ARTICLE
Presentation and Echocardiographic Markers of
Neonatal Hypertensive Cardiomyopathy
Amy L. Peterson, MDa, Peter C. Frommelt, MDa, Kathy Mussatto, BSNb
aDepartment of Pediatrics, Medical College of Wisconsin, Milwaukee, Wisconsin;bHerma Heart Center, Children’s Hospital of Wisconsin, Milwaukee, Wisconsin
The authors have indicated they have no financial relationships relevant to this article to disclose.
ABSTRACT
BACKGROUND.Systemic hypertension is a rare but important cause of neonatal heart failure. It is critical that this etiology be recognized and distinguished from other causes of myocardial dysfunction in young infants, because diagnostic studies, treatments, and prognoses are quite different.
METHODS.Between 1991 and 2005, 11 neonates were diagnosed as having neonatal cardiomyopathy and systemic hypertension through retrospective review of the Children’s Hospital of Wisconsin database.
RESULTS.All infants in the cohort were found to have systemic hypertension (blood pressure of ⬎95th percentile for gestational age and weight). Causes included renovascular disease (n⫽9), aortic obstruction secondary to thrombus (n⫽1), and steroid use (n⫽1). Echocardiography was performed at presentation for all patients because of cardiomegaly and/or hypertension. Echocardiographic findings were consistently striking for (1) left ventricular systolic dysfunction without chamber dilation, (2) concentric left ventricular hypertrophy, (3) left atrial dila-tion, and (4) aortomegaly. No anatomic aortic arch obstruction was identified, but Doppler findings for the descending thoracic aorta were uniformly consistent with elevated systemic vascular resistance. One patient died as a result of overwhelming thrombotic disease; all other patients responded to afterload reduction therapy with normalization of left ventricular systolic function during infancy.
CONCLUSIONS.Hypertensive cardiomyopathy can present in neonates with nonspe-cific symptoms and systemic hypertension. Because sometimes hypertension in infants is ignored or misinterpreted as agitation, echocardiography can provide critical markers of the disease.
www.pediatrics.org/cgi/doi/10.1542/ peds.2006-0631
doi:10.1542/peds.2006-0631
Key Words
cardiomyopathy, infant
Abbreviations
LA—left atrial LV—left ventricular
LVH—left ventricular hypertrophy
Accepted for publication Mar 31, 2006 Address correspondence to Peter C. Frommelt, MD, Medical College of Wisconsin, 8700 Watertown Plank Rd, Milwaukee, WI 53226. E-mail: pfrom@mcw.edu
S
YSTEMIC HYPERTENSION ISa rare but important cause of heart failure in neonates. Diagnostic studies, treatments, and prognoses are quite different for heart failure secondary to hypertension versus other causes; therefore, early recognition and prompt treatment are crucial for good outcomes in this group. In particular, systolic dysfunction secondary to hypertension can mimic dilated cardiomyopathy, a primary disorder of myocardial contractility. Therefore, echocardiographic evidence of poor myocardial function should not be presumed to be attributable to intrinsically defective myocardium; hypertension must be ruled out first.Echocardiographic evaluation of neonates with hy-pertension at Children’s Hospital of Wisconsin has led to the anecdotal observation of poor left ventricular (LV) systolic function, aortomegaly, and abnormal Doppler flow patterns. The aim of this study was to characterize echocardiographic findings for hypertensive neonates, specifically focusing on left heart and aortic dimensions, as well as indices of LV function.
METHODS
Between 1991 and 2005, 11 neonates were diagnosed as having neonatal cardiomyopathy and systemic hyper-tension through retrospective review of the Children’s Hospital of Wisconsin database. Eligible infants pre-sented with clinical and/or echocardiographic evidence of poor myocardial function or congestive heart failure between 0 and 28 days of age. These infants had docu-mented pretreatment blood pressure of ⬎95% confi-dence interval for postconceptual age and birth weight.1,2
Infants with congenital cardiac or extracardiac defects, known genetic or metabolic derangements, or gesta-tional age of ⬍37 weeks were excluded from analysis. Infants without echocardiographic evaluations per-formed on the day of presentation were also excluded. Institutional review board approval was granted for the study.
Chart review was performed to document patient presentation, cause of hypertension, clinical course, and outcome. Echocardiograms obtained on the day of pre-sentation were reviewed to obtain measurements of left atrial (LA) long-axis diameter, LV short-axis end systolic and end diastolic dimensions, and aortic end systolic diameter at 5 points, namely, the aortic annulus, the ascending aorta proximal to the innominate artery (as-cending aorta), the proximal aortic arch between the innominate and carotid arteries (transverse aortic arch), the aortic isthmus, and the abdominal aorta at the level of the diaphragm; values were compared with age- and size-matched normal values withzscores.3LV
shorten-ing fraction (LV end diastolic dimension ⫺LV end sys-tolic dimension/LV end diassys-tolic dimension) and ejec-tion fracejec-tion (LV end diastolic volume⫺LV end systolic volume/end diastolic volume) were calculated for each
patient. LV mass was calculated by using the area-length method indexed to body surface area.4
RESULTS
All infants in the cohort were found to have systemic hypertension as part of their inclusion criteria. Causes included renovascular disease (n⫽9), aortic obstruction secondary to thrombus (n⫽1), and steroid use (n⫽1). Many of the infants with renovascular disease (4 of 9 infants) had underlying thrombotic disease as a direct cause of their renovascular compromise.
Ages at presentation varied (4 –13 days of life), and initial symptoms were frequently nonspecific (6 of 11 infants were discharged after birth and returned with poor oral intake/vomiting, respiratory distress, and/or fever that led to readmission). The other infants were monitored in a NICU or PICU setting and had elevated blood pressures or heart failure noted during their clin-ical course (Table 1).
Echocardiography was performed at presentation for all patients, because of clinical concerns regarding car-diomegaly (noted on chest radiographs) and/or hyper-tension. Echocardiographic findings were consistently striking for (1) LV systolic dysfunction without chamber dilation (shortening fraction: 16⫾6%; normal:⬎28%; ejection fraction: 35 ⫾ 6%; normal: ⬎60%; LV end diastolic dimension: 2.1⫾0.3 cm; meanzscore: 0.30), (2) concentric LV hypertrophy (LVH) (mass: 61.8⫾19.3 g/m2; normal: 47.4 ⫾ 6.2 g/m2),5 (3) LA dilation (LA
diameter: 1.9 ⫾ 0.3 cm; mean z score: 2.86), and (4) aortomegaly (ascending aortic diameter: 10.4⫾2 mm; meanzscore: 1.84; aortic isthmus diameter: 7.7 ⫾1.8
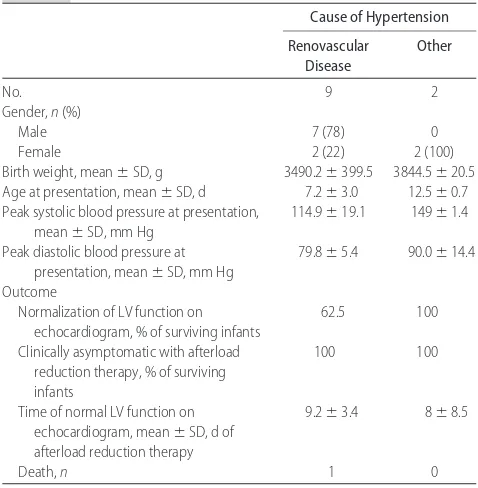
TABLE 1 Characteristics of Neonates
Cause of Hypertension
Renovascular Disease
Other
No. 9 2
Gender,n(%)
Male 7 (78) 0
Female 2 (22) 2 (100)
Birth weight, mean⫾SD, g 3490.2⫾399.5 3844.5⫾20.5
Age at presentation, mean⫾SD, d 7.2⫾3.0 12.5⫾0.7
Peak systolic blood pressure at presentation, mean⫾SD, mm Hg
114.9⫾19.1 149⫾1.4
Peak diastolic blood pressure at presentation, mean⫾SD, mm Hg
79.8⫾5.4 90.0⫾14.4
Outcome
Normalization of LV function on echocardiogram, % of surviving infants
62.5 100
Clinically asymptomatic with afterload reduction therapy, % of surviving infants
100 100
Time of normal LV function on echocardiogram, mean⫾SD, d of afterload reduction therapy
9.2⫾3.4 8⫾8.5
Death,n 1 0
PEDIATRICS Volume 118, Number 3, September 2006 e783
at Viet Nam:AAP Sponsored on August 29, 2020
www.aappublications.org/news
mm; meanzscore: 2.11; abdominal aortic diameter: 8.8 ⫾1 mm; meanzscore: 2.82; normal aortic valve ana-tomic features; annulus dimension: 7.3⫾0.5 mm; mean
z score: 0.37) (Fig 1 and Table 2). No anatomic aortic arch obstruction was identified, but Doppler study find-ings for the descending thoracic aorta were uniformly consistent with elevated systemic vascular resistance, for all 11 infants (Fig 2).
One patient died on day 8 of life, because of over-whelming thrombotic disease secondary to a prothrom-bin gene mutation. Of 10 surviving infants, all became clinically asymptomatic with afterload reduction ther-apy, and 7 had subsequent early echocardiographic eval-uations documenting rapid normalization of LV function (8.9 ⫾ 4.5 days after initiation of afterload reduction therapy), with persistence of aortic dilation (Table 1). All surviving infants were discharged with chronic afterload reduction therapy. One infant later underwent renal transplantation, because of end-stage renal disease, at 2 years of age. All surviving infants with identified throm-botic disease were discharged with low-molecular weight heparin injections, although none had an iden-tifiable cause, despite hematologic evaluation.
DISCUSSION
Neonates are considered to be hypertensive if their blood pressure is⬎95% confidence interval for infants of sim-ilar gestational age and size.1It is estimated that 2% of
infants in NICUs and 0.2% to 0.3% of healthy term infants have hypertension.6Neonatal systemic
hyperten-sion is usually secondary to renovascular or thrombotic events, which can be iatrogenic, often resulting from umbilical line placement in the neonatal period. It can also be caused by congenital anomalies such as aortic coarctation, acquired or intrinsic renal disease, or med-ications.1 In older children and adults, typically
hyper-tension causes concentric LVH and thickening of the vascular media. In neonates, however, hypertension has been noted to present acutely as congestive heart failure, often with the appearance of dilated cardiomyopathy.7
Correction of hypertension (either pharmacologically or
through nephrectomy) led to improvement or resolution in the case studies reviewed.8–10
Numerous case reports of systemic neonatal hyper-tension discuss echocardiographic findings. Typically, poor right ventricular and/or LV function is reported, specifically poor systolic function (decreased LV short-ening fraction). Some studies report concentric LVH with a thickened intraventricular septum; others report dilated cardiomyopathy.6–10Other causes of myocardial
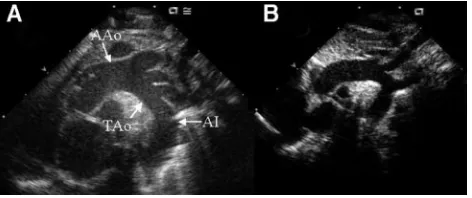
dysfunction in neonates include myocarditis, coronary anomalies, aortic valve disease, and coarctation of the aorta. Of the aforementioned causes, only coarctation has been associated with hypertension; the remaining causes are associated with hypotension. However, coarc-tation can be excluded on the basis of clinical examina-tion and echocardiographic findings. If a hypertensive neonate has myocardial dysfunction and coarctation has FIGURE 1
Representative aortic arch echocardiographic image from a suprasternal notch window in a neonate with hypertensive cardiomyopathy (A) and a normal neonate (B). Dramatic dilation of the aorta can be appreciated in the ascending aorta (AAo), the transverse aorta (TAo), and the aortic isthmus (AI), compared with the normal aorta. Distance markers separated by 1 cm are visible along the left side of each image.
TABLE 2 Aortic Measurements and LV Function, According to Echocardiographic Data
Mean⫾SD zScore, Mean⫾SD
LA diameter, cm 1.89⫾0.27 2.86⫾1.21
Aortic annulus diameter, cm 0.73⫾0.05 0.37⫾0.43
Ascending aorta diameter, cm 1.04⫾0.15 1.84⫾0.85
Proximal aortic arch diameter, cm 0.85⫾0.13 0.24⫾0.79
Aortic isthmus diameter, cm 0.77⫾0.18 2.11⫾1.35
Abdominal aorta diameter, cm 0.88⫾0.10 2.82⫾0.95
LV end diastolic dimension, cm 2.06⫾0.28 0.30⫾0.72
LV ejection fraction, % 35.10⫾6.01
LV shortening fraction, % 15.98⫾6.08
LV mass/BSA, g/m2 61.79⫾19.26
BSA indicates body surface area.
FIGURE 2
been ruled out, then the hypertension should be consid-ered the primary cause of the myocardial dysfunction. Evaluation to rule out thrombotic or renovascular dis-ease should then be initiated.
No documentation of aortomegaly or abnormal aortic Doppler flow patterns in hypertensive human neonates was found in a review of the literature. One study eval-uating aortic luminal diameter and aortic wall thickness demonstrated increased medial thickening in congeni-tally hypertensive mice, but thoracic aortic luminal di-ameters were not statistically larger in hypertensive mice than in their normotensive counterparts.11
The data presented here indicate that, when exposed to elevated arterial blood pressures, the neonatal aorta is capable of rapid significant distension. Okubo et al12
concluded that aortic distensibility varies with age and is low in infants but peaks in children 10 to 15 years of age. Robinson et al13reported that LV mass and aortic
disten-sibility are markers of cardiovascular disease in children with end-stage renal disease; specifically, as hyperten-sion and cardiovascular disease progress, LV mass in-creases and aortic distensibility dein-creases. Our data sug-gest that the opposite is true in neonates. Neonatal hypertension creates significant aortic dilation, which suggests increased distensibility in the neonatal period. Of note, the youngest infant in the study by Okubo et al12 was 7 days of age, and the distribution of neonates
versus infants in the study was not reported.
Hypertensive cardiomyopathy can present in neo-nates with nonspecific symptoms and systemic hyper-tension. Because hypertension in infants is sometimes ignored or misinterpreted as agitation, echocardiography can provide critical markers of the disease. Important findings that should lead to this diagnosis include de-pressed LV systolic function without chamber dilation, elevated LV mass, diastolic dysfunction with significant LA dilation, and aortomegaly with Doppler evidence of increased systemic vascular resistance. These findings should result in rapid recognition of the pathophysio-logic condition and should direct appropriate additional
studies and therapy. Identification of these echocardio-graphic changes associated with hypertension in a neo-nate differentiates this disease process from other causes of neonatal myocardial dysfunction.
REFERENCES
1. Flynn J. Neonatal hypertension: diagnosis and management. Pediatr Nephrol.2000;14:332–341
2. Zubrow AB, Hulman S, Kushner H, et al. Determinants of blood pressure in infants admitted to neonatal intensive care units: a prospective multicenter study. J Perinatol. 1995;15: 470 – 479
3. Sable C, Skeens ME, Martin G, et al. Pediatric normative data andz-scores revisited: echocardiographic data from a heterog-enous population of over 6,000 patients.J Am Soc Echocardiogr. 2004;17:503
4. Deveureux RB, Alonso DR, Lutas EM, et al. Echocardiographic assessment of left ventricular hypertrophy: comparison to nec-ropsy findings.Am J Cardiol.1986;57:450 – 458
5. Joyce JJ, Dickson PI, Qi N, et al. Normal right and left ventric-ular mass development during early infancy. Am J Cardiol. 2004;93:797– 801
6. Skalina ME, Kliegman RM, Fanaroff AA. Epidemiology and management of severe symptomatic neonatal hypertension. Am J Perinatol.1986;3:235–239
7. Mace SE, Hirschfield SS, Riggs TW. Echocardiographic and hemodynamic findings in isolated symptomatic coarctation of the aorta in infancy.Cathet Cardiovasc Diagn.1983;9:363–372 8. Hawkins KC, Watson AR, Rutter N. Neonatal hypertension and
cardiac failure.Eur J Pediatr.1995;154:148 –149
9. McGonigle LR, Beaudry MA, Coe JY. Recovery from neonatal myocardial dysfunction after treatment of acute hypertension. Arch Dis Child.1987;62:614 – 627
10. Saland JM, Mahony L, Baum M. Perinatal renal ischemia resulting in hypertensive cardiomyopathy.Pediatrics.2001;107: 185–187
11. Eccleston-Joyner C, Gray SD. Arterial hypertrophy in the fetal and neonatal spontaneously hypertensive rat. Hypertension. 1988;12:513–518
12. Okubo M, Ino T, Takahashi K, et al. Age dependency of stiff-ness of the abdominal aorta and the mechanical properties of the aorta in Kawasaki disease in children.Pediatr Cardiol.2001; 22:198 –203
13. Robinson RF, Nahata MC, Sparks E, et al. Abnormal left ven-tricular mass and aortic distensibility in pediatric dialysis pa-tients.Pediatr Nephrol.2005;20:64 – 68
PEDIATRICS Volume 118, Number 3, September 2006 e785
at Viet Nam:AAP Sponsored on August 29, 2020
www.aappublications.org/news
DOI: 10.1542/peds.2006-0631 originally published online July 31, 2006;
2006;118;e782
Pediatrics
Amy L. Peterson, Peter C. Frommelt and Kathy Mussatto
Cardiomyopathy
Presentation and Echocardiographic Markers of Neonatal Hypertensive
Services
Updated Information &
http://pediatrics.aappublications.org/content/118/3/e782
including high resolution figures, can be found at:
References
http://pediatrics.aappublications.org/content/118/3/e782#BIBL
This article cites 13 articles, 3 of which you can access for free at:
Subspecialty Collections
http://www.aappublications.org/cgi/collection/cardiology_sub
Cardiology
following collection(s):
This article, along with others on similar topics, appears in the
Permissions & Licensing
http://www.aappublications.org/site/misc/Permissions.xhtml
in its entirety can be found online at:
Information about reproducing this article in parts (figures, tables) or
Reprints
http://www.aappublications.org/site/misc/reprints.xhtml
DOI: 10.1542/peds.2006-0631 originally published online July 31, 2006;
2006;118;e782
Pediatrics
Amy L. Peterson, Peter C. Frommelt and Kathy Mussatto
Cardiomyopathy
Presentation and Echocardiographic Markers of Neonatal Hypertensive
http://pediatrics.aappublications.org/content/118/3/e782
located on the World Wide Web at:
The online version of this article, along with updated information and services, is
by the American Academy of Pediatrics. All rights reserved. Print ISSN: 1073-0397.
the American Academy of Pediatrics, 345 Park Avenue, Itasca, Illinois, 60143. Copyright © 2006 has been published continuously since 1948. Pediatrics is owned, published, and trademarked by Pediatrics is the official journal of the American Academy of Pediatrics. A monthly publication, it
at Viet Nam:AAP Sponsored on August 29, 2020
www.aappublications.org/news