organic papers
Acta Cryst.(2005). E61, o2663–o2664 doi:10.1107/S1600536805022968 Cuiet al. C
25H20FN3O2S
o2663
Acta Crystallographica Section E
Structure Reports
Online
ISSN 1600-5368
3-(4-Fluorophenyl)-2-(4-methylphenoxy)-5,8,9-trimethylthieno[3
000,2
000:5,6]pyrido[4,3-
d
]pyrimidin-4(3
H
)-one
Zeping Cui,aMingtian Li,b Jianchao Liuaand Hongwu Hea*
a
Key Laboratory of Pesticide and Chemical Biology, College of Chemistry, Central China Normal University, Wuhan, Hubei 430079, People’s Republic of China, andbDepartment of Chemistry, Central China Normal University, Wuhan, Hubei 430079, People’s Republic of China
Correspondence e-mail: cuizeping@yahoo.com.cn
Key indicators
Single-crystal X-ray study
T= 292 K
Mean(C–C) = 0.003 A˚
Rfactor = 0.050
wRfactor = 0.117
Data-to-parameter ratio = 16.9
For details of how these key indicators were automatically derived from the article, see http://journals.iucr.org/e.
#2005 International Union of Crystallography
Printed in Great Britain – all rights reserved
In the title molecule, C25H20FN3O2S, the central tricyclic
system is essentially planar. All bond lengths and angles are within normal ranges. The crystal packing is stabilized by–
stacking interactions and van der Waals forces.
Comment
Pyridine-containing heterocyclic compounds have been
intensively studied due to the biological activity they often demonstrate (Augusto et al., 1995). The title compound, (I), belongs to this family of heterocyclic compounds and we present its crystal structure here.
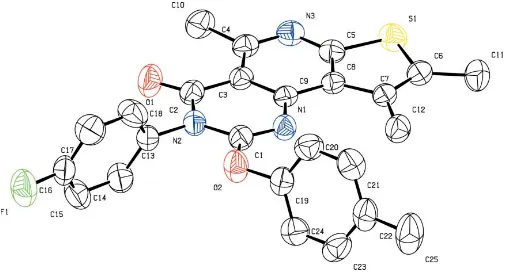
In (I) (Fig. 1), the C—S bond lengths [1.730 (2) and 1.744 (2) A˚ ] are greater than those observed in free thiophene [1.714 (s.u.) A˚; Bonham & Momany, 1963] and thieno[2,3-c ]-pyridine [1.728 (1) and 1.731 (1) A˚ ; Nerenzet al., 1997]. The C5—S1—C6 angle of 91.29 (10)in (I) is slightly less than that
observed in free thiophene [92.2 (2)]. As expected for a
non-protonated ring system, the C1—N1—C9 angle of 117.17 (16)
[image:1.610.255.411.308.461.2] [image:1.610.206.460.585.722.2]Received 4 July 2005 Accepted 18 July 2005 Online 23 July 2005
Figure 1
is smaller than 120 (Ghosh & Simonsen, 1993). The torsion
angles C4—C3—C9—N1 and C9—C8—C5—S1 are
179.41 (17) and 179.89 (13), respectively, showing the
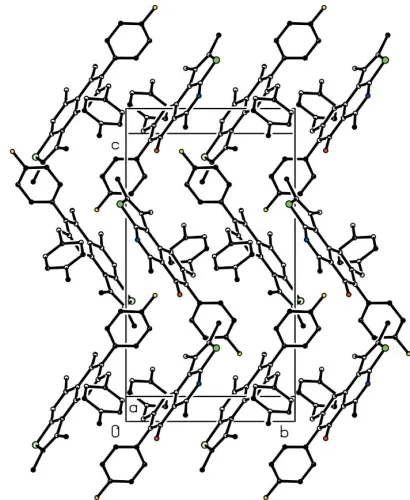
essen-tial planarity of the tricyclic system. The short intermolecular distances between the centroids of the thiophene (Cg1), pyridine (Cg2) and pyrimidine (Cg3) rings [Cg1 Cg2i = 3.525 (11) A˚ and Cg1 Cg3i= 3.516 (12) A˚ ; symmetry code:
(i) 2 x, 1y,z] indicate the existence of –stacking interactions, which stabilize the crystal packing (Fig. 2) toge-ther with van der Waals forces.
Experimental
To a solution of iminophosphorane (1 mmol) and 4-fluorophenyl isocyanate (1.1 mmol) in dry CH2Cl2(20 ml) was added
4-methyl-phenol (1.1 mmol) and a catalytic amount of K2CO3under N2at
room temperature. After filtration, the solid was recrystallized from acetonitrile. Colourless block-shaped crystals of the title compound were obtained by evaporation of the solvent over a period of one week.
Crystal data
C25H20FN3O2S
Mr= 445.50 Monoclinic,P21=c
a= 11.0623 (10) A˚
b= 10.4086 (9) A˚
c= 20.2368 (15) A˚
= 110.737 (4) V= 2179.2 (3) A˚3
Z= 4
Dx= 1.358 Mg m
3 MoKradiation Cell parameters from 2397
reflections
= 2.4–21.6 = 0.19 mm1
T= 292 (2) K Block, colourless 0.300.200.10 mm
Data collection
Bruker SMART CCD area-detector diffractometer
’and!scans
Absorption correction: multi-scan (SADABS; Bruker, 2000)
Tmin= 0.947,Tmax= 0.982 12598 measured reflections
4959 independent reflections 2909 reflections withI> 2(I)
Rint= 0.054
max= 27.5
h=14!9
k=13!13
l=24!26
Refinement
Refinement onF2
R[F2> 2(F2)] = 0.050
wR(F2) = 0.117
S= 0.89 4959 reflections 293 parameters
H-atom parameters constrained
w= 1/[2 (Fo
2
) + (0.0516P)2] whereP= (Fo2+ 2Fc2)/3 (/)max= 0.006
max= 0.20 e A˚
3
min=0.26 e A˚
3
C-bound H atoms were introduced at calculated positions and refined using a riding model, withUiso(H) = 1.2–1.5Ueq(C) and C—
H = 0.93–0.96 A˚ .
Data collection:SMART(Bruker, 2000); cell refinement:SAINT (Bruker, 2000); data reduction:SAINT; program(s) used to solve structure:SHELXS97(Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: SHELXTL (Bruker, 1997); software used to prepare material for publication:SHELXTL.
The authors acknowledge financial support from the
National Natural Science Foundation of China (No.
20072008).
References
Augusto, C. Y., Rosella, C. & Carlo, F. M. (1995).Tetrahedron,51, 12277– 12284.
Bonham, R. A. & Momany, F. A. (1963).J. Phys. Chem.67, 2474–2477. Bruker (1997). SHELXTL. Version 5.10. Bruker AXS Inc., Madison,
Wisconsin, USA.
Bruker (2000).SMART(Version 5.059),SAINT(Version 6.01) andSADABS
(Version 6.10). Bruker AXS Inc., Madison, Wisconsin, USA. Ghosh, R. & Simonsen, S. H. (1993).Acta Cryst.C49, 1031–1032. Nerenz, H., Grahn, W. & Jones, P. G. (1997).Acta Cryst.C53, 787–789. Sheldrick, G. M. (1997). SHELXS97 and SHELXL97. University of
[image:2.610.66.271.72.322.2]Go¨ttingen, Germany. Figure 2
supporting information
sup-1
Acta Cryst. (2005). E61, o2663–o2664
supporting information
Acta Cryst. (2005). E61, o2663–o2664 [https://doi.org/10.1107/S1600536805022968]
3-(4-Fluorophenyl)-2-(4-methylphenoxy)-5,8,9-trimethyl-thieno[3
′
,2
′
:5,6]pyrido[4,3-
d
]pyrimidin-4(3
H
)-one
Zeping Cui, Mingtian Li, Jianchao Liu and Hongwu He
3-(4-Fluorophenyl)-2-(4-methylphenoxy)-5,8,9- trimethylthieno[3′,2′:5,6]pyrido[4,3-d]pyrimidin-4(3H)-one
Crystal data
C25H20FN3O2S
Mr = 445.50
Monoclinic, P21/c
Hall symbol: -P 2ybc
a = 11.0623 (10) Å
b = 10.4086 (9) Å
c = 20.2368 (15) Å
β = 110.737 (4)°
V = 2179.2 (3) Å3
Z = 4
F(000) = 928
Dx = 1.358 Mg m−3
Mo Kα radiation, λ = 0.71073 Å Cell parameters from 2397 reflections
θ = 2.4–21.6°
µ = 0.19 mm−1
T = 292 K Block, colourless 0.30 × 0.20 × 0.10 mm
Data collection
Bruker SMART CCD area-detector diffractometer
Radiation source: fine-focus sealed tube Graphite monochromator
φ and ω scans
Absorption correction: multi-scan (SADABS; Bruker, 2000)
Tmin = 0.947, Tmax = 0.982
12598 measured reflections 4959 independent reflections 2909 reflections with I > 2σ(I)
Rint = 0.054
θmax = 27.5°, θmin = 2.0°
h = −14→9
k = −13→13
l = −24→26
Refinement
Refinement on F2
Least-squares matrix: full
R[F2 > 2σ(F2)] = 0.050
wR(F2) = 0.117
S = 0.89 4959 reflections 293 parameters 0 restraints
Primary atom site location: structure-invariant direct methods
Secondary atom site location: difference Fourier map
Hydrogen site location: inferred from neighbouring sites
H-atom parameters constrained
w = 1/[σ2(F
o2) + (0.0516P)2]
where P = (Fo2 + 2Fc2)/3
(Δ/σ)max = 0.006
Δρmax = 0.20 e Å−3
Special details
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes.
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2,
conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used
only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2
are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger.
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
x y z Uiso*/Ueq
C1 0.8452 (2) 0.16516 (19) −0.04561 (11) 0.0454 (5)
C2 1.00418 (19) 0.20374 (19) −0.10009 (10) 0.0456 (5)
C3 1.06724 (18) 0.28633 (18) −0.03935 (10) 0.0410 (5)
C4 1.18197 (19) 0.35666 (19) −0.03219 (11) 0.0445 (5)
C5 1.18292 (19) 0.43943 (18) 0.07233 (10) 0.0432 (5)
C6 1.1212 (2) 0.4915 (2) 0.17596 (10) 0.0482 (5)
C7 1.03619 (18) 0.40839 (18) 0.13231 (10) 0.0417 (5)
C8 1.07077 (17) 0.37696 (18) 0.07151 (9) 0.0385 (4)
C9 1.01150 (18) 0.29756 (18) 0.01296 (9) 0.0384 (4)
C10 1.2467 (2) 0.3522 (2) −0.08597 (11) 0.0598 (6)
H10A 1.3168 0.4126 −0.0733 0.090*
H10B 1.1852 0.3739 −0.1316 0.090*
H10C 1.2795 0.2673 −0.0874 0.090*
C11 1.1204 (2) 0.5490 (2) 0.24443 (11) 0.0646 (6)
H11A 1.0475 0.6055 0.2348 0.097*
H11B 1.1987 0.5966 0.2665 0.097*
H11C 1.1144 0.4815 0.2754 0.097*
C12 0.9214 (2) 0.3539 (2) 0.14644 (11) 0.0544 (6)
H12A 0.9185 0.3888 0.1898 0.082*
H12B 0.9291 0.2621 0.1503 0.082*
H12C 0.8434 0.3760 0.1083 0.082*
C13 0.81578 (19) 0.06377 (19) −0.15961 (10) 0.0440 (5)
C14 0.8536 (2) −0.0600 (2) −0.16256 (11) 0.0575 (6)
H14 0.9288 −0.0909 −0.1285 0.069*
C15 0.7793 (2) −0.1393 (2) −0.21660 (12) 0.0640 (6)
H15 0.8044 −0.2238 −0.2195 0.077*
C16 0.6699 (2) −0.0925 (2) −0.26502 (11) 0.0544 (6)
C17 0.6329 (2) 0.0312 (2) −0.26416 (12) 0.0673 (7)
H17 0.5590 0.0622 −0.2992 0.081*
C18 0.7067 (2) 0.1105 (2) −0.21039 (11) 0.0639 (6)
H18 0.6824 0.1956 −0.2087 0.077*
C19 0.67781 (19) 0.1224 (2) −0.00289 (10) 0.0487 (5)
C20 0.6140 (2) 0.2343 (2) −0.00177 (12) 0.0578 (6)
H20 0.6131 0.3012 −0.0324 0.069*
supporting information
sup-3
Acta Cryst. (2005). E61, o2663–o2664
H21 0.5085 0.3220 0.0476 0.074*
C22 0.5492 (2) 0.1471 (3) 0.09045 (11) 0.0593 (6)
C23 0.6157 (2) 0.0361 (3) 0.08784 (11) 0.0631 (6)
H23 0.6168 −0.0313 0.1182 0.076*
C24 0.6806 (2) 0.0227 (2) 0.04135 (11) 0.0563 (6)
H24 0.7251 −0.0525 0.0402 0.068*
C25 0.4740 (3) 0.1575 (3) 0.13960 (14) 0.0953 (10)
H25A 0.4062 0.0945 0.1268 0.143*
H25B 0.4371 0.2418 0.1360 0.143*
H25C 0.5309 0.1427 0.1873 0.143*
F1 0.59371 (13) −0.17201 (14) −0.31586 (7) 0.0800 (4)
N1 0.89993 (16) 0.23327 (16) 0.00921 (8) 0.0448 (4)
N2 0.88858 (15) 0.14488 (15) −0.10039 (8) 0.0440 (4)
N3 1.23908 (16) 0.43169 (16) 0.02338 (9) 0.0489 (4)
O1 1.04009 (15) 0.18189 (15) −0.14930 (8) 0.0645 (4)
O2 0.73459 (14) 0.10248 (15) −0.05488 (7) 0.0613 (4)
S1 1.24644 (5) 0.53432 (5) 0.14686 (3) 0.05487 (18)
Atomic displacement parameters (Å2)
U11 U22 U33 U12 U13 U23
C1 0.0476 (12) 0.0454 (12) 0.0463 (12) −0.0034 (10) 0.0207 (10) −0.0021 (10)
C2 0.0490 (13) 0.0477 (13) 0.0431 (12) 0.0065 (10) 0.0202 (10) 0.0041 (10)
C3 0.0422 (11) 0.0412 (11) 0.0401 (11) 0.0054 (9) 0.0153 (9) 0.0050 (9)
C4 0.0440 (12) 0.0431 (12) 0.0497 (12) 0.0054 (10) 0.0208 (10) 0.0080 (10)
C5 0.0394 (11) 0.0412 (12) 0.0463 (12) 0.0006 (9) 0.0120 (10) 0.0034 (9)
C6 0.0528 (13) 0.0464 (13) 0.0417 (12) 0.0038 (10) 0.0123 (10) 0.0044 (9)
C7 0.0450 (12) 0.0398 (12) 0.0375 (11) 0.0029 (9) 0.0113 (10) 0.0037 (9)
C8 0.0381 (11) 0.0381 (11) 0.0374 (11) 0.0020 (9) 0.0111 (9) 0.0041 (8)
C9 0.0404 (11) 0.0360 (11) 0.0391 (11) 0.0024 (9) 0.0144 (9) 0.0059 (9)
C10 0.0579 (14) 0.0670 (16) 0.0671 (15) −0.0036 (12) 0.0374 (12) 0.0036 (12)
C11 0.0737 (17) 0.0703 (16) 0.0448 (13) −0.0075 (13) 0.0150 (12) −0.0106 (11)
C12 0.0585 (14) 0.0592 (14) 0.0517 (13) −0.0045 (11) 0.0273 (11) −0.0051 (11)
C13 0.0491 (12) 0.0447 (13) 0.0389 (11) 0.0011 (10) 0.0163 (10) −0.0035 (9)
C14 0.0603 (14) 0.0580 (15) 0.0485 (13) 0.0124 (12) 0.0122 (11) −0.0029 (11)
C15 0.0760 (17) 0.0497 (14) 0.0651 (16) 0.0079 (12) 0.0235 (14) −0.0121 (12)
C16 0.0621 (15) 0.0627 (16) 0.0414 (12) −0.0114 (12) 0.0222 (12) −0.0131 (11)
C17 0.0650 (16) 0.0666 (17) 0.0536 (14) 0.0019 (13) 0.0004 (12) 0.0010 (13)
C18 0.0688 (16) 0.0472 (14) 0.0609 (15) 0.0095 (12) 0.0046 (13) 0.0000 (11)
C19 0.0428 (12) 0.0606 (15) 0.0452 (12) −0.0148 (11) 0.0186 (10) −0.0107 (11)
C20 0.0594 (15) 0.0566 (15) 0.0591 (14) −0.0057 (12) 0.0229 (12) 0.0038 (11)
C21 0.0544 (14) 0.0643 (17) 0.0713 (16) −0.0043 (12) 0.0271 (13) −0.0114 (13)
C22 0.0501 (14) 0.0782 (18) 0.0500 (14) −0.0242 (13) 0.0180 (11) −0.0197 (13)
C23 0.0649 (16) 0.0739 (18) 0.0455 (13) −0.0220 (14) 0.0133 (12) 0.0027 (12)
C24 0.0505 (13) 0.0572 (15) 0.0556 (14) −0.0069 (11) 0.0119 (11) −0.0021 (11)
C25 0.100 (2) 0.124 (3) 0.0846 (19) −0.0473 (19) 0.0608 (18) −0.0386 (18)
F1 0.0832 (10) 0.0903 (11) 0.0646 (9) −0.0244 (8) 0.0238 (8) −0.0316 (8)
N2 0.0481 (10) 0.0449 (10) 0.0408 (9) −0.0012 (8) 0.0180 (8) −0.0045 (8)
N3 0.0442 (10) 0.0491 (11) 0.0561 (11) −0.0009 (8) 0.0210 (9) 0.0041 (9)
O1 0.0712 (10) 0.0786 (12) 0.0558 (9) −0.0073 (9) 0.0373 (9) −0.0128 (8)
O2 0.0593 (9) 0.0722 (11) 0.0596 (9) −0.0261 (8) 0.0300 (8) −0.0233 (8)
S1 0.0501 (3) 0.0542 (4) 0.0547 (4) −0.0083 (3) 0.0116 (3) −0.0055 (3)
Geometric parameters (Å, º)
C1—N1 1.274 (2) C12—H12C 0.9600
C1—O2 1.340 (2) C13—C14 1.362 (3)
C1—N2 1.371 (2) C13—C18 1.368 (3)
C2—O1 1.217 (2) C13—N2 1.454 (2)
C2—N2 1.416 (2) C14—C15 1.385 (3)
C2—C3 1.459 (3) C14—H14 0.9300
C3—C9 1.406 (2) C15—C16 1.349 (3)
C3—C4 1.427 (3) C15—H15 0.9300
C4—N3 1.331 (2) C16—C17 1.353 (3)
C4—C10 1.500 (2) C16—F1 1.357 (2)
C5—N3 1.345 (2) C17—C18 1.380 (3)
C5—C8 1.396 (3) C17—H17 0.9300
C5—S1 1.730 (2) C18—H18 0.9300
C6—C7 1.351 (3) C19—C24 1.363 (3)
C6—C11 1.512 (3) C19—C20 1.366 (3)
C6—S1 1.744 (2) C19—O2 1.419 (2)
C7—C8 1.449 (2) C20—C21 1.380 (3)
C7—C12 1.508 (3) C20—H20 0.9300
C8—C9 1.403 (2) C21—C22 1.372 (3)
C9—N1 1.382 (2) C21—H21 0.9300
C10—H10A 0.9600 C22—C23 1.381 (3)
C10—H10B 0.9600 C22—C25 1.509 (3)
C10—H10C 0.9600 C23—C24 1.377 (3)
C11—H11A 0.9600 C23—H23 0.9300
C11—H11B 0.9600 C24—H24 0.9300
C11—H11C 0.9600 C25—H25A 0.9600
C12—H12A 0.9600 C25—H25B 0.9600
C12—H12B 0.9600 C25—H25C 0.9600
N1—C1—O2 121.41 (18) C18—C13—N2 119.76 (18)
N1—C1—N2 126.52 (18) C13—C14—C15 119.5 (2)
O2—C1—N2 112.06 (17) C13—C14—H14 120.2
O1—C2—N2 118.14 (18) C15—C14—H14 120.2
O1—C2—C3 127.02 (19) C16—C15—C14 119.1 (2)
N2—C2—C3 114.84 (16) C16—C15—H15 120.4
C9—C3—C4 119.49 (18) C14—C15—H15 120.4
C9—C3—C2 118.48 (17) C15—C16—C17 122.1 (2)
C4—C3—C2 122.03 (17) C15—C16—F1 119.1 (2)
N3—C4—C3 121.72 (17) C17—C16—F1 118.7 (2)
supporting information
sup-5
Acta Cryst. (2005). E61, o2663–o2664
C3—C4—C10 123.11 (19) C16—C17—H17 120.6
N3—C5—C8 127.14 (18) C18—C17—H17 120.6
N3—C5—S1 121.68 (15) C13—C18—C17 119.9 (2)
C8—C5—S1 111.18 (14) C13—C18—H18 120.1
C7—C6—C11 128.61 (19) C17—C18—H18 120.1
C7—C6—S1 113.27 (15) C24—C19—C20 121.9 (2)
C11—C6—S1 118.13 (16) C24—C19—O2 117.6 (2)
C6—C7—C8 111.58 (17) C20—C19—O2 120.26 (19)
C6—C7—C12 123.71 (17) C19—C20—C21 118.5 (2)
C8—C7—C12 124.70 (18) C19—C20—H20 120.8
C5—C8—C9 115.62 (17) C21—C20—H20 120.8
C5—C8—C7 112.68 (18) C22—C21—C20 121.6 (2)
C9—C8—C7 131.70 (17) C22—C21—H21 119.2
N1—C9—C8 118.22 (16) C20—C21—H21 119.2
N1—C9—C3 122.66 (17) C21—C22—C23 118.0 (2)
C8—C9—C3 119.12 (17) C21—C22—C25 121.3 (2)
C4—C10—H10A 109.5 C23—C22—C25 120.7 (2)
C4—C10—H10B 109.5 C24—C23—C22 121.6 (2)
H10A—C10—H10B 109.5 C24—C23—H23 119.2
C4—C10—H10C 109.5 C22—C23—H23 119.2
H10A—C10—H10C 109.5 C19—C24—C23 118.4 (2)
H10B—C10—H10C 109.5 C19—C24—H24 120.8
C6—C11—H11A 109.5 C23—C24—H24 120.8
C6—C11—H11B 109.5 C22—C25—H25A 109.5
H11A—C11—H11B 109.5 C22—C25—H25B 109.5
C6—C11—H11C 109.5 H25A—C25—H25B 109.5
H11A—C11—H11C 109.5 C22—C25—H25C 109.5
H11B—C11—H11C 109.5 H25A—C25—H25C 109.5
C7—C12—H12A 109.5 H25B—C25—H25C 109.5
C7—C12—H12B 109.5 C1—N1—C9 117.17 (16)
H12A—C12—H12B 109.5 C1—N2—C2 120.26 (17)
C7—C12—H12C 109.5 C1—N2—C13 120.36 (16)
H12A—C12—H12C 109.5 C2—N2—C13 119.38 (15)
H12B—C12—H12C 109.5 C4—N3—C5 116.91 (16)
C14—C13—C18 120.4 (2) C1—O2—C19 116.58 (15)
C14—C13—N2 119.75 (18) C5—S1—C6 91.29 (10)
O1—C2—C3—C9 −178.91 (19) C24—C19—C20—C21 0.0 (3)
N2—C2—C3—C9 1.5 (3) O2—C19—C20—C21 174.66 (17)
O1—C2—C3—C4 1.9 (3) C19—C20—C21—C22 −1.2 (3)
N2—C2—C3—C4 −177.61 (17) C20—C21—C22—C23 1.6 (3)
C9—C3—C4—N3 0.9 (3) C20—C21—C22—C25 −176.6 (2)
C2—C3—C4—N3 −179.92 (17) C21—C22—C23—C24 −1.0 (3)
C9—C3—C4—C10 −178.93 (18) C25—C22—C23—C24 177.3 (2)
C2—C3—C4—C10 0.2 (3) C20—C19—C24—C23 0.6 (3)
C11—C6—C7—C8 179.56 (19) O2—C19—C24—C23 −174.16 (17)
S1—C6—C7—C8 −0.6 (2) C22—C23—C24—C19 −0.1 (3)
S1—C6—C7—C12 178.34 (15) N2—C1—N1—C9 2.7 (3)
N3—C5—C8—C9 0.2 (3) C8—C9—N1—C1 177.64 (17)
S1—C5—C8—C9 179.89 (13) C3—C9—N1—C1 −2.4 (3)
N3—C5—C8—C7 −179.16 (18) N1—C1—N2—C2 −0.9 (3)
S1—C5—C8—C7 0.6 (2) O2—C1—N2—C2 179.79 (17)
C6—C7—C8—C5 0.0 (2) N1—C1—N2—C13 179.67 (19)
C12—C7—C8—C5 −178.90 (18) O2—C1—N2—C13 0.3 (3)
C6—C7—C8—C9 −179.17 (19) O1—C2—N2—C1 179.08 (17)
C12—C7—C8—C9 1.9 (3) C3—C2—N2—C1 −1.3 (3)
C5—C8—C9—N1 −179.93 (16) O1—C2—N2—C13 −1.5 (3)
C7—C8—C9—N1 −0.8 (3) C3—C2—N2—C13 178.11 (16)
C5—C8—C9—C3 0.1 (3) C14—C13—N2—C1 −100.1 (2)
C7—C8—C9—C3 179.25 (18) C18—C13—N2—C1 77.2 (2)
C4—C3—C9—N1 179.41 (17) C14—C13—N2—C2 80.5 (2)
C2—C3—C9—N1 0.2 (3) C18—C13—N2—C2 −102.3 (2)
C4—C3—C9—C8 −0.6 (3) C3—C4—N3—C5 −0.7 (3)
C2—C3—C9—C8 −179.77 (17) C10—C4—N3—C5 179.18 (17)
C18—C13—C14—C15 −1.4 (3) C8—C5—N3—C4 0.1 (3)
N2—C13—C14—C15 175.84 (18) S1—C5—N3—C4 −179.55 (15)
C13—C14—C15—C16 −0.6 (3) N1—C1—O2—C19 3.8 (3)
C14—C15—C16—C17 2.6 (3) N2—C1—O2—C19 −176.81 (16)
C14—C15—C16—F1 −176.68 (19) C24—C19—O2—C1 −109.5 (2)
C15—C16—C17—C18 −2.6 (4) C20—C19—O2—C1 75.7 (2)
F1—C16—C17—C18 176.7 (2) N3—C5—S1—C6 178.99 (17)
C14—C13—C18—C17 1.4 (3) C8—C5—S1—C6 −0.74 (15)
N2—C13—C18—C17 −175.82 (19) C7—C6—S1—C5 0.78 (16)