systematic reviews of case control or cohort or studies
High quality case control or cohort studies with a very low risk of confounding or bias and a
high probability that the relationship is causal
2+
Well-conducted case control or cohort studies with a low risk of confounding or bias and a
moderate probability that the relationship is causal
2–
Case control or cohort studies with a high risk of confounding or bias and a significant risk
that the relationship is not causal
3
Non-analytic studies, e.g. case reports, case series
Findings
Six systematic reviews
2-7were located and deemed relevant for this report.
The best evidence probably comes from Slater (2013)
4. This systematic review evaluated
post-operative morbidity and hernia recurrence after ventral hernia repair with biological mesh. It was one
of the most methodologically sound of all of the reviews included in this report and included 27 studies
with a total of 1125 participants. The median of reported means were 54.5 years for age, 32.0 kgm
-2for BMI
‡, and 150 cm
2for size of the defect. All median ASA
§scores reported were 3. There were no
statistically significant differences found between subgroups with respect to age or BMI.
The primary studies included almost exclusively three of the commercially available biologic meshes:
Alloderm (derived from human cadaver dermis), Permacol (from porcine dermis), and Surgisis (from
porcine small intestinal submucosa).
Mortality was reported in 19 studies (n=879 patients) and 36 (4.1%) patients died within 30 days of
surgery; all other deaths were noted as unrelated to ventral hernia repair.
Recurrence was reported in 17 studies (n=531 patients) with an overall weighted pooled recurrence
rate of 13.8% (95%CI
**: 7.6 – 21.3). See Table 1 below for details. The recurrence rate was around 3%
for clean/clean-contaminated cases, increased to 23% for contaminated/dirty cases, and 19% for
"complicated" cases. The odds of a recurrence in clean/clean-contaminated cases was statistically
significantly less than the odds of a recurrence in contaminated/dirty or "complicated" cases (OR
††=
55.9 [95%CI: 8.5 – 2321.3; p<0.0001] and 39.0 [95% CI: 6.5 – 1,581.0; p<0.0001] respectively). No
significant difference was found between contaminated/dirty and the "complicated" subgroup
[p=0.2233].
‡ body mass index
§ American Society of Anesthesiologists score - a global score that assesses the physical status of patients before surgery ** 95% confidence interval
Table 1: Study characteristics and recurrence rates for ventral hernia repair with biologic grafts
Reference / Year MINORS
index‡‡ Graft used
No. of patients Mean size of defect (cm2) No. of C/D procedures (%) No. of recurrences (%) Follow-up (range) Clean/Clean-Contaminated Buinewicz 20048 10 Alloderm 36 86.4 0 (0) 1 (2.8) 20 (8–32) Glasberg 20069 12 Alloderm 54 166 0 (0) 0 (0) 18.5 (9–30) Gupta 200610 10 Alloderm 33 NS 0 (0) 8 (24.2) 29 (NS) Boehmler 200911 11 Alloderm 31 NS 0 (0) 0 (0) 20.4 (NS) Helton 200512 13 Surgisis 21 202 0 (0) 0 (0) 14 (2–29) Gupta 200610 10 Surgisis 38 NS 0 (0) 0 (0) 19 (NS)
Overall C/CC (95% CI)† Various 213 2.9% (0.2–8.3)
Contaminated/dirty Buinewicz 20048 10 Alloderm 8 86.4 8 (100) 1 (4.5) 20 (8–32) Alaedeen 200713 11 Alloderm 11 NS 11 (100) 2 (18.2) 14 (2–68) Taner 200914 13 Alloderm 11 NA 13 (100) 0 (0) 12 (64) Ueno 200415 13 Surgisis 20 NS 20 (100) 6 (30.0) 15.7 (NS) Helton 200512 13 Surgisis 19 202 19 (100) 8 (42.1) 14 (2–29) Gupta 200610 10 Surgisis 3 NS 3 (100) 0 (0) 29 (NS) Connolly 200816 10 Permacol 12 NA 12 (100) 5 (41.7) 29 (16–84)
Overall C/D (95% CI)† Various 84 23.1% (11.3–37.6)
Complicated* Kolker 200517 8 Alloderm 16 NS 9 (56.3) 0 (0) 16 (9–23) Jin 200718 10 Alloderm 37 122 16 (43.2) 12 (32.4) 22.2 (15–37) Candage 200819 12 Alloderm 46 NS 17 (37.0) 14 (30.4) 12.1 (NS) Lee 200920 11 Alloderm 68 NS 54 (79.4) 21 (30.9) 15.4 (NS) Parker 200621 10 Permacol 9 NS 5 (55.6) 1 (11.1) 18.2 (NS) Shaikh 200722 12 Permacol 20 180 5 (25.0) 3 (15) 18 (6–36) Hsu 200923 13 Permacol 28 150 NS 3 (10.7) 16 (10–23) Loganathan 201024 10 Permacol 10 NS NS 1 (10.0) 12 (3–62)
Overall ‘‘complicated’’(95% CI)† Various 234 19.4% (11.4–29.0)
Overall combined (95% CI) † Various 531 13.8% (7.6–21.3)
C/CC = clean/clean-contaminated; C/D = contaminated/dirty; NA = not available; NS = not specified.
*This subgroup contains groups of patients for which the outcomes could not be stratified per wound class and/or that contained cases with unclear surgical indications that were classified by the corresponding authors as either ‘‘complex,’’ ‘‘complicated,’’ ‘‘potentially contaminated,’’ or ‘‘high risk.’’
†Weighted pooled proportion using the random-effects (DerSimonian-Laird) model.
†† odds ratio
With regards to surgical morbidity, Slater (2013)
4found 46% of patients experienced some
complication, infection being the most common (21%). There were 249 (16%) patients with wound
infections and 12 (5%) had to have their biologic mesh removed due to infection. Not surprisingly, the
odds of an infection was significantly greater in the contaminated/dirty and "complicated" groups
compared to the clean/clean-contaminated group (OR = 1.9 [95%CI: 1.24
–2.91; p=0.0016] and 1.7
[95%CI: 1.16 – 2.39; p=0.004] respectively).
Table 2: Surgical morbiditiesSurgical morbidity Weighted pooled rate (%)
All 46.3% (95%CI 33.3-59.6)
Wound infection 15.9% (95%CI: 9.8-23.2) Seroma 14.2% (95%CI: 9.5 – 19.5) Haematoma 3.0% (95%CI: 1.4 – 5.3) Deep wound dehiscence 8.6% (95%CI: 6.2 – 11.2) Enterocutaneous fistula 6.5% (95%CI: 3.5 – 10.3)
Other complications reported include seroma,
haematoma, and fistula formation, and wound
dehiscence and their frequencies are summarised in
Table 2. Other infectious complications were
intra-abdominal abscess in 2.4% and another 2.7% termed
"miscellaneous".
Laxity of the repair was reported in only 8 studies (n=451) and occurred in 51 patients (10.5% [95%CI:
3.7–20.3]). All cases of laxity were reported with the use of Alloderm.
Fourteen studies with 380 patients adequately reported recurrence and/or laxity by type of closure. In
patients who underwent reinforcement, the recurrence rate was 8.1% (95%CI: 2.2–17.2) and laxity
was reported in 9.7% (95%CI: 0.1–32.1) of cases. In those whose defect was bridged, the recurrence
rate was 21.8% (95%CI: 7.5–40.9) and laxity was seen in 21.0% (95%CI: 14.8–28.0). It was not
possible to relate component separation technique or the type of anatomic graft position to any study
outcome because of inadequate reporting.
These results are similar to those found in another good
quality systematic review by Bellows (2013)
5of 60
studies with a total of 1241 participants. Overall
mortality was 4% and recurrence rate
§§was 15.2%
(range: 0–100%) at a mean follow-up period of 13.6
months. Recurrence rates by type of biological mesh
were as follows: Alloderm 23.3% at 14.6 months
average up, Surgisis 7.4% at 15.4 months
follow-up, Permacol 7.5% at 10.7 months, and Strattice 14.3%
at 10.1 months average follow-up. Surgical site
occurrence rates are summarised in Table 3 and shows
that a surgical site occurrence was reported in just over
half of cases with post-operative infection being the
most common occurrence (17%). Formation of a
haematoma or seroma was the second most common
complication and occurred in 12% of cases.
Table 3: Surgical site occurrences
Surgical site occurrences Rate (number)
Total 52.8% (491/930)
Post-operative infection 16.9% (157/930) Hematoma/seroma 12.0% (112/930)
Pain 4.7% (44/930)
Bulging/abdominal wall laxity 4.2% (39/930) Superficial dehiscence 3.8% (35/930) Fistula 3.2% (30/930) Acute mechanical failure/evisceration 2.0% (19/930) Skin necrosis 0.9% (8/930) Mesh reaction/rejection 0.9% (8/930) Poor mesh integration 0.9% (8/930) Mesh disintegration 0.5% (5/930)
Flap necrosis 0.3% (3/930)
Other 2.5% (23/930)
The other four systematic reviews
2 3 6 7are not of as good quality as Slater (2013)
4or Bellows (2013)
5but are included here for completeness.
Beale (2012)
2looked at biological meshes used in open ventral hernia repairs (including
post-traumatic reconstruction) with the express aim to make comparisons between biologic products with
respect to hernia recurrence and surgical site complications. The mean recurrence rates for Alloderm
was 21% (range 0 – 73%), Permacol 11% (range 0 – 15%), and Surgisis 8% (range 0 – 17%). The
mean 'surgical site occurrence' (which included all reported cases of haematoma, seroma, wound
infection, dehiscence, or need for graft removal for each study) for Alloderm was 31% (range 0 – 60%),
Permacol 25% (range 0
–40%), and Surgisis 40% (range 20
–41%). Statistical analysis using a
Scatterwhite
t
test found a significantly higher rate of recurrence and surgical site occurrence when
using Alloderm compared to Permacol [p=0.006 and 0.04 respectively], however, when an analysis of
variance method was used, there was no significant differences in recurrence or surgical site
occurrence [p=0.34 and 0.52 respectively]. Subgroup analysis by infection, BMI, prior hernia repair or
surgical technique was not possible making confounding by these factors a distinct possibility.
Another systematic review by Smart (2012)
7assessed the outcomes reported in the available
literature of various biological meshes used in mixed primary
***/incisional abdominal hernia repairs.
They found recurrence rates for Alloderm to be between 0% and 100%, between 0% and 41% for
Permacol, and 0% to 30% for Surgisis. Of the 45 studies (with n=1888 participants) included here,
only 8 were comparative with a mixture of comparators including other biological mesh, no
mesh/suture only, and synthetic mesh. Again the clinical heterogeneity (in study design, populations
studied, types of mesh used, surgical technique, outcome measures, and follow-up periods) present in
the papers included makes definitive conclusions difficult.
Zhong (2011)
3evaluated the use of acellular dermal matrix (ADM) for abdominal wall reconstruction
with the aim to identify factors that may affect outcomes located 30 primary studies. Recurrence rates
varied between 0% and 80% in 30 studies. Abdominal laxity was only reported in ten studies and
ranged from 0% to 83%. Delayed wound healing occurred in up to 64% of participants and
infection-related complications (including wound infection, cellulitis, and abdominal abscess) were as high as
40% The authors concluded that the "large range of recurrence rates and the lack of level I or II
evidence combined with considerable heterogeneity of design, patient populations, surgical technique,
indications, and duration of follow-up make it very difficult to come to any firm conclusions or to
perform a meaningful meta-analysis".
Hiles (2009)
6investigated the efficacy of biological grafts for any hernia repair and located 36 papers
that addressed the repair of ventral hernias
†††. The authors' found an unadjusted pooled mean
recurrence rate of 13% (range: 0 – 50%) for ventral hernia repairs over a mean follow-up duration of
12 months. The recurrence rate was 18% (range: 0 – 50%) for contaminated surgical fields and 9%
(range: 0 – 24%) for clean fields. Unadjusted pooled recurrence rates by biologic mesh can be seen in
Table 3 below.
*** this includes open abdomen, parastomal herniae, 'abdominal wall defects', and TRAM procedures (for breast reconstruction) ††† this included 'abdominal wall' & 'incisional' hernias, as well as 'open abdomen' and 'transverse rectus abdominis
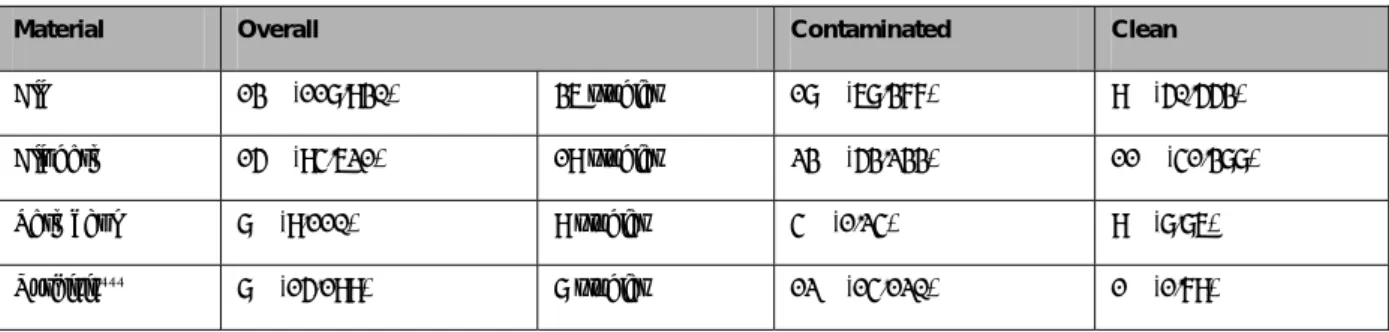
Table 3: Ventral Hernia Recurrence Rate (failures/total patients treated) by material and state of surgical field
Material Overall Contaminated Clean
All 13% (118/930) 36 studies 18% (68/377) 9% (50/553)
Alloderm 15% (94/621) 19 studies 23% (53/233) 11% (41/388)
Permacol 8% (9/110) 9 studies 4% (1/24) 9% (8/86)
Surgisis‡‡‡ 8% (15/199) 8 studies 12% (14/120) 1% (1/79)
The characteristics of all of the primary studies used in the systematic reviews in this report have been
summarised in Appendix 2.
3.
Additional Information
Synthetic mesh
A recent meta-analysis
25of open repairs of incisional hernia using synthetic mesh found an overall
infection rate of 13.6%. Meshes were removed in 26% of infected cases.
Autologous repair
Techniques using autologous tissue like the component separation technique yield similar rates of
recurrence (18.2% in one meta-analysis)
27to repairs using biologic mesh.
Expert opinion
A 2011 narrative review
28of available prosthetics for ventral hernia repair (including synthetic and
biologic materials) concluded: "The use of synthetic mesh remains an appropriate solution for most
ventral hernia repairs…Preliminary studies have demonstrated that the newer biologic prosthetics are
reasonable
options for hernia repair in contaminated fields and for large abdominal wall defects;
however, more studies need to be done before advocating the use of these biologics in other settings."
Another 2010 narrative review
29of biologic mesh for abdominal wall reconstruction concluded that
even though these meshes have offered "significant options" for surgeons (especially for complex and
contaminated cases), there are still important questions to be answered. The author cautions that
these products should be rigorously evaluated before their widespread use, in particular their exact
indications and contraindications.
Peer-review opinion
Peer-review was sought from a New Zealand expert, who gave the following advice on biological
mesh:
there is little evidence to support the routine use
the small incremental gain in infection may not be offset by their significant expense
there may be (as yet unproved) use in combination with autologous tissue repair
there may be benefit for gynaecological pelvic repair and paediatric abdominal defect repair
modern synthetic mesh is lightweight with wide pores; there is good tissue integration and
resistance to infection; when infected, salvage by vacuum-assisted closure may be possible
because of the cost, biological mesh should be restricted to specialist use until the results of
future trials emerge
4.
Limitations
There is a paucity of randomised controlled trials; the majority of studies are case series which are
highly susceptible to bias (selection, information and publication bias).
A wound classification system was not used in most primary studies.
There is a high degree of clinical heterogeneity with respect to study population, surgical technique,
length of follow-up, and outcome measures used.
The comparison of recurrence rates between synthetic and biological mesh is indirect so should be
interpreted cautiously.
5.
Conclusions
Based on six systematic reviews, the rate of hernia recurrence after the use of biological mesh for
ventral/incisional hernia repair appears to be in the order of 15% (range 0% to 100%) over a follow-up
period of about 1 year. This is similar to the recurrence rate observed for synthetic mesh (reported to
be between 17% and 30% in a long-term follow-up study)
26.
Infection is the most common complication reported (approximately 16-17%) and required removal of
the biological mesh in 5% of cases (compared to the removal of synthetic mesh because of infection in
26% of cases). Infection rate with synthetic meshes was 14% in a recent meta-analysis.
In summary, even considering the limitations of the research evidence, complication rates (in particular
rates of recurrence and infection) appear to be similar when comparing biological and synthetic
meshes. There is also limited indirect evidence that if the surgical site becomes infected the biological
mesh may not have to be removed as often as synthetic mesh.
6.
Appendix 1: Evidence Tables
(in alphabetical order)
Reference and study design Studies Intervention/comparison Outcome measure Results/effect size Conclusions
Beale et al. (2012). "The role of biologic mesh in abdominal wall reconstruction: a systematic review of the current literature."
American Journal of Surgery 204(4): 510-7.
USA
Included studies: Bellows 2007, Blatnik 2008, Buinewicz 2004, Candage 2008, Catena 2007, de Moya 2008, Diaz 2006, Diaz 2009, Espinosa-de-los-Monteros 2007, Gupta 2006, Gupta 2006, Helton 2005, Holton 2005, Hsu 2009, Jin 2007, Kim 2006, Ko 2009, Kolker 2005, Lee 2009, Limpert 2009, Lin 2009, Lipman 2008, Misra 2008, Nemeth 2009, Parker 2006, Patton 2007, Schuster 2006, Scott 2006, Shaikh 2007, Singh 2008, Trevino 2006 Level of evidence: 1- Number of studies: N=29 (Permacol 4, Surgisis 3, Veritas 1, and Alloderm 23)§§§
Total number of patients in the studies: n=1257 Inclusion criteria: original studies, age, ≥18 yr, human, English language, post-traumatic reconstruction, ventral hernia, incisional hernia
Exclusion criteria: parastomal hernia, inguinal hernia, laparoscopic hernia repair, hiatal hernia, case reports, books
Databases used: Pubmed/Medline Description of the
methodological assessment of studies:. Assigned a level of evidence based on the Oxford Centre for Evidence-Based Medicine i.e. based on study design only
Fixed or variable effects: not reported
Heterogeneity: +++ (in design, data collection, patient population, surgical technique, & length of follow-up)
Intervention: Ventral/incisional hernia repair or post-traumatic reconstruction of abdominal wall using biological meshes Length of treatment: mean follow-up period was 12.6 months (Alloderm), 15 months (Permacol), and 17.7 months (Surgisis)
Comparator (N=4): one compared Alloderm vs. Prolene mesh [Ko 2009]; another, two implantation techniques using same biological mesh [Jin 2007]; one prospective study investigating Permacol only [Shaikh 2007]; and the last prospective compared Alloderm with Surgisis [Gupta 2006].
Co-interventions: not reported
Hernia recurrence (mean, %)
Surgical site occurrence [SSO] (included haematoma, seroma, wound infection, dehiscence, or need for graft removal) Overall: 19% Alloderm: 21% Permacol: 11% Surgisis: 8% Overall: 30% Alloderm: 31% Permacol 25% Surgisis 40% No significant difference in recurrence rates or SSO among the 3 meshes using analysis of variance [p=0.34, p=0.52 respectively] Recurrence rates and SSO were significantly higher when comparing Alloderm with Permacol when Scatterwhite t-test used [p=0.006 and 0.04 respectively]
"Biologic mesh does play a beneficial role in abdominal wall reconstruction although allograft acellular dermal matrix [Alloderm] does have a higher recurrence rate as compared with xenograft products, which limits its current role in hernia repair." Biological mesh have similar recurrence rates as compared to synthetic mesh
Not able to control for confounding factors that affect recurrence and SSO e.g. infection, BMI, prior hernia repair, and repair technique
Study type: Systematic review
Comments: Highly susceptible to bias due to a limited search (only 1 database) and no formal appraisal of study quality (i.e. a level of evidence was assigned according to study design alone). The results were combined even though there was considerable heterogeneity in study design, participant numbers, and follow-up periods. Even though the heterogeneity of studies would preclude combining results, the recurrence estimates may be useful as a very rough "ballpark" figure.
Reference and study design
Participants Intervention/comparison Outcome measure Results/effect size Conclusions
Bellows (2013).
"Repair of incisional hernias with biological prosthesis: a systematic review of current evidence."
American Journal of Surgery 205(1): 85-101.
USA
Studies included: Adedeji 2002, Alaedeen 2007, Asham 2006, Awad 2010, Bachman 2009, Baillie 2007, Bluebond-Langner 2008, Boutros 2010, Brewer 2011, Butler 2011, Canda 2009, Candage 2008, Catena 2007, Cavallaro 2010, Chavarriaga 2010, Chuo 2008, Cobb 2005, Coyle 2010, Dorafshar 2009, Edelman 2010, Eid 2004, Espinosa-de-los-Monteros 2007, Gentile 2011 Gupta 2006, Helton 2005, Hirsch 2004, Holton 2005, Hsu 2009, Jin 2007, Kim 2006, Ko 2009, Kolker 2005, Lee 2009, Limpert 2009, Lin 2009, Lipman 2007, Liyanage 2006, Martins 2010, McDonald 2005, Misra 2008, Morrison 2009, Nemeth 2009, O'Brien 2011, Parker 2006, Parra 2010, Patton 2007, Pomahac 2010, Saettele 2006, Sailes 2011, Satterwhite 2011, Schuster 2006, Shaikh 2007, Shaihk 2011, Singh 2008, Tong 2011, Trevino 2006, Tung 2006, Walker 2009, Wietfeldt 2009, Wotton 2009 Level of evidence: 1++ Number of studies: N=60 Total number of patients in the studies: n=1241 Inclusion criteria: studies of patients with incisional/ventral hernia repaired with
commercially available biological prosthesis; the outcomes evaluated were reported separately for each type of hernia repair; and the patients were adult (age, ≥18 yr) subjects and follow-up data were available; all types of clinical study design; English language
Exclusion criteria: congenital abdominal wall defects, and femoral, inguinal, hiatal, diaphragmatic, parastomal, or sacral hernias; animal studies, reviews, and articles that investigated the application of biological prosthesis for transverse rectus abdominis
myocutaneous (TRAM) flap repairs or open abdomens from trauma patients. Databases used: MEDLINE, EMBASE, & Cochrane Register of Controlled Trials to Oct 2011 using subject headings and text words Description of the
methodological assessment of studies: used a modified version of the methodological index for non-randomised studies [MINORS]; studies that scored 0 on items 2, 5, or 7, or had a total score <9 were considered to be of poor quality
Fixed or variable effects: not applicable
Heterogeneity: +++ (both statistical and clinical i.e. populations studied, repair
Description of
treatment/procedure studied: Biological grafts for
incisional/ventral hernia repair Length of treatment (mean): 13.6 months (range 0.17 – 60) Description of comparison: three retrospective comparative studies; Cobb 2005 polypropylene/expanded polytetrafluoroethylene mesh; Espinoso-de-los-Monteros 2007 no human cadaveric acellular dermis; Ko 2009 soft polypropylene mesh
Co-interventions: none reported
Overall mortality: Overall weighted average recurrence rate:
Weighted average recurrence rate:
Alloderm Surgisis Permacol Strattice Surgical site occurrences (unweighted):
Overall rate
Postoperative infection Hematoma/seroma Pain
Bulging/abdominal wall laxity Superficial dehiscence Fistula Acute mechanical failure/evisceration Skin necrosis Mesh reaction/rejection Poor mesh integration Mesh disintegration Flap necrosis Other 4.0% (48/1212) 15.2% (range 0-100%) 23.2% (170/732) 7.4% (10/135) 7.5% (12/160) 14.3% (16/112) 52.8% (491/930) 16.9% (157/930) 12.0% (112/930) 4.7% (44/930) 4.2% (39/930) 3.8% (35/930) 3.2% (30/930) 2.0% (19/930) 0.9% (8/930) 0.9% (8/930) 0.9% (8/930) 0.5% (5/930) 0.3% (3/930) 2.5% (23/930) There is an insufficient level of high-quality evidence in the literature on the value of
bioprosthetics for incisional hernia repair.
techniques, outcome measures, follow-up times) Study type: Systematic review
Comments: Well conducted systematic review. Searched three databases. Narrow focus i.e. incisional ventral hernia repairs. Methodological assessment well done and appropriate. Weighted variance approach used to calculate pooled recurrence rate. Appropriately cautious conclusion.
Reference and study
design Studies Intervention/comparison Outcome measure Results/effect size Conclusions
Hiles, M., et al. (2009)6
"Are biologic grafts effective for hernia repair? A systematic review of the literature."
Surgical Innovation 16 (1): 26-37.
USA
Included studies: Adedeji 2002, Alaedeen 2007, Armellino 2006, Bellows 2007, Bluebond-Langner 2008, Buinewicz 2004, Butler 2005, Catena 2005, Catena 2007, Cobb 2005, Diaz 2006, Eid 2004, Espinosa-de-los- Monteros 2007, Ferguson 2007, Franklin 2004, Glasberg 2006, Gu 2008, Gupta 2006, Helton 2005, Hirsch 2004, Holton 2005, Jin 2007, Kim 2006, Kolker 2005, Liyanage 2006, Misra 2008, Napolitano 2004, Parker 2006, Patton 2007, Pentlow 2008, Saettele 2007, Schuster 2006, Scott 2006, Shaikh 2007, Treviño 2006, Tung 2006, Ueno 2004
Number of studies for ventral hernia****: N = 36 (8 Surgisis;
19 Alloderm; 9 Permacol) Total number of patients (n = 930)
Inclusion criteria: Children and adults with ventral, para-oesophageal, hiatus, inguinal, diaphragmatic, gastroschisis, or congentital hernia; "biologic" grafts including human or animal tissues (chemically cross-linked or not) of submucosal,
pericardial or dermal sources; available for purchase in US; published in peer-reviewed publications for hernia repair in humans; subsequent studies from same authors with longer follow-up or additional subjects
Exclusion criteria: Preclinical studies; pelvic
floor/anterior/posterior repairs; number of recurrences or duration of study could not be determined; studies prior to studies from same authors with longer follow-up or
Intervention studied: Surgical hernia (ventral,
paraoesophageal, hiatus, inguinal, diaphragmatic, gastroschisis, or congenital) repair using biological grafts Mean length of follow-up (months):
Surgisis 19 mo
Alloderm 12 mo
Permacol 14 mo
Description of comparison: only one study was a direct comparison between different bio meshes for inguinal hernia Co-interventions: none reported
Recurrence†††† (range) Average longevity‡‡‡‡ (months) Ventral hernias: Overall: 13% (0-50%) - clean field 9% (0-24%) - infected 18% (0-30%) By material (clean vs. infected): - Surgisis 8% (1% vs. 12%) - Alloderm 15% (11% vs. 23%) -Permacol 8% (9% vs. 4%) Overall: 13.2 months - clean 12.8 - infected 13.5 By material (clean vs. infected): - Surgisis§§§§ 17.9 (17.5 vs. 18.1 mo) - Alloderm***** 13.4 (11.8 vs. 11.0 mo) - Permacol††††† 13.3 (12.9 vs. 14.8 mo)
From 36 studies of various designs, a recurrence rate of approximately 13% and longevity of around 13 months was derived. Recurrence rate varied from 0% to 50%
Overall the recurrence rate in infected surgical fields was approximately 18% (range 0-50%) compared to 9% (range 0-24%) in clean fields. There are significant limitations to the interpretation of these findings as there are considerable validity issues present i.e.
selection/publication bias
**** includes abdominal wall & incisional hernia, TRAM flap, or open abdomen
†††† calculated by summing the total number of cases of recurrence and dividing by all subjects reported across all papers in the group, regardless of hernia type
‡‡‡‡ calculated by summing the products of number of patients in each study & the mean follow-up time in months (i.e. total patient-months) and dividing this sum by the total number of subjects,
regardless of hernia type
§§§§ 6 studies ***** 17 studies †††††
Level of evidence: 1-
additional subjects
Databases used: MEDLINE to March 31 2008
Description of the
methodological assessment of studies: each paper was assigned a level of evidence based on study design alone
Fixed or variable effects: not addressed
Heterogeneity: +++ Study type: Systematic review
Comments: Highly susceptible to bias due to a limited search (only 1 database) and no formal appraisal of study quality (i.e. a level of evidence was assigned according to study design alone). Short duration of follow-up and high rates of loss-to-follow-up very likely underestimates recurrence rate. The results were combined even though there was considerable heterogeneity in study design, participant numbers, and follow-up periods. However, this was still included as a 'ball-park' estimate of recurrence but will be given less weight than other, better quality studies. Limited outcomes reported i.e. recurrence & longevity only, due to inconsistency in reporting of other outcomes e.g. seroma, wound infection.
Reference and study design
Participants Intervention/comparison Outcome measure Results/effect size Conclusions
Slater (2013).
"Biologic grafts for ventral hernia repair: a systematic review."
American Journal of Surgery 205(2): 220-30.
The Netherlands
Studies included: Alaedeen 2007, Bellows 2007, Boehmler 2009, Buinewicz 2004, Candage 2008, Catena 2007, Chavarriaga 2010, Connolly 2008, Diaz 2009, Glasberg 2006, Gupta 2006, Helton 2005, Hsu 2009, Jin 2007, Kim 2006, Kolker 2005, Lee 2009, Lin 2009, Loganathan 2010, Maurice 2009, Misra 2008, Parker 2006, Patton 2007, Scott 2006, Shaikh 2007, Taner 2009, Ueno 2004 Level of evidence: 1++ Number of studies: N=27 (N=17 for primary outcome; N=25 for secondary outcomes)
Total number of patients in the studies: n= 531 for recurrence; n= 879 for mortality; n=1125 for surgical morbidity; n=451 for laxity Inclusion criteria: all adults over the age of 18 yrs who underwent definitive repair of all types of ventral hernia (i.e. incisional, epigastric, Spiegel [lateral ventral], and [para-]umbilical) as well as reconstruction of large defects created during surgery (e.g. TRAM flap reconstruction , and tumour resection) or trauma; the biological graft had to be the sole graft material used; biological grafts placed as reinforcement or bridging material were also included.
Description of
treatment/procedure studied: Biological grafts for ventral hernia repair (including incisional, epigastric, Spiegel [lateral ventral], and [para-]umbilical hernias, reconstruction of large defects created during surgery (e.g. TRAM flap reconstruction , and tumour resection) or trauma
Length of treatment (mean): 18 months (range 12 – 29) Description of comparison (placebo): none as all case series
Co-interventions: none reported
Primary outcome: Overall recurrence rate Recurrence by wound class: - Clean/clean-contaminated - Contaminated/dirty - "Complicated"
Recurrence by surgical technique: - Reinforcement
- Bridged
Secondary outcomes: Mortality
Overall surgical morbidity rate Surgical morbidity by wound class: - Clean/clean-contaminated - Contaminated/dirty - "Complicated"
Surgical morbidity by biologic graft - Alloderm
- Permacol - Surgisis
Weighted pooled proportion, % (95% confidence interval) 13.8% (7.6 – 21.3) 2.9% ( 0.2 – 8.3) 23.1% (11.3 – 37.6) 19.4% (11.4 – 29.0) 8.1% (2.2 – 17.2) 21.8% (7.5 – 40.9) 36 (4.1%) 46.3% (33.3 – 59.6) 32.5% (9.2 – 61.9) 52.4% (23.8 – 80.2) 50.3% (33.4 – 67.2) 46.5% (29.7–63.7) 28.7% (17.2–41.8) 45.7% (36.2–55.3)
No randomized trials are available to properly evaluate biologic grafts for ventral hernia repair. The current evidence suggests that biologic grafts perform similarly to other surgical options. Biologic grafts are associated with a high salvage rate when faced with infection.
Exclusion criteria: studies with less than 7 patients were excluded
Databases used: MEDLINE, Embase, Cochrane, Central Register of Controlled Trials to Nov 2010
Description of the
methodological assessment of studies: used a modified version of the methodological index for non-randomised studies [MINORS]; studies that scored 0 on items 2, 5, or 7, (see below in comments) or had a total score <8 were excluded from analysis of the primary outcome.
Fixed or variable effects: random effects model used Heterogeneity: Recurrence in clean/clean-contaminated hernia repairs I2 = 68%;
contaminated/dirty I2 = 53%;
"complicated" I2 = 64% i.e. all
show moderate heterogeneity
Overall wound infection rate Infection rate by wound class: - Clean/clean-contaminated - Contaminated/dirty - "Complicated"
Infection rate by biologic graft: - Alloderm
- Permacol - Surgisis
Overall seroma formation rate Seroma formation by biologic graft - Alloderm
- Permacol - Surgisis Haematoma
Deep wound dehiscence Enterocutaneous fistula Overall laxity rate
Laxity by surgical technique: - Reinforcement - Bridged 15.9% (9.8 – 23.3) 4.7% (0.1–20.6) 19.3% (6.8–36.2) 22.3% (15.3–13.3) 17.1% (9.3–6.7) 6.1% (1.7–3.0) 14.9% (0.4–44.8) 14.2% (9.5 – 19.5) 13.2% (8.8–18.4) 7.6% (1.9–16.4) 26.0% (.3–59.2) 3.0% (1.4 – 5.3) 8.6% (6.2 – 11.2) 6.5% (3.5 – 10.3) 10.5% (3.7 – 20.3) 9.7% (0.1 – 32.1) 21.0% (14.8 – 28.0) Study type: Systematic review
Comments: A well conducted systematic review and meta-analysis. There was an extensive and robust search, with explicit, clear inclusion & exclusion criteria. The methodological assessment was well done and appropriate. A weighted pooled estimate of rates was conducted. Heterogeneity was formally measured (I2). No exclusion table. Median modified MINORS score was 11/14 (range
8-14)
MINORS: Score of 0 on Item 2 = less than 5 patients included; on item 5 = surgical methods incomplete; on item 7 = endpoints not reported
Reference and study design Studies Intervention/comparison Outcome measure Results
(comparative studies only) Conclusions
Smart, et al. (2012). "Biological meshes: a review of their use in abdominal wall hernia repairs."
Surgeon Journal of the Royal Colleges of Surgeons of Edinburgh & Ireland 10(3): 159-71.
Number of studies: N = 45 Total number of patients in the studies: n = 1888 Inclusion criteria: Randomized controlled trials (RCTs), prospective controlled or uncontrolled studies and retrospective cohort studies of
Intervention: Ventral or incisional hernia repair using biological mesh
Length of treatment: variable (range: 0.3 to 68 months) Comparison (N=8): 8 studies involved a comparator(s), either other meshes (synthetic
Recurrence rate (%) Gupta (n=74) ventral hernia
Alloderm 24.2% Surgisis 0% Espinosa-de-los-Monteros (n=76) incisional hernia Alloderm 5% no Alloderm 20%
Sailes (n=545) abdominal wall
There is only sufficient data about three meshes, Alloderm, Permacol, & Surgisis.
The absence of comparative data doesn't allow for definitive conclusions. However, the available
UK
Included studies: Alaedeen 2007, Armellino 2006, Aycock 2007, Bellows 2007, Blatnik 2008, Brewer 2011, Buinewicz 2004, Candage 2008, Catena 2007, Cobb 2005, Connolly 2008, De Moya 2008 , Diaz 2006, Eid 2004, Espinosa-de-los-Monteros 2007, Franklin 2002, Franklin 2004, Franklin 2008, Gupta 2006, Helton 2005, Holton 2005, Hsu 2009, Jin 2007, Kim 2006, Ko 2009, Kolker 2005, Lin 2009, Loganathan 2010, Maurice 2009, Misra 2008, Parker 2006, Patton 2007, Sailes 2010, Schuster 2006, Scott 2006, Shaikh 2007, Smart 2011, Ueno 2004 Level of evidence: 1+
primary (aka ventral) & incisional abdominal wall ‡‡‡‡‡
repairs using biological mesh in English to the end of September 2011
Exclusion criteria: Reviews, case reports,
congress/meeting abstracts; inguinal hernia repairs Databases used: PubMed, MEDLINE, The Cochrane library, clinical trials registers and articles which came from the reference list of other articles.
Description of the
methodological assessment of studies: criteria published in 2009 by the Oxford Centre for Evidence-Based Medicine Levels of Evidence i.e. based on study design alone
Fixed or variable effects: not applicable due to lack of RCTs and quality of data Heterogeneity: +++ (in design, data collection, patient population, surgical technique, & length of follow-up)
or biologic), or suture with no mesh
Co-interventions: not reported
reconstruction Alloderm 19% Permacol 7.7% Synthetic 25.6% Component separation only 12% Brewer (n= 84) ventral or incisional hernia in transplant recipients i.e. immunosuppressed
Alloderm 23.5%
Synthetic 76.9%
Primary repair 36.0% Cobb (n=139) laparoscopic ventral hernia repair
Permacol 6.6%
PP/ePTFE 1.2%
Connolly (n=61) intestinal fistula and open abdomen
NB: these are rates of recurrence of fistulae not recurrence of hernia
Suture 0%
Permacol 41%
Synthetic 76%
Eid (n=71) ventral hernia after gastric bypass
Surgisis 0%
Primary repair 22%
Ko (n=54) midline incisional hernia with component separation technique
Alloderm 46%
polypropylene 11%
studies evaluated in this review suggest that Alloderm has the highest recurrence rates while Surgisis performs reasonably well in clean or clean-contaminated fields, and Permacol had the lowest failure rate and longest time to failure, particularly in
contaminated fields.
Study type: Systematic review
Comments: Reasonably conducted SR with appropriately cautious interpretation of findings. Again, a level of evidence system was used based on study design, not methodological quality. Significant heterogeneity in design, data collection, patient population, surgical technique, & length of follow-up. Comparative studies only are reported in this evidence table – see the paper or report for all results.
PP/ePTFE, composite polypropylene and expanded polytetrafluoroethylene mesh
Reference and study design Studies Intervention/comparison Outcome measure Results/effect size Conclusions
Zhong, et al. (2011). Number of studies: N = 30 Intervention: Abdominal wall Hernia recurrence (follow-up) Cobb 2005: There is a deficiency of data
"Outcomes after abdominal wall reconstruction using acellular dermal matrix: A systematic review." Journal of Plastic,
Reconstructive and Aesthetic Surgery 64 (12): 1562-1571. Canada/USA
Included studies: Bellows 2007, Blatnik 2008, Bluebond-Langer 2008, Buinewicz 2004, Butler 2005, Candage 2008, Cobb 2005, de Moya 2008, Diaz 2006, Diaz 2009, Espinosa-de-los-Monteros 2007, Gu 2008, Gupta 2006, Guy 2003, Hsu 2009, Jin 2007, Kim 2006, Kolker 2005, Lee 2009, Lin 2009, Maurice 2009, Nemeth 2009, Parker 2006, Patton 2007, Schuster 2006, Scott 2006, Shinall 2010, Singh 2008, Tang 2009, Weitfeldt 2009 Level of evidence: 1+
(only 4 comparative studies located – all the rest were case series)
Total number of patients in the studies: 1351
Inclusion criteria: prospective or retrospective human studies in English using the search terms 'dermal matrix' and 'abdomen' or 'hernia' Exclusion criteria: animal studies, case reports, review/articles that only dealt with ADM for repair of congenital abdominal wall defects, hiatal, parastomal or inguinal hernias and enterocutaneous fistulae. Databases used: MEDLINE as of 31 October 2010; references of included studies were screened to identify other citations not captured in MEDLINE search
Description of the
methodological assessment of studies: each paper was assigned a level of evidence based on study design
Fixed or variable effects: not applicable
Heterogeneity: +++ (in design, data collection, patient population, surgical technique, & length of follow-up)
reconstruction using acellular dermal matrix (ADM) Length of treatment: 0-53 months (unreported in 2 studies)
Comparison (N=4): 2 compared HADM vs. other materials (including PSISM & polyprop mesh); another, PADM vs. CPEPM; and one compared HADM used in a bridged repair vs. reinforced repair with components separation
Co-interventions: none reported
Abdominal wall laxity (N = 10)
Other complications PADM vs. composite 6% vs. 1% ; NS (14m vs. 31m) Gupta 2006: HADM vs. PSISM 24% vs. 0% ; no stat analysis (18 m vs. 29m) Espinosa-de-los-Monteros 2007:
HADM vs. polyprop mesh 5% vs. 20% ; sig (15m vs. 13m) Jin 2007:
HADM bridged vs. reinforced CS
80% vs. 20% ; sig (24m vs. 13m)
All other studies (case series): range 0-80% (F/U range: 3m to 43m) 0% recurrence in 8 studies Range 0-83% Delayed healing in up to 64% Infection-related up to 40% Seroma 27%
derived from high level-of-evidence studies. The existing studies fail to provide conclusive evidence about whether ADM may be more useful i.e. less hernia recurrence, than synthetic materials, or what positioning of ADM, suture type, etc may lead to better long-term outcomes.
Study type: Systematic review
Comments: Susceptible to bias because only one database searched (Medline) and no formal appraisal of study quality (only assignment to level of evidence based on study design). However, mitigated by authors not attempting a meta-analysis. Limited number (4) of comparative studies and studies investigating porcine or bovine ADM. Significant heterogeneity.
ADM, acellular dermal matrix; HADM, human acellular dermal matrix; PADM, porcine acellular dermal matrix; PSISM, porcine small intestinal submucosa material; CPEPM, composite polypropylene and expanded polytetrafluoroethylene mesh; polyprop, polypropylene; CS, component separation; sig, statistically significant; NS, not statistically significant; m. months; vs., versus
7.
Appendix 2: Summary of primary studies cited in the systematic reviews
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence Contaminated
field?
Adedeji 2002 Permacol Abdominal wall 1 12 months 0% Y
Alaedeen 2007 Alloderm Ventral (2 fascial bridge, 8 component separation) 10 Mean 14 months (2-68) 20% Y
Armellino 2006 Permacol Incisional 6 3-24 months 0% Y
Asham 2006 Alloderm Abdominal wall reconstruction (inlay) after small bowel
transplantation
1 24 months 0% NS
Awad 2010 Strattice Infected or contaminated incisional hernia 80 12 months 21% Y
Aycock 2007 Alloderm Parastomal hernia 11 9.4 months (1-21) 27% N
Surgisis Ventral hernia (component separation) 2 Median 6 months (0.25-9) 0% NS
Bachman 2009
FlexHD Ventral hernia (component separation) 2 Median 6 months (0.25-9) 0% NS
Permacol Complex abdominal wall reconstruction (bridged) 1 5 months 0% NS
Baillie 2007
Alloderm Complex abdominal wall reconstruction (bridged) 1 5 months 0% NS
Alloderm Abdominal wall reconstruction (underlay) 1 Median 9 months (2-16) 0% N
Bellows 2007
Abdominal wall reconstruction (underlay) 20 Median 9 months (2-16) 30% Y
Blatnik 2008 Alloderm Abdominal wall reconstruction / complex ventral hernias
(interposition)
11 24 months (18-37) 80% Y
Alloderm Ventral 7 Average 9 months (1-14) 0% N
Bluebond-Langner 2008
Ventral 20 Average 9 months (1-14) 35% Y
Boehmler 2009 Alloderm Ventral hernia 31 20.4 months 0% NS
Boutros 2010 Surgisis Abdominal wall closure after intrperitoneal chemotherapy
(underlay)
3 Mean 6.3 months 0% NS
Alloderm Ventral/incisional hernia in transplant patients
(immunosuppressed)
34 26 months 24% NS
Synthetic mesh Ventral/incisional hernia in transplant patients (immunosuppressed)
26 26 months 77% NS
Brewer 2011
Primary repair Ventral/incisional hernia in transplant patients (immunosuppressed)
Contaminated
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence
field?
Alloderm Incisional or TRAM (overlay; multilayer interposition) 36 Mean 20 months (8-32) 6% N
Buinewicz 2004
Incisional or TRAM§§§§§ (overlay; multilayer interposition) 8 Mean 20 months (8-32) 0% Y
Butler 2005 Alloderm Abdominal/pelvic/chest wall reconstruction after tumour
resection
13 Mean 6.4 months (2-13) 0% mixed******
Butler 2011 Strattice Ventral hernias in cancer patients (bridged-inlay;
component separation + reinforced-inlay)
38 Mean 12.4 months 3% NS
Candage 2008 Alloderm Abdominal wall hernia†††††† / (20 bridged/underlay, 26
reinforced/ overlay/underlay/sandwich)
46 12 months (1-39) 30%‡‡‡‡‡‡ Y
Catena 2007 Permacol Incisional (underlay) 7 Mean 11 months (7-18) 0% Y
Cavallaro 2010 Tutomesh Abdominal wall reconstruction (retrorectus) 2 Mean 54 months (48-60) 0% Y
Chavarriaga 2010 Collamend Abdominal wall reconstruction (reinforced/inlay, overlay,
interposition +/- component separation)
18 Mean 7.3 months (2-14) 44% NS
Chuo 2008 Permacol Closure of abdominal wound dehiscence with exposed
bowel (bridged-underlay)
1 15 months 0% NS
Permacol Ventral/incisional & recurrent hernia / laparoscopic ventral repair
55 Average 14 months 6% N
Cobb 2005
Composite§§§§§§
mesh
Ventral/incisional & recurrent hernia / laparoscopic ventral repair
84 31 months******* 1% N
Permacol Open abdomen and intestinal fistula 12 Median 29 months 41% Y
Synthetic mesh Open abdomen and intestinal fistula 17 Median 29 months 76% Y
Connolly 2008
Suture repair Open abdomen and intestinal fistula 34 Median 29 months 0% Y
Coyle 2010 Permacol Strangulated umbilical hernia (underlay) 1 1.4 months 0% Y
de Moya 2008 Alloderm Open abdomen / large open abdominal wounds (underlay) 10 12 months (1-12) 10% NS
Diaz 2006 Alloderm Ventral (inlay, interposition, onlay, component separation) 64 9 months 16% N
§§§§§ transverse rectus abdominis myocutaneous flap (breast reconstruction surgery)
****** 7 had bacterial contamination of surgical site
†††††† 34 of 46 were complex ventral hernia
‡‡‡‡‡‡ "6 recurrent hernias and 8 eventration of the prosthesis" [abstract]
§§§§§§ composite polypropylene/expanded polytetrafluorethylene mesh
Contaminated
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence
field?
Ventral (inlay, interposition, onlay, component separation) 11 9 months 18% Y
Diaz 2009 Alloderm Complex ventral hernia repair (inlay, onlay, interposition,
component separation)
240 9 months (0-38) 17% NS
Dorafshar 2009 Alloderm Post-bariatric laxity (onlay) 1 24 months 0% NS
Edelman 2010 Surgisis Umbilical hernia 16 Mean 12 months 6% NS
Surgisis Ventral hernia 12 Mean 13 months (7-18) 0% N
Eid 2004
Primary repair Ventral hernia 59 Mean 20 months 22% N
Alloderm Incisional (recurrent) / component separation + overlay 37 Median 15 months 5% N
Espinosa-de-los- Monteros 2007
Polypropylene Incisional (recurrent) / polypropylene + overlay 39 Median 13 months 20% NS
Ferguson 2007 Surgisis TRAM flap 1 14 months 0% N
Franklin 2002 Surgisis Incisional/ventral or inguinal hernia 25 Median 15 months (1-20) 0% Y
Franklin 2004 Surgisis Incisional/ventral or inguinal hernia 58
(including 25 from the 2002
study)
Mean 19 months (1-30)††††††† 0% Y
Franklin 2008 Surgisis Incisional, inguinal and umbilical hernia 116 Mean 52 ± 21 months 5% Y
Gentile 2011 Permacol Complex abdominal hernia repair (bridged) 1 12 months 0% NS
Glasberg 2006 Alloderm TRAM flaps 54 Mean 18.5 months (9-30) 0% N
Gu 2008 Alloderm Abdominal wall reconstruction (underlay + omental flap) 3 3 months (1-11) 0% N
Surgisis Ventral hernia repair 38 Mean 29 months 0% N
Surgisis Ventral hernia repair 3 Mean 29 months 0% Y
Gupta 2006
Alloderm Ventral hernia repair 33 Mean 18 months 24% N
Guy 2003 Alloderm Abdominal compartment syndrome / interposition 9 20 months (11-30) 11% ?
Surgisis Ventral hernia repair 22 Median 14 months (2-29) 5% N
Helton 2005
Surgisis Ventral hernia repair 31 Median 14 months (2-29) 26% Y
Contaminated
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence
field?
Hirsch 2004 Alloderm Abdominal wall 1 9 months 0% Y
Holton 2005 Alloderm Abdominal wall reconstruction (various) 46 6 months (0.25-32) 11% N
Hsu 2009 Permacol Abdominal wall reconstruction (underlay) 28 16 months (10-23) 11% N
Alloderm Ventral / bridged (interposition/underlay) 11 24 months (18-37) 80% Y
Jin 2007
Ventral / reinforced component separation (overlay/underlay/sandwich)
26 13 months (6-13) 20% Y
Kim 2006 Alloderm Ventral / high risk / recurrent (underlay + component
separation
29 6 months (0-16) 10% Y‡‡‡‡‡‡‡
Alloderm Midline incisional hernia (component separation) 26 17 months 46% mixed
Ko 2009
Polypropylene Midline incisional hernia (component separation) 28 16 months 11% mixed
Kolker 2005 Alloderm Incisional and recurrent (multilayer with underlay, overlay,
component separation)
16 Mean 16 months (9-23) 0% mixed§§§§§§§
Lee 2009 Alloderm Abdominal wall reconstruction (underlay) 68 15 month 27% ?
Limpert 2009 Veritas Abdominal wall repair 30 Mean 22 months (4-37) 17% NS
Lin 2009 Alloderm Abdominal wall reconstruction (underlay, overlay,
interposition)
144 5 months (0-23) 27% Y
Lipman 2008 Alloderm Massive incisional hernias (serial excision of Gore-Tex
mesh with reapproximation of fascia with Alloderm with component separation)
8 Mean 10 months 13% NS
Liyanage 2006 Permacol Ventral 1 12 months 0% Y
Loganathan 2010 Permacol Complicated hernia 15 Median 12 months 13% Y
Martins 2010 Alloderm Richter's hernia repair 1 6 months 0% NS
Maurice 2009 Alloderm Abdominal wall reconstruction (underlay, overlay,
interposition)
63 7 months (0-24) 41% NS
McDonald 2005 Alloderm Recurrent hernia (interposition retorectus) 5 Mean 1.9 months (0.2-8) 60% Y
Misra 2008 Alloderm Ventral/incisional hernias 70 NS (up to 1 yr) 20% mixed********
‡‡‡‡‡‡‡ 4/29 were clean
§§§§§§§ 9/16 had infections
Contaminated
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence
field?
Morrison 2009 Alloderm Massive ventral hernia 1 60 months 100% NS
Napolitano 2004 Surgisis Incisional 6 Mean 24 months (3-36) 0% N
Nemeth 2009 Alloderm Complex torso reconstruction after tumour resection
(inlay)
13 43 months (41-53) 8% (20% of
surviving patients)
Y
O'Brien 2011 Permacol Recurrent ventral hernia (onlay) 1 24 months 0% NS
Parker 2006 Permacol Abdominal wall / complicated fascial defects (underlay) 9 Mean 18 months 11% Y
Parra 2010 Strattice Potentially contaminated ventral hernia 3 Mean 6 months 0% Y
Patton 2007 Alloderm Abdominal wall reconstruction (interposition, onlay) 67 Mean 10 months (0-38) 18% Y
Pentlow 2008 Permacol Abdominal wall 5 36 months (n=1), others less 20.0 N
Pomahac 2010 XenMatrix Abdominal wall reconstruction (underlay +/- interposition;
approx midline +/- component separation + reinforced-underlay)
16 Mean 16.5 months 6% NS
Saettele 2007 Permacol Incisional 1 14 months 0% N
No mesh Abdominal wall reconstruction (component separation
only)
225 12% NS Y
Alloderm Abdominal wall reconstruction 100 19% NS Y
Permacol Abdominal wall reconstruction 13 8% NS Y
Sailes 2010
Synthetic mesh Abdominal wall reconstruction 207 26% NS Y
Satterwhite 2012 Permacol Abdominal wall reconstruction (approx midline +/-
component separation + reinforced sandwich)
19 Mean 11 months (1-33) 0% NS
Schuster 2006 Alloderm Ventral / contaminated abdominal wall fascial defects
(interposition)
18 Mean 9 months (5-27) 50% Y
Scott 2006 Alloderm Open abdomen (underlay) 37 1 month (longest 36) 0% N
Shaikh 2007 Permacol Acute & chronic abdominal wall defect (underlay) 20 Median 18 months (6-36) 15% NS
Shaikh 2011 Permacol Ventral hernia recurrence (onlay) 1 6 months 100%
Shinall 2010 Alloderm Open abdomen paediatric patients (interposition) 5 13 months (3-21) 0% NS
Study/yr Material Condition/procedure Cases Follow-up (range) Recurrence Contaminated field? (interposition)
Smart 2011 Permacol Parastomal hernia 27 16.6 months 56% Y
Taner 2009 Alloderm Enterocutaneous fistula 11 12 months (+/-4) 0% Y
Abdominal wall reconstruction after tumour resection (interposition)
6 4 months (3-21) 0% NS
Tang 2009 Alloderm
Abdominal wall reconstruction after tumour resection (interposition + TFL, interposition + omental flap)
21 4 months (3-21) 0% NS
Tong 2011 Surgisis Ruptured umbilical hernia 1 12 months 0% NS
Treviño 2006 Surgisis Incisional hernia repair (sandwich) 5 Median 10 months (3-12) 0% Y
Tung 2006 Alloderm Abdominal wall 1 24 months 0% Y
Ueno 2004 Surgisis Ventral or inguinal 20 Mean 16 months 30% Y
Walker Permacol Abdominal wall reconstruction 1 3 months 0% NS
Weitfeldt 2009 Bovine ADM Abdominal wall reconstruction (interposition) 5 10 months (9-17) 20% NS
Wotton 2009 Permacol Abdominal wall reconstruction 1 4 months 0% NS