Original Article
Prophylactic ozone administration reduces renal
ischemia-reperfusion injury in the rat
Oznur Kal1, Ishak Akillioglu2, Ali Kal3, Esin Celik4, Mustafa Yilmaz5, Merih Onal6, Ozkan Onal7
1Department of Nephrology, Baskent University Medical Faculty, Konya, Turkey; 2Department of Pediatric
Surgery, Baskent University Medical Faculty, Konya, Turkey; 3Department of Ophtalmology, Baskent University
Medical Faculty, Konya, Turkey; 4Department of Pathology, Selcuk University Medical Faculty, Konya, Turkey; 5Department of Histology and Embryology, Selcuk University Medical Faculty, Konya, Turkey; 6Department of
Otorhinolaryngology, Meram Educational and Training Hospital, Konya, Turkey; 7Department of Anesthesiology
and Reanimation, Selcuk University Medical Faculty, Konya, Turkey
Received January 20, 2016; Accepted June 8, 2016; Epub July 15, 2016; Published July 30, 2016
Abstract: Background: The objective of this study was to examine the role of ozone oxidative preconditioning after
renal IR (ischemia reperfusion) injury. Methods: Twenty-eight Wistar rats were randomized into four groups: sham operated (S), IR, ozone (O), and O+IR. The S group was administered physiological saline (PS) intraperitoneally (i.p.) for seven days. The IR group was subjected to renal ischemia for 1 h by occlusion of the left renal artery and vein, followed by reperfusion for 2 h. The O group was administered ozone i.p. for seven days. In the O+IR group, ozone was administered i.p. for seven days before the IR procedure. IR injury (as in the IR group) was induced on the eight day. Laboratory analyses of renal tissue samples for superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and malondialdehyde (MDA) were performed. Results: The total oxidant score (TOS) and total antioxidant capacity (TAC) of the blood samples were also analyzed. The degree of renal injury was highest in the IR group. In the O+IR group, renal injury was decreased. The antioxidant parameters were increased in the O group. The oxidant param-eters were highest in the IR group. Conclusion: Ozone preconditioning ameliorated renal IR injury, with a significant decrease observed in the renal IR injury score.
Keywords: Ischemia reperfusion injury, kidney, ozone, apoptosis
Introduction
Ozone gas was first utilized medically during World War I by the German physician Dr. Albert Wolff in local surgical interventions to prevent wound infections [1]. Since then, it has been used as an antibacterial and antiviral disinfec-tion agent [2]. Clinical studies have investigat-ed the use of ozone in combating ischemia, inflammation, infection, pressure sores, perito-nitis, and peripheral vascular diseases in dia-betic and nondiadia-betic patients, stroke patients, and others [3]. Following findings that low doses of ozone gas stimulated antioxidant endogenous systems, ozone therapy has been applied in the treatment of a variety of diseas-es in experimental and clinical studidiseas-es. Thdiseas-ese include cerebrovascular ischemia [4], chronic ulcers [5], arteriosclerosis obliterans [6], immune deficiency [7], hepatic steatosis [8],
charac-terized by a complex sequence of events, which is triggered by direct cellular damage caused by hypoxic processes [14]. Ischemic tissue reper-fusion aggravates acute ischemic injury via the formation of reactive oxygen and nitrogen com-ponents. This is followed by renal dysfunction and damage produced by an inflammatory cas-cade, resulting in the eventual death of renal cells [14].
To the best of our knowledge, there have been only a few reports of the role of ozone precondi-tioning in renal IR injury [15-17]. Given the lim-ited number of studies and the effectiveness of ozone treatment in renal IR injury shown in ear-lier studies, the present study investigated the effect of ozone on renal IR injury by evaluating the total antioxidant capacity (TAC) and total oxidant score (TOS) of the blood and malondial-dehyde (MDA), glutathione peroxidase (GSH-Px), and superoxide dismutase (SOD) levels in renal tissue samples.
Material and methods
The current study was approved by the Selcuk University Animal Ethics Committee and was performed in accordance with the National Institutes of Health guidelines and as recom-mended by the Science Council of Japan or the National Research Council’s criteria (NIH No. 86-23) for the use of experimental animals. Female Wistar rats weighing 250-300 g were used in this study. The animals were housed under a 12-h light/12-h dark cycle at a temper-ature of 24±3°C. The animals were fed with a standard pellet diet and water ad libitum. Experimental groups
The number of rats in the groups was deter-mined according to a previous study [18] and limited to 28 to eliminate excessive sacrifica-tion. The animals were randomly divided into four groups: sham operated (S) (n=7), IR, ozone (O), and O+IR. The S group was given physiologi-cal saline (PS) intraperitoneally (i.p.) for seven days. On the eight day, the right and left renal pedicles were exposed after a midline incision, and laparotomy was performed, but IR injury was not induced. The IR group (n=7) was sub-jected to renal ischemia for 1 h by occlusion of the left renal artery and vein with a nontrau-matic clamp, followed by reperfusion for 2 h and neither PS nor ozone was administered.
The O group (n=7) was given ozone at a dosage of 1 mg/kg in a single dose administered i.p. each day for seven days. On the eight day, the right and left renal pedicles were exposed after a midline incision, and laparotomy was per-formed, but IR injury was not induced. The O+IR group (n=7) was administered 1 mg/kg of ozone i.p. for seven days before the creation of IR injury (as in the IR group) on the eight day.
Ozone preconditioning
Ozone was generated by an ozone generator machine (Humazon Promedic, Humares® GmbH, Germany), which controls the gas flow rate and ozone concentration in real time using a built-in UV spectrometer. The ozone flow rate was kept constant at 3 L/min in a gas mixture of 97% O2+3% O3, representing a concentration of 60 μg/mL. The volume of the gaseous mix-ture administered to each animal was approxi-mately 3.5-4.5 mL. Tygon polymer tubes and single-use silicon-treated polypropylene syring-es (ozone rsyring-esistant) were used throughout the reaction to ensure containment of ozone and consistency of concentrations. The first dose of ozone was administered seven days before ischemia. In total, seven doses of ozone were administered. The duration of ozone adminis-tration and the number of doses were based on previous studies [6, 19].
IR model
Histomorphological evaluation of renal tissue
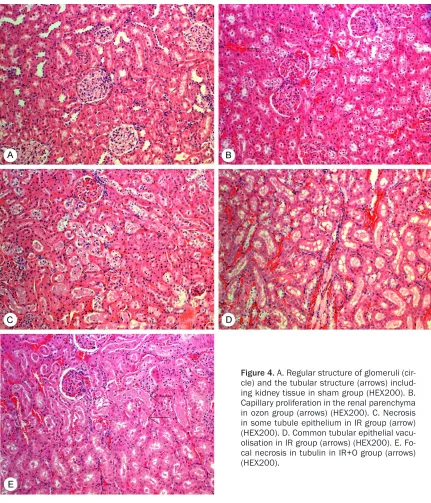
Both kidneys of each animal were removed for histopathological evaluation. The renal tissue samples were fixed in a freshly prepared forma-lin solution at room temperature and processed for embedding in paraffin. The paraffin-embed-ded kidney tissues were sectioned into 4-μm thick sections, placed on polylysine-coated slides, and stained with hematoxylin-eosin (H&E). One pathologist blinded to the animal groups evaluated the renal tissue sections exposed to IR by light microscopy. The following were assessed: structural changes in the proxi-mal tubules (tubular atrophy, loss of the tubular brush border, vacuolization, tubular dilatation, cast formation), mononuclear cell (MNC) infil-tration, glomerular changes (GCs), total score of histopathological damage (TSHD), and ne- crotic and apoptotic cells. Cross-sectional images were scored semiquantitatively in terms of tubulointerstitial damage. The scoring sys-tem used was 0, absent; 1, mild; 2, intermedi-ate; and 3, severe [20]. The results were observed using a BX-51 Research Microscope System and a DP70 Image Acquisition System (Olympus, Tokyo, Japan). The total histopatho-logical injury score per kidney was calculated by adding all the scores.
To evaluate the apoptosis of the renal tissues at the cellular level, the TUNEL (terminal deoxy-nucleotidyl transferase dUTP nick end) assay and Insitu Cell Death (POD, Roche, Germany) kit were used, according to the manufacturer’s instructions [21]. They were visualized digitally at 40× magnification, and counts were made in at least three different regions.
Biochemical analysis
For the biochemical analysis, the renal tissues were washed twice with a cold saline solution, placed in glass bottles, and stored in a deep freezer at -80°C until processing. The frozen renal tissues were homogenized in ice-cold phosphate buffer (pH 7.4) using a homogeniza-tor (Ultra Turrax IKA T18 Basic, IKA Labortecnic, Staufen, Germany). The homogenates were centrifuged at 14,000 rpm (7,530 g) at 4°C for 10 min, and the supernatant was analyzed. The total protein concentration of the renal tissue homogenates was determined using the meth-od of Lowry et al. [22], with bovine serum albu-min as the standard. MDA, GSH-Px, and SOD
were measured in the renal tissue samples, and TOS and TAC were measured in the blood samples. In these analyses, 2 mL of intracardi-ac blood were drawn 2 h after reperfusion. The blood samples were centrifuged at 3,500 rpm for 15 min, and the serum was collected and stored at -80°C until processing.
Measurement of MDA
The lipid peroxidation product and kidney tis-sues were homogenized in 1.15% KCl solution. A 100 µl aliquot of the homogenate was added to a reaction mixture containing 200 µl 8.1% sodium dodecyl sulfate, 1,500 µl 20% acetic acid (pH 3.5), 1,500 µl 0.8% thiobarbituric acid, and 700 µl distilled water. Samples were then boiled for 1 h at 95°C and centrifuged at 3,000 g for 10 min. The absorbance of supernatant was measured by spectrophotometry at 650 nm. The MDA was assayed using a calorimetric reaction with thiobarbituric acid as described by Shin et al. [23]. The concentration of MDA was expressed as nmol/mg protein.
Measurement of SOD
The activity of SOD was evaluated by inhibition of nitro blue tetrazolium reduction by superox-ide anions generated by the xanthine-xanthine oxide system, according to the method of Sun et al. [24]. A commercial assay kit (Nnjing Jiancheng Biological Product, Nanjing China) was used. The calculated SOD activity was expressed as U/g protein.
Measurement of GSH-Px
A Cayman GSH-Px assay kit (Cayman Chemical, USA) was used to measure the activity of GSH-Px. The efficacy of the treatment was assessed by measuring the tissue levels of GSH-Px using the method of Paglia and Valentine [25]. GSH-Px activity is coupled to the oxidation of NADPH by glutathione reductase. The oxidation of NADPH at 37°C was measured using a spectro-photometer. The absorbance at 340 nm was recorded, and GSH-Px activity was recorded as U/g protein in the tissue sample.
Measurement of the TAC
reactions, which are initiated by the produced
[image:4.612.92.525.76.254.2]hydroxyl radicals, is measured. The TAC was measured using an Aeroset 2.0 analyzer and Cayman’s total antioxidant score kit (cayman
Figure 1. The histopathological evaluation markers of the four groups. a: P<0.0001 compared with the 1st group. b:
P<0.0001 compared with the 2nd group. c: P<0.0001 compared with the 3rd group. d: P<0.0001 compared with the
4th group.
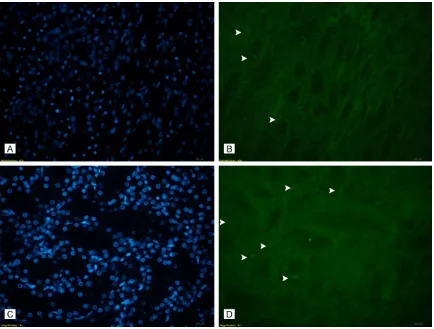
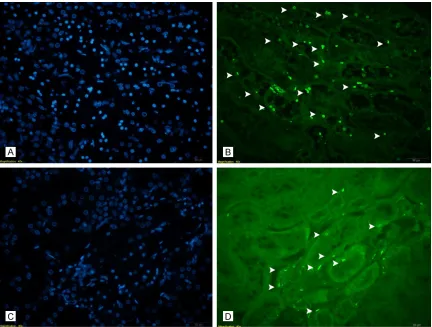
Figure 2. Apoptotic cell labeling with TUNEL (A) (S Group, DAPI), (B) (S Group, TUNEL): very few apoptotic cells were
[image:4.612.90.524.313.642.2]Chemical, USA) according to the manufactur-er’s instructions. The results were expressed as μmol Trolox Eq/L. The reaction rate was cali-brated with Trolox, which is widely used as a standard in TAC measurement assays.
Measurement of the TOS
The serum TOS levels of the blood samples were determined using an automated colori-metric measurement method developed by Erel [27]. The serum TOS levels of the blood sam-ples were determined using an Aeroset 2.0 analyzer and a commercial Cayman’s TOS kit (Cayman Chemical, USA). The assay is based on the oxidation of ferrous ion to ferric ions in the presence of various oxidant species in acid-ic medium and the measurement of the ferracid-ic ions by xylenol orange. The color intensity, which denotes the total amount of oxidant mol-ecules in the sample, was measured spectro-photometrically. The assay was calibrated with hydrogen peroxide (H2O2). The results are
expressed as micromolar hydrogen peroxide equivalent per liter (μmol H2O2 Eq/L).
Statistical analysis
Statistical analysis was performed using SPSS 15.0 for Windows (SPSS Inc., IL, USA). The results are expressed as mean ± standard deviation. The differences in pathological find-ings between the study groups were analyzed using the Kruskal-Wallis test. When an overall statistically significant difference was observed, pairwise comparisons were performed using the Mann-Whitney U test. Results were consid-ered statistically significant if the two-tailed P was <0.05.
Results
Histopathological results (Figures 1-4)
[image:5.612.91.523.70.397.2]Apoptotic index (AI): The AI was significantly dif-ferent between the four groups (P trend
Figure 3. Apoptotic cell labelling staining with TUNEL (A) (IR group, DAPI), (B) (IR group, TUNEL): quite high number
<0.0001) (Table 1). In the pairwise analysis, the AI was higher in the IR group than in the O+IR (P=0.002) and higher in the S (P=0.002 group than in the O (P=0.002). The AI was high-er in the O+IR group than in the S (P=0.002) and O groups (P=0.002). The AI was higher in the S group than in the O group (P=0.002). Mononuclear cell infiltration (MCI): The MCI was significantly different between the four groups (P trend <0.0001) (Table 1). In the pair-wise analysis, the MCI was higher in the IR
group than in the O+IR group (P=0.002) and higher in the S (P=0.002) group than in the O group. The MCI was higher in O+IR group than in the S group, but the difference was not sta-tistically significant (P=0.606) compared to that of the O group (P=0.002). The MCI was higher in the S group than in the O group, but the difference was not statistically different (P=0.591).
[image:6.612.90.521.68.571.2]Proximal tubuler changes (PTC): The PTC was significantly different between the four groups
Figure 4. A. Regular structure of glomeruli
(P trend <0.0001) (Table 1). In the pairwise analysis, the PTC was higher in the IR group than in the O+IR group (P=0.008) and higher in the O group than the S group, but the difference was not statistically significant (P=1.00). The PTC was higher in the O+IR group than in the S (P=0.002) and O groups, but the difference was not statistically significant (P=1.00). Glomerular changes (GCs): The GCs were sig-nificantly different between the four groups (P trend <0.0001) (Table 1). In the pairwise
analy-sis, the level of GCs was greater in the IR group than in the O+IR (P=0.000) group and greater in the O (P=0.000) group than in the S group (P=0.000).
[image:7.612.91.523.86.165.2]Total score of histopathological damage (TSHD): TSHD was significantly different be- tween the four groups (P trend <0.0001) (Table 1). In the pairwise analysis, TSHD was higher in the IR group than in the O+IR (P=0.001) group and higher in the S group (P=0.000) than in the O group (P=0.000). TSHD was higher in the IR Table 1. The histopathological evaluation markers of the four groups
Parameter (S) (N=7) (IR) (N=7) (O) (N=7) (O+IR) (N=7) Ptrend
AI 4.81±0.48b,c,d 20.7±1.23a,c,d 2.2±0.42a,b,d 10.5±0.66a,b,c <0.0001
MCI 0.43±0.53b 1.43±0.53a,c,d 0.29±0.48b,d 0.57±0.53b,c <0.0001
PTC 0.0±0.00d 2.29±0.75d 0.0±0.00 1.00±0.57a,b <0.0001
GC 0.0±0.00b,c 1.14±0.37a,c,d 0.0±0.00a,b 0.0±0.00b <0.0001
TSHD 0.43±0.53b,d 4.86±0.90a,c,d 0.29±0.48b,d 1.57±0.78a,b,c <0.0001
a: P<0.0001 compared with the S group. b: P<0.0001 compared with the IR group. c: P<0.0001 compared with the O group. d:
[image:7.612.95.524.217.385.2]P<0.0001 compared with the O+IR group.
Figure 5. The oxidative markers of the four groups. a: P<0.0001 compared with the sham group. b: P<0.0001
com-pared with the IR group. c: P<0.0001 compared with the Ozone group. d: P<0.0001 compared with the Ozone+IR
group.
Table 2. The biochemical markers of the four groups
Parameter (S) (N=7) IR (N=7) O (N=7) O+IR (N=7) Ptrend
Superoxide dismutase 1.26±0.06b,c,d 0.91±0.12a,c,d 1.64±0.03a,b,d 1.46±0.01a,b,c <0.0001
Glutathione peroxidase 1.57±0.4c,d 1.59±0.03c,d 2.50±0.03a,b,d 2.11±0.02a,b,c <0.0001
Malondialdehyde 6.06±0.09b,c,d 7.39±0.22a,c,d 4.50±0.31a,b,d 5.44±0.14a,b,c <0.0001
Total antioxidant capacity 2.50±0.04b,c,d 1.84±0.03a,c,d 4.58±0.34a,b,d 3.52±0.19a,b,c <0.0001
Total oxidant status 8.35±0.36b,c,d 14.27±0.63a,c,d 4.93±0.53a,b,d 7.35±0.54a,b,c <0.0001
a: P<0.0001 compared with the S group. b: P<0.0001 compared with the IR group. c: P<0.0001 compared with the O group. d:
[image:7.612.90.524.459.542.2]group than in the S (P=0.001) and O (P=0.001) groups. TSHD was higher in the S group than in the O group, but the difference was not statisti-cally significant (P=0.591).
Biochemical results (Figure 5)
SOD: The level of SOD was significantly differ-ent between the four groups (P trend: 0.001) (Table 2). In the pairwise comparison, the level of SOD was significantly higher in the O group than in the S group, and higher in the IR group than in the O+IR group (P for all 0.001). The level of SOD was significantly higher in the O+IR group than in the S and IR groups (P for all 0.001). The level of SOD was also significantly higher in the S group than in the IR group (P=0.001).
GSH-Px: The level of GSH-Px was significantly different between the four groups (P trend< 0.0001) (Table 2). In the pairwise comparison, the level of GSH-Px was significantly higher in the O group than in the S group and higher in the IR group than in the O+IR group (P for all 0.001). The level of GSH-Px was significantly higher in the O+IR group than in the S and IR groups (P for all 0.001). The level of GSH-Px was not significantly different between the S and IR groups (P=0.445).
MDA: The level of MDA was significantly differ-ent between the four groups (P trend<0.0001) (Table 2). In the pairwise comparison, the level of MDA was significantly higher in the IR group than in the S, O and O+IR groups (P for all, 0.001). The level was also higher in the S group than in O and O+IR groups (P for all, 0.001). The level of MDA was higher in the O+IR group than in the O group (P=0.001).
TAC: The TAC was significantly different between the four groups (P trend<0.0001) (Table 2). In the pairwise comparison, the TAC was signifi-cantly higher in the O group than in the S, IR, and O+IR groups (P for all, 0.001). The TAC was significantly lower in the IR group than in the S and O+IR groups (P=0.001 and 0.001, respec-tively). The TAC was also lower in the S group than in the O+IR group (P=0.001).
TOS: The TOS was significantly different between the four groups (P trend <0.0001) (Table 2). In the pairwise comparison, the TOS was significantly higher in the IR group than in the S, O and O+IR groups (P for all, 0.001). The
TOS was also higher in the S group than in the O and O+IR groups (P for all, 0.001). The TOS was higher in the O+IR group than in the O group (P=0.001).
Discussion
Renal IR injury is a common cause of acute renal injury, and it is characterized by high mor-tality and morbidity [28]. Although there have been many studies of renal IR injury [29, 30], a therapeutic strategy for renal IR has not been developed until recently. Oxidative stress is the main mechanism underling IR injury [31]. It results in free oxygen radicals attacking the reperfused tissue. The binding of these radi-cals to lipids, proteins, and nucleic acids on cell membranes causes lipid peroxidation, leading
to tissue damage [31]. The expression of
adhe-sion molecules in response to vascular dys-function results in transmigration of polymor-phonuclear leukocytes into renal tissues and immune system activation, especially of T cells and cell adhesion molecules [32]. The subse-quent production of reactive oxygen species (ROS) and nitric oxide contribute to the destruc-tion and dysfuncdestruc-tion of renal tissue [32]. Low doses of ozone can promote oxidative precon-ditioning by enhancing the activity antioxidant endogenous systems, thereby contributing to the preservation of the redox state of the cell [5]. A previous study demonstrated that ozone supports cellular antioxidant systems and pre-vents septic shock by mediating the expression of ROS/RNS [33]. According to the results of the present study, ozone used alone or in com-bination significantly reduced MNC infiltration, glomerulotubular changes, and total damage scores in IR injury. The significant decrease in apoptosis in the O+IR group suggested that could be useful in the treatment of IR.
In the present study, lipid peroxidation, which is an oxidative stress marker of MDA, was increased in the kidneys of the IR group. Levels of the antioxidant enzymes SOD and GSH-Px were elevated in the O group. These results are similar to those of previous studies, which reported that ozone prevented apoptosis and exerted an anti-inflammatory effect in renal IR injury [33] and that ozone decreased long-term renal fibrosis in IR injury [34].
TOS levels after both IR and ozone exposure. The significant elevation in the plasma TOS level in the IR groups may be related to an increase in renal oxidant stress after the IR injury. The plasma TAC of the animals treated with ozone was remarkably increased, which suggests augmentation of renal antioxidant defenses. A previous study also reported a sig-nificant reduction in the serum TAC level of an IR group, in addition to increases in plasma MDA concentrations [35].
The results of the present study indicated that ozone pretreatment activated renal antioxidant mechanisms, which may be involved in confer-ring renal protection against IR injury. The pre-treatment with ozone before IR injury decreased oxidative injury, as demonstrated by significant-ly reduced serum MDA concentrations in the O group. After the administration of ozone, it immediately dissolves in plasma and reacts with macromolecular glycoproteins, such as carbohydrates and polypeptides. The oxidation of these compounds results in the formation of H2O2 and lipid oxidation products, which act as electron donors. H2O2 acts immediately and disappears (early and short-acting messenger) and is able to act as an ozone messenger, which acts as a regulator of signal transduction and is an important mediator of host defense and immune responses [5]. In contrast to H2O2, lipid oxidation products are distributed through-out the tissues, and they have long-lasting effects, stimulating the innate immune system and aiding cell survival in response to IR injury [36]. The beneficial actions of ozone havebeen demonstrated previously, with the gas used as a therapeutic agent in experimental and clinical
studies [2, 3, 6, 10]. Our previous in vivo study
showed that ozone attenuated intestinal IR injury [19]. We also showed that ozone amelio-rated eye IR injury, cochlea IR injury, and flap IR injury (unpublished data).
Renal IR injury associated with renal transplan-tation is an unresolved problem in clinical prac-tice. The experimental model used in this work mimicked the IR injury that occurs during renal transplantation procedures. The model can be used to shed light on complex processes of acute renal damage that cannot be studied in humans due to ethical concerns. The mecha-nisms underlying the positive effects of ozone are not fully understood. However, various
mechanisms have been proposed [36]. Among these, the most important is up-regulation of cellular antioxidant enzyme activity and regula-tion of ROS [37], which protects the host against pathophysiological conditions mediat-ed by ROS.
In the current study, the application of ozone i.p. for seven days had a beneficial effect on both antioxidant and histopathological values in renal hypoxic/reperfusion injury. In common with the findings of an earlier study by Oztosun et al. [15], ozone protected renal cells and life-time against IR injury. In contrast to the study by Oztosun et al., TAC, which is an antioxidant marker, and TOS, which is an oxidative marker, were evaluated in the present study. In addi-tion, the levels of the antioxidants SOD and GSH-Px and those of the oxidant MDA in kidney tissue were evaluated. The results of these analyses corroborated the histopathological findings and the results of other biochemical tests.
In this study, lipid peroxidation, which is an oxi-dative stress marker for MDA, was increased in the kidneys of the IR-induced rats. The activi-ties of the antioxidant enzymes SOD and GSH-Px were also increased in the ozone treated groups. These results are similar to those reported in previous studies of IR injury in other organs, such as liver [2], heart [6], soft tissue [38], intestines [19], and rat skeletal muscle [39]. Ozone was also shown to protect against IR injury in a testicular torsion model [40] and ovary torsion model [10]. The results of the present study are consistent with those of the above studies, with ozone providing protection against renal IR injury. In clinical use, particu-larly in liver, kidney, eye, and composite tissue transplantation, the administration of ozone prior to transplantation may provide protection from IR injury, which occurs frequently in such procedures. However, the use of ozone in humans remains controversial. Therefore, its clinical use is restricted. Further studies of the preventive and protective effects of ozone in patients with IR injury are required.
administering ozone both before and after IR injury would yield more reliable results. To shed light on this issue, we administered ozone before and after IR in a study of the effect of ozone on flap viability (unpublished data). The results demonstrated the healing effect of ozone on IR injury. A second limitation of this study is that the animals were not housed in metabolic cages. Thus, this study lacks data on other well-documented renal injury biomarkers, such as creatinine clearance and urine microprotein.
Conclusion
Various studies have shown that ozone therapy is beneficial for the treatment of different clini-cal entities [2, 3, 6, 10, 11]. The findings of the present study suggest that ozone may provide renal protection against IR injury by decreasing tissue damage and increasing the activity of antioxidant endogenous systems in a rat model. Further studies are required to explain the mechanisms mediating the protective effect of ozone on renal IR injury.
Disclosure of conflict of interest
None.
Abbreviations
S, sham operated group; TAC, total antioxidant capacity; IR, ischemia reperfusion; PS, physio-logical saline; O, O3, ozone; MNC, mononuclear cell; O+IR, ozone+ischemia reperfusion group; GCs, glomerular changes; SOD, superoxide dis-mutase; TSHD, total score of histopathological damage; GSH-Px, glutathione peroxidase; MDA, Malondialdehyde; TUNEL, terminal deoxynucle-otidyl transferase dUTP nick end; H2O2, hydro-gen peroxide; TOS, total oxidant score; AI, Apoptotic index; PTC, Proximal tubuler changes; GC, Glomeruler changes.
Address correspondence to: Oznur Kal, Department
of Nephrology, Baskent University Medical Faculty Konya Hospital, Konya, Turkey. Tel: +905065167270; E-mail: dr.oznurkal@yahoo.com
References
[1] León OS, Menéndez S, Merino N, Castillo R, Sam S, Pérez L, Cruz E, Bocci V. Ozone oxida-tive preconditioning: a protection against cel-lular damage by free radicals. Mediators In-flamm 1998; 7: 289-94.
[2] Ajamieh HH, Menéndez S, Martínez-Sánchez G, Candelario-Jalil E, Re L, Giuliani A, Fernán-dez OS. Effects of ozone oxidative precondi-tioning on nitric oxide generation and cellular redox balance in a rat model of hepatic isch-aemia-reperfusion. Liver Int 2004; 24: 55-62. [3] Martinez G, Al-Dalain SM, Menendez S,
Guil-iani A, Leon OS. Ozone treatment reduces blood oxidative stress and pancreas damage in a streptozotocin-induced diabetes model in rats. Acta Farm Bonaerense2005; 24: 491-7. [4] Torossian A, Ruehlmann S, Eberhart L,
Midde-ke M, Wulf H, Bauhofer A. Pre-treatment with ozonized oxygen (O3) aggravates inflammation in septic rats. Inflamm Res 2004; 53 Suppl 2: S122-5.
[5] Zamora ZB, Borrego A, López OY, Delgado R, González R, Menéndez S, Hernández F, Schulz S. Effects of ozone oxidative preconditioning on TNF-alpha release and antioxidant-prooxi-dant intracellular balance in mice during endo-toxic shock. Mediators Inflamm 2005; 2005: 16-22.
[6] Merin O, Attias E, Elstein D, Schwalb H, Bitran D, Zimran A, Silberman S. Ozone administra-tion reduces reperfusion injury in an isolated rat heart model. J Card Surg 2007; 22: 339-42.
[7] Salmon M, Koto H, Lynch OT, Haddad EB, Lamb NJ, Quinlan GJ, Barnes PJ, Chung KF. Prolifera-tion of airway epithelium after ozone exposure: effect of apocynin and dexamethasone. Am J Respir Crit Care Med 1998; 157: 970-7. [8] Toward TJ, Broadley KJ. Airway function,
oede-ma, cell infiltration and nitric oxide generation in conscious ozone-exposed guinea-pigs: ef-fects of dexamethasone and rolipram. Br J Pharmacol 2002; 136: 735-45.
[9] Re L, Mawsouf MN, Menéndez S, León OS, Sánchez GM, Hernández F. Ozone therapy: clinical and basic evidence of its therapeutic potential. Arch Med Res 2008; 39: 17-26. [10] Aslan MK, Boybeyi Ö, Şenyücel MF, Ayva Ş,
Kısa Ü, Aksoy N, Soyer T, Cesur Ö, Çakmak M. Protective effect of intraperitoneal ozone ap-plication in experimental ovarian ischemia/re-perfusion injury. J Pediatr Surg 2012; 47: 1730-4.
[11] Di Filippo C, Capuano A, Rinaldi B, Luongo M, Lettieri B, Rossi F, D’Amico M. Intraperitoneal oxygen/ozone treatment decreases the forma-tion of experimental postsurgical peritoneal adhesions and the levels/activity of the local ubiquitin-proteasome system. Mediators In-flamm 2011; 2011: 606718.
STE-MI Trial): rationale and study design. Clin Car-diol 2015; 38: 259-66.
[13] Hsu CY, Chertow GM, McCulloch CE, Fan D, Or-doñez JD, Go AS. Nonrecovery of kidney func-tion and death after acute on chronic renal failure. Clin J Am Soc Nephrol 2009; 4: 891-8. [14] Tsung A, Hoffman RA, Izuishi K, Critchlow ND,
Nakao A, Chan MH, Lotze MT, Geller DA, Billiar TR. Hepatic ischemia/reperfusion injury in-volves functional TLR4 signaling in nonparen-chymal cells. J Immunol 2005; 175: 7661. [15] Oztosun M, Akgul EO, Cakir E, Cayci T, Uysal B,
Ogur R, Ozcan A, Ozgurtas T, Guven A, Kork-maz A. The effects of medical ozone therapy on renal ischemia/reperfusion injury. Ren Fail 2012; 34: 921-5.
[16] Caliskan B, Guven A, Ozler M, Cayci T, Ozcan A, Bedir O, Surer I, Korkmaz A. Ozone therapy prevents renal inflammation and fibrosis in a rat model of acute pyelonephritis. Scand J Clin Lab Invest 2011; 71: 473-80.
[17] Demirbag S, Uysal B, Guven A, Cayci T, Ozler M, Ozcan A, Kaldirim U, Surer I, Korkmaz A. Ef-fects of medical ozone therapy on acetamino-phen-induced nephrotoxicity in rats. Ren Fail 2010; 32: 493-7.
[18] Amasyali AS, Akkurt A, Kazan E, Yilmaz M, Erol B, Yildiz Y, Erol H. The protective effect of tadalafil on IMA (ischemia modified albumin) levels in experimental renal ischemia-reperfu-sion injury. Int J Clin Exp Med 2015; 8: 15766-72.
[19] Onal O, Yetisir F, Sarer AE, Zeybek ND, Onal CO, Yurekli B, Celik HT, Sirma A, Kılıc M. Prophylac-tic Ozone Administration Reduces Intestinal Mucosa Injury Induced by Intestinal Ischemia-Reperfusion in the Rat. Mediators Inflamm 2015; 2015: 792016.
[20] Bagcik E, Ozkardesler S, Boztas N, Ugur Ergur B, Akan M, Guneli M, Ozbilgin S. Effects of dex-medetomidine in conjunction with remote ischemic preconditioning on renal ischemia-reperfusion injury in rats. Rev Bras Anestesiol 2014; 64: 382-90.
[21] Vyvyan Howard C. Unbiased Stereology. BIOS Scientific Publishers 1998. pp. 20.
[22] Lowry OH, Rosebrough NJ, Farr A, Randall AR. Protein measurement with folin phenol re-agent. J Biol Chem 1951; 193: 256-75. [23] Shin MS, Angel MF, Im MJ, Manson PN. Effect
of 21-Aminosteroid U74389F on skin- ap sur-vival after secondary ischemia. Plast Reconstr Surg 1994; 94: 661-6.
[24] Sun Y, Oberley LW, and Li Y. A simple method for clinical assay of superoxide dismutase. Clin Chem 1988; 34: 497-500.
[25] Paglia DE, Valentine WN. Studies on the quan-titative and qualitative characterization of
erythrocyte glutathione peroxidase. J Lab Clin Med 1967; 70: 158-69.
[26] Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cat-ion. Clin Biochem 2004; 37: 277-85.
[27] Erel O. A new automated colorimetric method for measuring total oxidant status. Clin Bio-chem 2005; 38: 1103-11.
[28] Landry GJ, Lau IH, Liem TK, Mitchell EL, Mo-neta GL. Adjunctive renal artery revasculariza-tion during juxtarenal and suprarenal abdomi-nal aortic aneurysm repairs. Am J Surg 2010; 199: 641-5.
[29] Wen X, Murugan R, Peng Z, Kellum JA. Patho-physiology of acute kidney injury:a new per-spective. Contrib Nephrol 2010; 165: 39-45. [30] Wang Z, Liu Y, Han Y, Guan W, Kou X, Fu J, Yang
D, Ren H, He D, Zhou L, Zeng C. Protective ef-fects of aliskiren on ischemia-reperfusion in-duced renal injury in rats. Eur J Pharmacol 2013; 718: 160-6.
[31] McCord JM. The evolution of free radicals and oxidative stress. Am J Med 2000; 108: 652-9. [32] Rabb H, O’Meara YM, Maderna P, Coleman P,
Brady HR. Leukocytes, cell adhesion mole-cules and ischemic acute renal failure. Kidney Int 1997; 51: 1463-8.
[33] Zamora ZB, Borrego A, López OY, Delgado R, González R, Menéndez S, Hernández F, Schulz S. Effects of ozone oxidative preconditioning on TNF-alpha release and antioxidant-prooxi-dant intracellular balance in mice during endo-toxic shock. MediatorsInflamm 2005; 2005: 16-22.
[34] Wang L, Chen H, Liu XH, Chen ZY, Weng XD, Qiu T, Liu L, Zhu HC. Ozone oxidative precondition-ing inhibits Renal fibrosis induced by ischemia and reperfusion injury in rats. Exp Ther Med 2014; 8: 1764-8.
[35] Kongkham S, Sriwong S, Tasanarong A. Protec-tive effect of alpha tocopherol on contrast-in-duced nephropathy in rats. Nefrologia 2013; 33: 116.
[36] Bocci VA. Scientific and medical aspects of ozone therapy: State of the art. Arch Med Res 2006; 37: 425-35.
[37] Bocci V. Ozone as Janus: this controversial gas can be either toxic or medically useful. Media-tors Inflamm 2004; 13: 3-11.
[39] Koca K, Yurttaş Y, Yıldız C, Caycı T, Uysal B, Korkmaz A. Effect of hyperbaric oxygen and ozone preconditioning on oxidative/nitrosative stress induced by tourniquetischemia/reperfu-sion in rat skeletal muscle. Acta Orthop Trau-matol Turc 2010; 44: 476-83.