Figure 3.0-2
Chapter 3: Big Ideas
Introduction to Organic
3.1 Life’s molecular diversity is based on the
properties of carbon
• Almost all the molecules a cell makes are
composed of carbon bonded to
• other carbons and
• atoms of other elements.
• Carbon-based molecules are called organic
3.1 Life’s molecular diversity is based on the
properties of carbon
• By sharing electrons, carbon can
• bond to four other atoms and
• branch in up to four directions.
• Methane (CH4) is one of the simplest organic
compounds.
• Four covalent bonds link four hydrogen atoms to the carbon atom.
Figure 3.1a
The four single bonds of carbon point to the corners of a tetrahedron.
H
H H
H
3.1 Life’s molecular diversity is based on the
properties of carbon
• Methane and other compounds composed of only
carbon and hydrogen are called hydrocarbons.
• Carbon, with attached hydrogens, can form chains
3.1 Life’s molecular diversity is based on the
properties of carbon
• A carbon skeleton is a chain of carbon atoms that
can differ in length and be
• straight,
• branched, or
3.1 Life’s molecular diversity is based on the
properties of carbon
• Compounds with the same formula but different
Figure 3.1b-0
Butane
Length: Carbon skeletons vary in length.
Propane 1-Butene 2-Butene
Double bonds: Carbon skeletons may have double bonds, which can vary in location.
Double bond
Isobutane Cyclohexane Benzene
Figure 3.1b-1
Length: Carbon skeletons vary in length.
Figure 3.1b-2
Butane Isobutane
Figure 3.1b-3
1-Butene 2-Butene
Double bonds: Carbon skeletons may have double bonds, which can vary in location.
Figure 3.1b-4
Cyclohexane Benzene
3.2 A few chemical groups are key to the
functioning of biological molecules
• The unique properties of an organic compound
depend on
• the size and shape of its carbon skeleton and
• the groups of atoms that are attached to that
skeleton.
• The sex hormones testosterone and estradiol (a
Figure 3.2-0
3.2 A few chemical groups are key to the
functioning of biological molecules
• Table 3.2 illustrates six chemical groups important
in the chemistry of life.
• The first five groups are called functional groups;
they affect a molecule’s function in a characteristic way.
• These five groups are polar, so compounds
containing them are typically hydrophilic
3.2 A few chemical groups are key to the
functioning of biological molecules
• A sixth group, a methyl group,
• consists of a carbon bonded to three hydrogen atoms,
• is nonpolar and not reactive, but
3.2 A few chemical groups are key to the
functioning of biological molecules
• The functional groups are
• hydroxyl group—a hydrogen bonded to an oxygen,
• carbonyl group—a carbon linked by a double bond to an oxygen atom,
• carboxyl group—a carbon double-bonded to both an oxygen and a hydroxyl group,
3.3 Cells make large molecules from a limited
set of small molecules
• There are four classes of molecules important to
organisms:
1. carbohydrates,
2. lipids,
3. proteins, and
3.3 Cells make large molecules from a limited
set of small molecules
• The four classes of biological molecules contain
very large molecules.
• They are often called macromolecules because of their large size.
• They are also called polymers because they are
made from identical or similar building blocks strung together.
3.3 Cells make large molecules from a limited
set of small molecules
• Monomers are linked together to form polymers
through dehydration reactions, which remove
water.
• Polymers are broken apart by hydrolysis, the
addition of water.
• These reactions are mediated by enzymes,
3.3 Cells make large molecules from a limited
set of small molecules
• A cell makes a large number of polymers from a
small group of monomers. For example,
• proteins are made from only 20 different amino acids and
• DNA is built from just four kinds of monomers called nucleotides.
• The monomers used to make polymers are
Figure 3.3-0
Short polymer Unlinked monomer
Dehydration reaction forms a new bond
H2O
Longer polymer
Hydrolysis breaks a bond
Figure 3.3-1-1
Short polymer Unlinked
Figure 3.3-1-2
Short polymer Unlinked
monomer
Dehydration reaction forms a new bond
Figure 3.3-2-2
Hydrolysis
breaks a bond
3.4 Monosaccharides are the simplest
carbohydrates
• Carbohydrates range from small sugar molecules
(monomers) to large polysaccharides.
• Sugar monomers are monosaccharides, such as
those found in
• fructose,
• glucose, and
3.4 Monosaccharides are the simplest
carbohydrates
• Monosaccharides can be hooked together by
dehydration reactions to form
• more complex sugars and
3.4 Monosaccharides are the simplest
carbohydrates
• The carbon skeletons of monosaccharides vary in
length.
• Glucose and fructose are six carbons long.
• Others have three to seven carbon atoms.
• Monosaccharides are
• the main fuels for cellular work and
• used as raw materials to manufacture other organic
Figure 3.4b
3.4 Monosaccharides are the simplest
carbohydrates
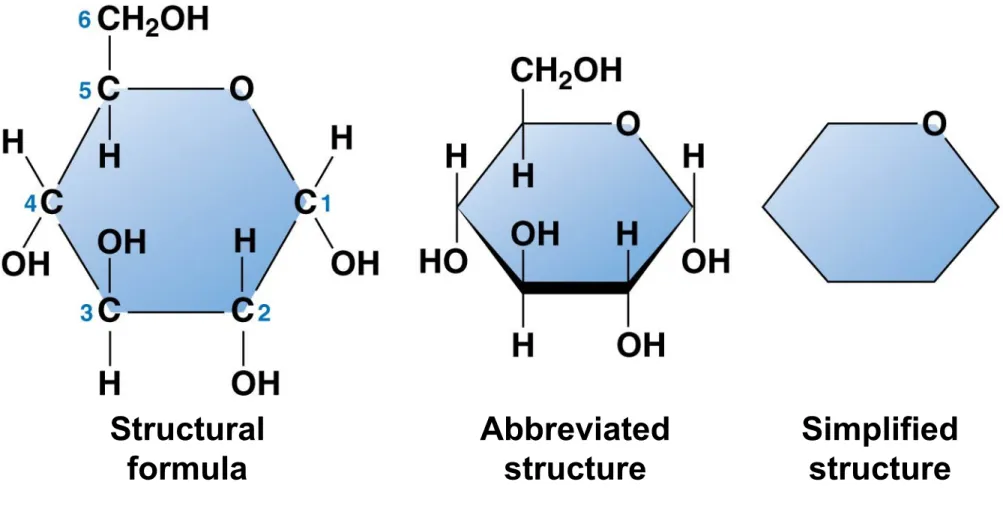
• Many monosaccharides form rings.
• The ring diagram may be
• abbreviated by not showing the carbon atoms at the corners of the ring and
Figure 3.4c
Structural formula
Abbreviated structure
3.5 Two monosaccharides are linked to form
a disaccharide
• Two monosaccharides (monomers) can bond to
form a disaccharide in a dehydration reaction.
• The disaccharide sucrose is formed by combining
• a glucose monomer and
• a fructose monomer.
• The disaccharide maltose is formed from two
Figure 3.5-1
Figure 3.5-2
Glucose Glucose
3.6 CONNECTION: What is high-fructose corn
syrup, and is it to blame for obesity?
• Most sodas and fruit drinks contain high-fructose
corn syrup (HFCS).
• Fructose is sweeter than glucose.
• To make HFCS, glucose atoms are rearranged to
3.6 CONNECTION: What is high-fructose corn
syrup, and is it to blame for obesity?
• High-fructose corn syrup (HFCS)
• is used to sweeten many beverages,
• consists of about 55% fructose and 45% glucose, and
• is a mixture with about the same proportions as in sucrose.
• Most scientific studies have not shown health
3.6 CONNECTION: What is high-fructose corn
syrup, and is it to blame for obesity?
• Although HFCS consumption has declined
somewhat in recent years, obesity rates continue to rise, with almost 36% of U.S. adults now
considered obese.
• Good health is promoted by
• a diverse diet of proteins, fats, vitamins, minerals, complex carbohydrates, and
3.7 Polysaccharides are long chains of sugar
units
• Polysaccharides are macromolecules, polymers
composed of thousands of monosaccharides.
• Polysaccharides may function as
• storage molecules or
3.7 Polysaccharides are long chains of sugar
units
• Starch is
• a polysaccharide,
• composed of glucose monomers, and
• used by plants for energy storage.
• Glycogen is
• a polysaccharide,
• composed of glucose monomers, and
3.7 Polysaccharides are long chains of sugar
units
• Cellulose
• is a polymer of glucose and
• forms plant cell walls.
• Chitin is
• a polysaccharide,
• used by insects and crustaceans to build an exoskeleton, and
Figure 3.7-0
Starch granules in
a potato tuber cell Starch
Glycogen granules in muscle
tissue
Cellulose microfibrils in a plant cell wall
Cellulose
molecules Hydrogen bonds
Cellulose Glycogen
Figure 3.7-1
Starch granules in
a potato tuber cell Starch
Figure 3.7-2
Glycogen granules in muscle
tissue
Figure 3.7-3
Cellulose microfibrils in a plant cell wall
Cellulose
molecules Hydrogen bonds
3.7 Polysaccharides are long chains of
sugar units
• Polysaccharides are usually hydrophilic
(water-loving).
• Bath towels, for example, are often made of
3.8 Fats are lipids that are mostly
energy-storage molecules
• Lipids
• are water insoluble (hydrophobic, or water-fearing) compounds,
• are important in long-term energy storage,
• contain twice as much energy as a polysaccharide, and
3.8 Fats are lipids that are mostly
energy-storage molecules
• Lipids differ from carbohydrates, proteins, and
nucleic acids in that they are
• not huge molecules and
• not built from monomers.
3.8 Fats are lipids that are mostly
energy-storage molecules
• We will consider three types of lipids:
1. fats,
2. phospholipids, and
3. steroids.
• A fat is a large lipid made from two kinds of
smaller molecules:
3.8 Fats are lipids that are mostly
energy-storage molecules
• A fatty acid can link to glycerol by a dehydration
reaction.
• A fat contains one glycerol linked to three fatty
acids.
• Fats are often called triglycerides because of their
Figure 3.8a
Glycerol
Fatty acid
3.8 Fats are lipids that are mostly
energy-storage molecules
• Some fatty acids contain one or more double
bonds, forming unsaturated fatty acids.
• These have one fewer hydrogen atom on each carbon of the double bond.
• These double bonds cause kinks or bends in the carbon chain, preventing them from packing
together tightly and solidifying at room temperature.
• Fats with the maximum number of hydrogens are
3.8 Fats are lipids that are mostly
energy-storage molecules
• Unsaturated fats are referred to as oils.
• Most animal fats are saturated fats.
• Hydrogenated vegetable oils are unsaturated fats
that have been converted to saturated fats by adding hydrogen.
• This hydrogenation creates trans fats, which are
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• In the 1890s, the process of adding hydrogen
atoms to unsaturated oils was developed to
• reduce spoilage of oils,
• help oils withstand repeated heating for frying, and • reduce consumption of animal fats, thought to be
less healthy than partially hydrogenated vegetable oils.
• By the 1990s, partially hydrogenated oils were
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• Recent research has shown that trans fats pose an even greater health risk than saturated fats.
• One study estimated that eliminating trans fats from the
food supply could prevent up to one in five heart attacks!
• In 2006, the U.S. Food and Drug Administration
required the listing of trans fat on food labels.
• Many cities and states have passed laws to eliminate
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• Experimental studies have tested the health risks
of consuming trans fats.
• In experimental controlled feeding trials, the diets of participants contained different proportions of
saturated, unsaturated, and partially hydrogenated fats.
• For ethical and practical reasons, controlled feeding trials are usually fairly short in duration, involve only limited dietary changes, generally use healthy
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• Observational scientific studies on dietary health
effects can
• extend over a longer time period,
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• The Nurses’ Health Study
• began in 1976 with more than 120,000 female nurses and
3.9 SCIENTIFIC THINKING: Scientific studies
document the health risks of trans fats
• The results indicate that
• for each 5% increase in saturated fat, there was a 17% increase in the risk of heart disease and
• for each 2% increase in trans fat, there is a 93% increase in risk.
• Trans fats are indeed a greater health risk than
3.10 Phospholipids and steroids are
important lipids with a variety of functions
• Phospholipids are the major component of all cell
membranes.
• Phospholipids are structurally similar to fats.
• Fats contain three fatty acids attached to glycerol.
Figure 3.10
Phosphate group
Glycerol
Hydrophilic heads
Hydrophobic tails
Symbol for phospholipid
Water
Figure 3.10a
Phosphate group
Glycerol
Hydrophilic heads
3.10 Phospholipids and steroids are
important lipids with a variety of functions
• Phospholipids cluster into a bilayer of
phospholipids.
• The hydrophilic heads are in contact with
• the water of the environment and
• the internal part of the cell.
• The hydrophobic tails cluster together in the center
Figure 3.10b
Hydrophilic heads
Hydrophobic tails
Symbol for phospholipid
Water
3.10 Phospholipids and steroids are
important lipids with a variety of functions
• Steroids are lipids in which the carbon skeleton
contains four fused rings.
• Cholesterol is
• a common component in animal cell membranes and
3.11 CONNECTION: Anabolic steroids pose
health risks
• Anabolic steroids are synthetic variants of the
male hormone testosterone that are abused by some athletes with serious consequences,
including
• violent mood swings,
• depression,
• liver damage,
3.12 Proteins have a wide range of functions
and structures
• Proteins are
• involved in nearly every dynamic function in your body and
• very diverse, with tens of thousands of different
proteins, each with a specific structure and function, in the human body.
• Proteins are composed of differing arrangements
3.12 Proteins have a wide range of functions
and structures
• Probably the most important role for proteins is as
enzymes, proteins that
• serve as catalysts and
3.12 Proteins have a wide range of functions
and structures
• Other types of proteins include
• transport proteins embedded in cell membranes, which move sugar molecules and other nutrients into your cells,
• defensive proteins, such as antibodies of the immune system,
3.12 Proteins have a wide range of functions
and structures
• Other types of proteins include (continued)
• receptor proteins, built into cell membranes, which receive and transmit signals into your cells,
• contractile proteins found within muscle cells,
• structural proteins such as collagen, which form the long, strong fibers of connective tissues, and
3.12 Proteins have a wide range of functions
and structures
• The functions of different types of proteins depend
on their individual shapes.
• A polypeptide chain contains hundreds or
thousands of amino acids linked by peptide bonds.
• The amino acid sequence causes the polypeptide
Figure 3.12a
Figure 3.12b
3.12 Proteins have a wide range of functions
and structures
• If a protein’s shape is altered, it can no longer
function.
• In the process of denaturation, a protein
• unravels,
• loses its specific shape, and
• loses its function.
• Proteins can be denatured by changes in salt
3.13 Proteins are made from amino acids
linked by peptide bonds
• Amino acids all have
• an amino group and
• a carboxyl group (which makes it an acid).
• Also bonded to the central carbon is
• a hydrogen atom and
• a chemical group symbolized by R, which
Figure 3.13a
Amino group
3.13 Proteins are made from amino acids
linked by peptide bonds
• Amino acids are classified as either
• hydrophobic or
Figure 3.13b
Hydrophobic Hydrophilic
3.13 Proteins are made from amino acids
linked by peptide bonds
• Amino acid monomers are linked together in a
dehydration reaction,
• joining the carboxyl group of one amino acid to the amino group of the next amino acid, and
• creating a peptide bond.
• Additional amino acids can be added by the same
process to create a chain of amino acids called a
Figure 3.13c-1
Carboxyl group
Amino group
Figure 3.13c-2 Carboxyl group Amino group Amino acid Dehydration reaction Peptide bond Dipeptide H2O
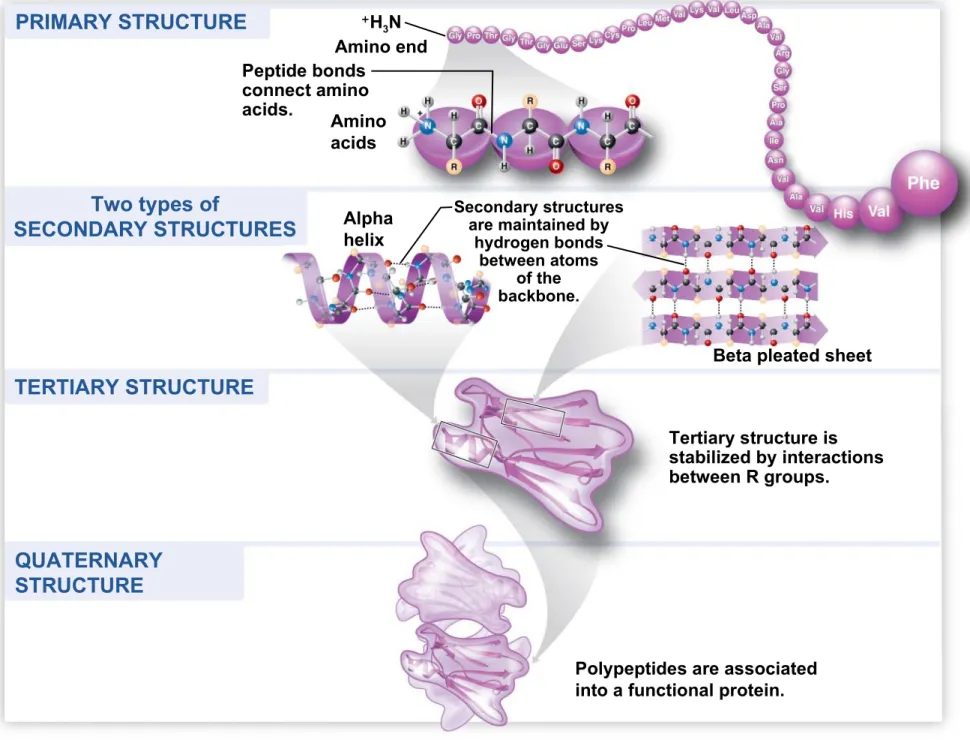
3.14 VISUALIZING THE CONCEPT: A
protein’s functional shape results from four
levels of structure
• A protein can have four levels of structure:
1. primary structure,
2. secondary structure,
3. tertiary structure, and
Figure 3.14-0-1
Amino acids
+H
3N Amino end Peptide bonds
connect amino acids.
Figure 3.14-0-2 Amino acids +H 3N Amino end Peptide bonds connect amino acids. Alpha helix Secondary structures are maintained by
hydrogen bonds between atoms
of the backbone.
Beta pleated sheet
PRIMARY STRUCTURE
Two types of
Figure 3.14-0-3 Amino acids +H 3N Amino end Peptide bonds connect amino acids. Alpha helix Secondary structures are maintained by
hydrogen bonds between atoms
of the backbone.
Beta pleated sheet
Tertiary structure is stabilized by interactions between R groups.
TERTIARY STRUCTURE PRIMARY STRUCTURE
Two types of
Figure 3.14-0-4 Amino acids +H 3N Amino end Peptide bonds connect amino acids. Alpha helix Secondary structures are maintained by
hydrogen bonds between atoms
of the backbone.
Beta pleated sheet
Tertiary structure is stabilized by interactions between R groups.
TERTIARY STRUCTURE PRIMARY STRUCTURE
QUATERNARY
Two types of
Figure 3.14-0 Amino acids +H 3N Amino end Peptide bonds connect amino acids. Alpha helix Secondary structures are maintained by
hydrogen bonds between atoms
of the backbone.
Beta pleated sheet
Tertiary structure is stabilized by interactions between R groups.
TERTIARY STRUCTURE PRIMARY STRUCTURE
Two types of
SECONDARY STRUCTURES
Figure 3.14-2
Alpha helix
Secondary structures are maintained by
hydrogen bonds between atoms
of the backbone.
Beta pleated sheet
Figure 3.14-3
Tertiary structure is stabilized by interactions
between R groups.
Figure 3.14-4
Polypeptides are associated into a functional protein.
3.15 DNA and RNA are the two types of
nucleic acids
• The amino acid sequence of a polypeptide is
programmed by a discrete unit of inheritance known as a gene.
• Genes consist of DNA (deoxyribonucleic acid), a
type of nucleic acid.
• DNA is inherited from an organism’s parents.
• DNA provides directions for its own replication.
• DNA programs a cell’s activities by directing the
3.15 DNA and RNA are the two types of
nucleic acids
• DNA does not build proteins directly.
• DNA works through an intermediary, RNA
(ribonucleic acid).
• DNA is transcribed into RNA in a cell’s nucleus.
Figure 3.15-1
Gene
Figure 3.15-2
Gene
Transcription
DNA
RNA
Figure 3.15-3
Gene
Transcription
Translation
Amino acid
DNA
RNA
Protein
3.16 Nucleic acids are polymers of
nucleotides
• DNA (deoxyribonucleic acid) and RNA (ribonucleic
acid) are composed of monomers called
nucleotides.
• Nucleotides have three parts:
1. a five-carbon sugar called ribose in RNA and deoxyribose in DNA,
2. a phosphate group, and
Figure 3.16a
Sugar Phosphate
group
Nitrogenous base
3.16 Nucleic acids are polymers of
nucleotides
• DNA nitrogenous bases are
• adenine (A),
• thymine (T),
• cytosine (C), and
• guanine (G).
• RNA also has A, C, and G, but instead of T, it has
3.16 Nucleic acids are polymers of
nucleotides
• A nucleic acid polymer, a polynucleotide, forms
from the nucleotide monomers when the
phosphate of one nucleotide bonds to the sugar of the next nucleotide by dehydration reactions.
• This produces a repeating sugar-phosphate
Figure 3.16b
Nucleotide A
T
C
G
3.16 Nucleic acids are polymers of
nucleotides
• RNA is usually a single polynucleotide strand.
• DNA is a double helix, in which two
polynucleotide strands wrap around each other.
• The two strands are associated because particular bases always hydrogen-bond to one another.
3.17 EVOLUTION CONNECTION: Lactose
tolerance is a recent event in human evolution
• The majority of people
• stop producing the enzyme lactase in early childhood and
• do not easily digest the milk sugar lactose after childhood.
• Lactose tolerance represents
• a relatively recent mutation in the human genome and
3.17 EVOLUTION CONNECTION: Lactose
tolerance is a recent event in human evolution
• Researchers identified three new mutations in 43
ethnic groups in East Africa that keep the lactase gene permanently turned on.
• The mutations
• are all different from each other and from the European mutation,