The origin and evolution of the nervous system
Full text
Figure
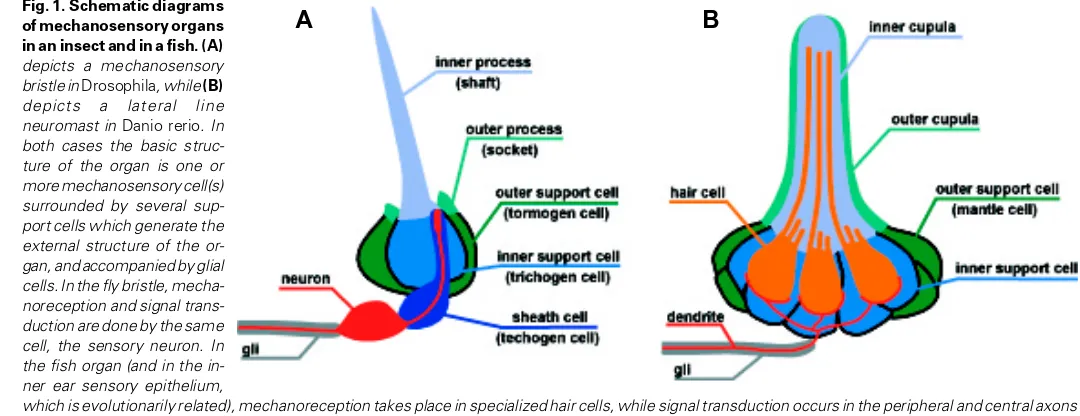
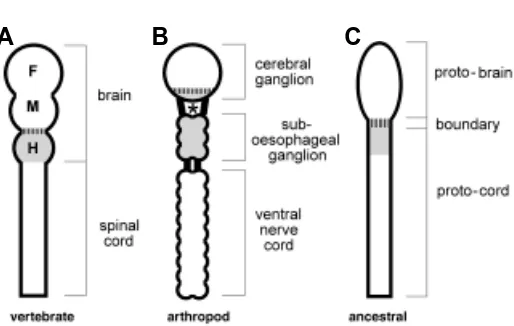
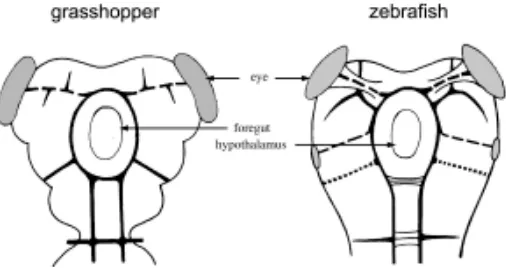
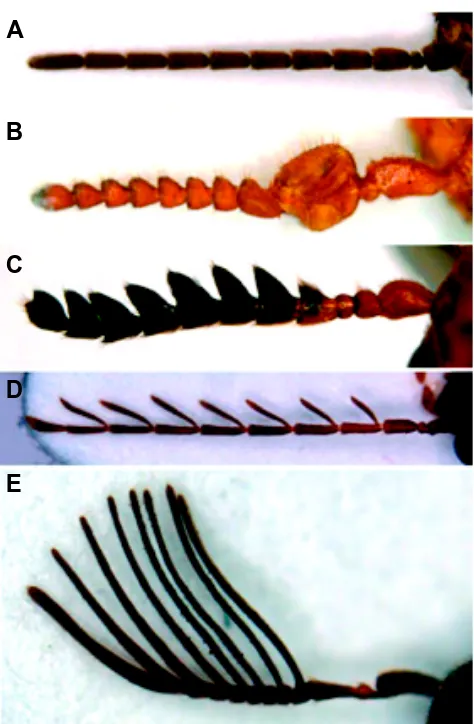
Related documents
Which of the following statements best describes the SEC rules relating to bookkeeping
Potential problems over which investors feel they have greater control—worries that can be addressed personally or mitigated via having greater financial means—do not tend to keep
This study significantly expands the number of wild Musa species where nuclear genome size and genomic distri- bution of rDNA loci is known.. SSR genotyping identified Musa species
Copyright © 2012, Asian Business Consortium | ABR Page 121 Human Resource Management Practices and Firms Performance in Bangladesh: An Empirical Study on
a) Except for free-fall lifeboats, fenders shall be fitted on both sides of lifeboats installed on mobile offshore units which are not of ship design. b) The lifeboat shall
The basic parameters for the model can be determined based on the population number in this area, the birth rate, the available water resources (including
We can provide the following, tentative evidence for this: adopting the baseline specification — Case (i) — from our own Monte Carlo setup we ran simulations where we dropped the
Asian American M3 members are most likely to work with their spouse/partner; women of color who have worked with domestic partners are much more evenly divided than Caucasian women