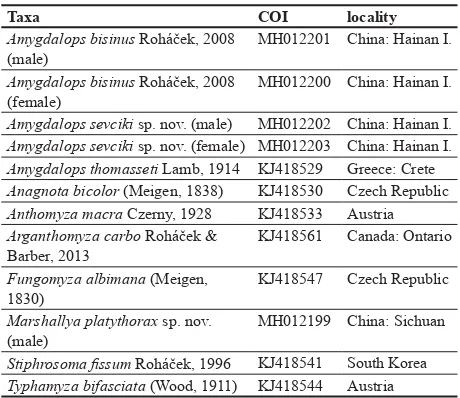
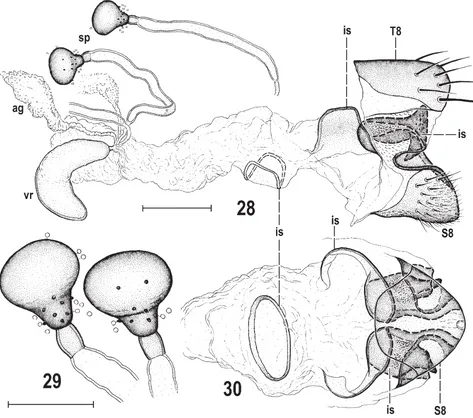
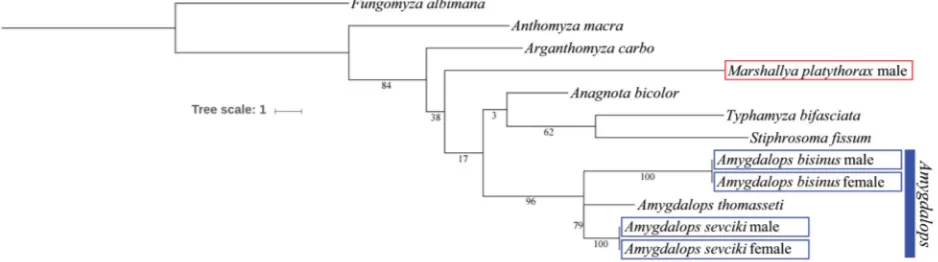
First Anthomyzidae (Diptera) from China: a new genus, six new species and new records
Full text
Figure
Related documents
We will also undoubtedly see a strengthening of outsourced incident response adoption, as firms plan for breaches by forging relationships with services providers, typically on
The management of capital structure, which reflects the financial leverage of companies and the way by which the capital of company is applied by the managers, is one of the
Next, we calculated the change in each local authority in overall cycling modal share from 2001 to 2011, and the changes in each local authority in the ratios of female cyclists
Yet as my case study shows, culture plays a significant role: in the Blue Mountains, a culture of ambivalent expectation regarding fire risk, characteristically Australian
wanie biochemicznych przyczyn tego stanu. Badania dotyczące genezy przyczyn choroby uwarunkowanej genetycznie, znanej jako hiper-cholesterolemia rodzinna, będącej
Appropriate timing of tooth extraction, the use of hard/soft tissue aug- mentation techniques and ideal implant positioning help to facilitate a good aesthetic and functional
However, the deep infiltration of Haitian historical events—such as the United States Occupation of Haiti from 1915 to 1934 and the Haitian Massacre in the Dominican Republic in
The new species is clearly distinguished from this species by the 9th and 10th antennomeres of the new species being as long as wide but transverse in scapularis; the abdomen of