Native plant species richness in non-native Pinus contorta forest
Full text
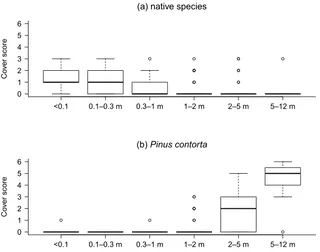
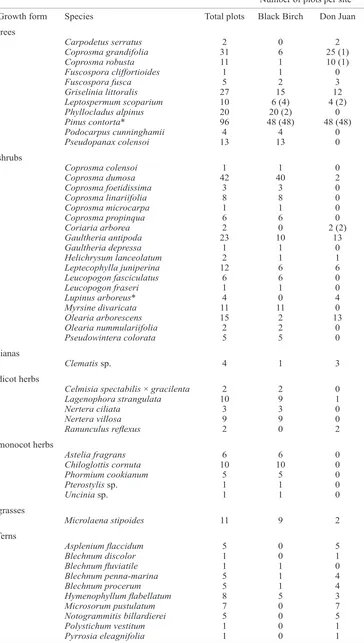
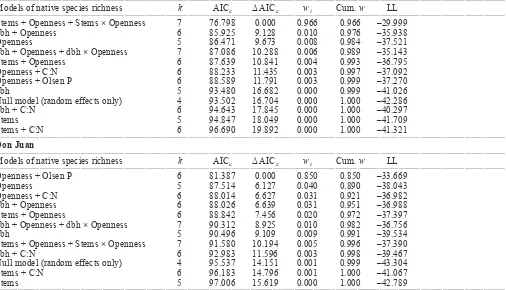
Figure
Related documents
Further, a recent meta- analysis has shown that, in sibutramine-administered obese patients, subjects who achieved weight losses of over 4 kg in the first 3 months of treatment were
Liver resection in the elderly: a retrospective cohort study of 460 patients - feasible and safe. Bartsch F, Baumgart J, Hoppe-Lotichius M, Schmidtmann I, Heinrich S,
The next chapter will take a closer examination of intrinsic value and value commensurability by detailing Moore’s specific theory about the compounding of values within wholes
The total long run average cost is $0.34 per vehicle km traveled, including user ®xed and variable costs, the cost of time to both the driver and pas- senger in traveling and
general, GPs were rather satisfied with their job with the exception of “hours of work,” “physical working condition,” and “income.” Job satis- faction differed only
When adult and child labor are substitutes (such as when IS are non-binding and both adults and children perform the same light tasks in the domestic sector), only the labor
The Markovic Case’, 1 J Int’l Crim Justice (2003) 406).. no question that the acts carried out by Germany occurred in the course of wartime activities and constituted the