Disease
Sakina B. Naqvi and Allan J. Collins
Infectious complications in individuals with chronic kidney disease (CKD) pose a significant source of morbidity and mortality. The overall scope of major infectious complications has, however, received little attention even though some of these events may be preventable. We reviewed infectious hospitalization rates in the CKD and end-stage renal disease (ESRD) populations, comparing them with the non-CKD and non-ESRD groups. We also reviewed preventive vaccination rates for influenza, pneumonia, and pneumococcal pneumonia to assess areas of potential improvement. We reviewed the medical literature and present findings based on hospitalization rates for pneumonia, sepsis/ bacteremia, and urinary tract infections in the Medicare CKD, ESRD, and non-CKD populations. Vaccination rates were determined from submitted claims for services with specific codes for the vaccinations. Regardless of the primary cause for the development of CKD, primary kidney disease or secondary to hypertension, diabetes mellitus, or other chronic condition, patient outcomes after the development of infections were 3 to 4 times worse than in the non-CKD population. Influenza vaccination rates were 52%, far less than the target of 90%. Pneumococcal pneumonia vaccination rate was only 13.5%, far less than recommended. CKD is associated with significant major infectious complications, which occur at rates 3 to 4 times the general population. Providers can improve prevention by using fewer dialysis catheters and increasing vaccination rates for influenza and pneumococcal pneumonia.
© 2006 by the National Kidney Foundation, Inc.
Index Words: Chronic kidney disease; Infectious complication; Vaccination
C
hronic kidney disease (CKD) is fastemerging as a major public health prob-lem in the 21st century. According to the Na-tional Kidney Foundation, 4.5% of the United States population (more than 14 million peo-ple) suffer from CKD.1New classification sys-tems standardize categories for the various stages of kidney damage. The National Kid-ney Foundation Disease Outcomes Quality Initiative guidelines define CKD as kidney damage or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 for at least 3 months. Three intermediary stages follow, with kidney failure or end-stage renal disease (ESRD), as the final stage, defined by a glo-merular filtration rate of less than 15 mL/min per 1.73 m2.1
Many people within the large group of CKD patients are also affected by cardiovas-cular disease (CVD) as well as infectious com-plications. CVD is in fact twice as common in CKD patients as in the general population, and it advances at twice the rate.2Although a great deal of attention has been paid to CVD, not enough has been said about infectious complications, which closely follow CVD in frequency and seriousness. The 2 complica-tions appear to be closely linked; infection is an inflammatory state and as such may be
implicated in the development of atheroscle-rotic disease,3–5and the risk of the developing CVD is increased in the 6 months after an infection (Fig 1).
The CKD population is predisposed to ad-verse infectious events because of overwhelm-ing uremia, which is associated with alter-ations in primary host defense mechanisms and increases the risk of bacterial infections. Neutrophils exhibit impaired chemotaxis, ox-idative metabolism, phagocytic activity, de-granulation, intracellular killing, and dys-regulated programmed cell death. Factors contributing to neutrophil dysfunction in-clude malnutrition, trace element deficiencies, iron overload, impaired glucose metabolism, hyperparathyroidism, dialysis, and uremic re-tention solutes. These immunologic abnor-malities are complicated by the use of immu-nosuppressive drugs to treat and control
From the Chronic Disease Research Group, Minneapolis Medical Research Foundation, Minneapolis, MN; and Depart-ment of Medicine, University of Minnesota, Minneapolis, MN. Address correspondence to Allan J. Collins, MD, FACP, Chronic Disease Research Group, 914 South Eighth Street, Suite S-206, Minneapolis, MN 55404. E-mail: acollins@cdrg.org
© 2006 by the National Kidney Foundation, Inc. 1548-5595/06/1303-0003$32.00/0
doi:10.1053/j.ackd.2006.04.004
underlying diseases and exacerbated by nutri-tional deficiencies, the dialysis procedure, and the disruption of cutaneous and mucosal bar-riers to infection.6 – 8 Epidemiological studies suggest that ESRD patients have a higher risk of contracting bacterial infections and that the 3 most commonly seen infectious complica-tions are urinary tract infeccomplica-tions (UTI), pneu-monia, and sepsis.
This article sets out to review literature related to CKD and the incidence of these complications in pre- and postintervention populations. Within the CKD population, we discuss patients who reach ESRD and receive hemodialysis, peritoneal dialysis or kidney transplantation. Patients in each of these groups are susceptible to similar infections; however, the different modalities lead to com-plications that vary in magnitude. The infec-tious complications in the non-ESRD popula-tion will also be discussed and compared with the event rates for dialysis patients and the non-CKD population.
Infectious Complications
The incidence of the commonly seen infec-tious complications is approximately 3 times greater among CKD patients who have not yet initiated dialysis than in the general
popula-tion, with UTI, pneumonia, and sepsis in de-scending order of prevalence. Notably, raw death rates after infection follow a different pattern in the CKD and non-CKD patient groups, with sepsis, pneumonia, and UTI in descending order of prevalence for both (Fig 2). The higher UTI susceptibility in the CKD group may be explained, in part, by a greater incidence of urinary obstructions, which in turn leads to infections,5 commonly seen in those with benign prostatic hypertrophy, kid-ney stones. and urinary tract cancers.9Patients with ESRD treated by dialysis have higher annual mortality rates caused by sepsis
com-0 1 2 3 4 5 6 6 12 18 24 30 36 42 48 54 60 Month A d ju s te d r e la tiv e r is k
Fig 1. Congestive heart failure after septicemia. (From American Society of Nephrology [ASN] 2003 United States Renal Data System [USRDS] One Hour Talk, available at www.usrds.org.)
Fig 2. Raw death rates after infection by CKD status. (From American Society of Nephrology [ASN] 2003 United States Renal Data System [USRDS] One Hour Talk, available at
pared with the general population, even after stratification for age, race, and diabetes.
Overall, the annual percentage of mortality secondary to sepsis is approximately 100- to 300-fold higher in dialysis patients.10The uri-nary tract, which may not be recognized as an important source of infection among dialysis patients because of their minimal urine out-put,11 is responsible for the highest rates of hospitalization followed by pulmonary infec-tions (Fig 3). Other potential sources of infec-tion include the skin, the dialysis water treat-ment system, and dialyzer reuse, which can cause septicemia in a small minority of pa-tients.
Hemodialysis patients, during the normal course of treatment, are exposed to several infectious risks, and the majority of patients require at least 1 hospitalization every year for treatment of infections.12The type of vascular access in use plays an important role in the subsequent development of bloodstream in-fections. Central venous catheters signifi-cantly increase the risk of bacteremia in hemo-dialysis patients13; those with temporary catheters have been shown to have a 50% higher risk of septicemia than patients with a native fistula. Maximizing the use of arterio-venous fistula as hemodialysis access is likely to lower infection risk.
Old, nonfunctional, clotted prosthetic arte-riovenous grafts have recently been recog-nized as a frequent cause of bacteremia and morbidity among hemodialysis patients.14 In-fected grafts should be surgically excised without delay and systemic antibiotics deliv-ered. At present, there are no prospective data
to address the question of routine excision of old grafts; a high-risk population should be identified and actively managed.
Chronic hemodialysis has been recognized as a risk factor for the development of infec-tive endocarditis (IE) since the early 1960s,15 and mortality rates for IE in the hemodialysis population are high. The study by Strom et al16 put the relative risk of IE in dialysis pa-tients compared with the general population at 16.9. Prevention and early detection of this complication are imperative.
Compared with hemodialysis patients, in whom the predominant morbidity is from car-diovascular complications, peritoneal dialysis patients are more often hospitalized for infec-tions, with peritonitis secondary to catheter tunnel infections as the most common cause of morbidity.17 Peritonitis usually results from decreased host phagocytic efficiency with de-pressed phagocytosis and bactericidal capac-ity of peritoneal macrophages.18 During epi-sodes of peritonitis, fluid movement is reversed, away from the lymphatics and peri-toneal membrane and toward the cavity. As a result, bloodstream infections are rare, yet there is no mistaking the impact of these in-fections on peritoneal membrane function with the loss of ultrafiltration capacity and subsequent difficulties in fluid removal.
Most peritonitis episodes are caused by gram-positive bacteria.18Staphylococcus aureus
or epidermitis are common, and often associ-ated with a catheter infection, frequently re-quiring catheter removal for resolution.S au-reus infections in peritoneal dialysis patients are especially common in nasal carriers, as the bacteria move from the nasal reservoir to the hands and skin and from there to the access site.
Compared with hemodialysis and perito-neal dialysis patients, kidney transplant recip-ients are at the lowest risk for infection, al-though United States Renal Data System data indicate that the annual percentage of mortal-ity secondary to sepsis is 20-fold higher in kidney transplant recipients than in the gen-eral population.10
Factors contributing to a higher incidence of infection among transplant patients include ex-tremes of age, low body mass index, and the presence of diabetes. Interestingly, the infection Fig 3. Raw infection rates by CKD status. (From
American Society of Nephrology [ASN] 2003 United States Renal Data System [USRDS] One Hour Talk, available at www.usrds.org.)
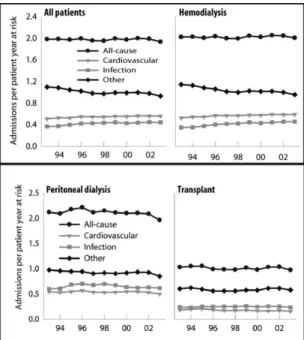
rate is also higher for recipients of deceased-donor organs than for recipients of live-deceased-donor organs. Over the last 10 years, the risk of infec-tion status posttransplant has remained lower than in the population on dialysis (Fig 4), most likely because of better transplant techniques and improved immunosuppression.
Infections in CKD Patients
Although studies have addressed host de-fense abnormalities in the CKD population, the magnitude of the morbidity has received less attention than in the ESRD population.
Figure 5shows infectious hospitalization rates for pneumonia, bacteremia/sepsis, and UTI in the non-CKD, CKD, and dialysis popula-tions.17,19
Compared with the non-CKD population, the rates of pneumonia are 3 times greater in the CKD population and 5 times greater in the dialysis population. Of particular interest, the length of hospital stays for pneumonia in the CKD and dialysis populations are very similar and 4 to 6 times longer than those in the non-CKD population (Fig 5). In fact, pneumo-nia as a complication in the CKD population appears to be more severe than previously appreciated.
Bacteremia/sepsis patterns are also quite different when comparing the non-CKD, CKD, and dialysis populations.20 Admission rates for this infectious event are 4 times greater in the CKD population than in the non-CKD population but almost 10 times greater in the dialysis population than in the non-CKD population. These complication rates highlight the extraordinary risk of infec-tions that CKD patients, especially dialysis patients, face, placing them in an environment of repeated inflammatory stimulation.
Fig 5. Adjusted hospital days: infection. Fig 4. Adjusted all-cause and cause-specific
Preventive Approaches to Major Infections in the CKD Population
The problem of infections in CKD patients may need greater attention, particularly re-lated to vaccinations. Because a randomized trial of influenza or pneumococcal vaccination is unlikely to occur and the complication rates from these vaccines is very low, addressing this major risk with more intensive effort seems reasonable. Currently, only 56% of di-alysis and transplant patients receive influ-enza vaccinations each year despite the Cen-ters for Disease Control and Prevention (CDC) Healthy People 2010 target objective of 90%.
Equally surprising are the low rates of in-fluenza vaccinations in those less than 65 years old. In light of a recent study by Gilbert-son et al21showing significantly lower risk of infectious hospitalizations and infectious death in patients who receive influenza vacci-nations, perhaps performance measures for providers should be implemented to address this gap in a potentially effective treatment.
Rates of pneumococcal vaccination are also surprising low, at only 13.5% per year.17 Tracking these vaccinations is very difficult because no integrated set of information is transmitted to dialysis units from hospitals, nursing homes, or skilled nursing facilities, or from primary care physician offices. Pneumo-coccal vaccinations may be more important than previously considered. Recent reports in-dicate reduced rates of severe pneumococcal infections in the general population during a period of increased vaccinations of children with new pneumococcal vaccine, a so-called herd immunity.22 Repeated pneumococcal vaccination may be nonproblematic; the CDC has not reported adverse effects of vaccina-tions given within 2 years, although the pack-age insert labeling recommends revaccination every 5 years.23 There is little doubt that the CKD and ESRD populations are at substantial risk for pneumonia, yet it appears little is being done to prevent this major source of morbidity and mortality with known vaccina-tions.
Acute hepatitis B virus (HBV) infection in uremic patients on dialysis is generally mild or asymptomatic, but these patients have a higher incidence of chronic HBV infection
compared with immunocompetent per-sons.24,25 Infection control measures and the use of hepatitis B vaccine have significantly reduced the annual incidence of HBV infec-tion among patients on dialysis, from 3.0% to 0.05% between 1976 and 1997.26
Preventive strategies to reduce the future incidence of IE in long-term dialysis patients should be used and include scrupulous atten-tion to asepsis in line inseratten-tion and toileting, antibiotic locks in dialysis catheters,27 vacci-nation against staphylococci (though uremic patients characteristically respond poorly to this immunization),28and consideration of us-ing antibiotic-impregnated dual-lumen tun-neled catheters when available.29
The incidence of diabetes mellitus as a cause of ESRD has increased, as have related complications. Preventive techniques that may be used by dialysis centers include foot assessments, laboratory testing and mainte-nance of strict glycemic control, diabetes self-management education, nutritional counsel-ing, and annual eye examinations.
Conclusion
Despite improvements in infection-control practices and dialysis techniques, bacterial and viral infections are a major cause of mor-bidity and mortality among patients on long-term hemodialysis or peritoneal dialysis; many of these deaths may be vaccine prevent-able.6,30 The Advisory Committee on Immu-nization Practices currently recommends a single 0.5 mL dose of the 23-valent pneumo-coccal polysaccharide vaccine administered intramuscularly or subcutaneously to all dial-ysis patients 2 years of age or older22,31 and the influenza vaccine administered annually, before the beginning of the influenza season, for all dialysis patients 6 months of age or older. Topical mupirocin andS aureus conju-gate vaccine use have shown promise as alter-native prevention methods in those patients for whom catheter use is unavoidable.32 The need for preventive measures to reduce pneu-monia seems obvious, and they can be imple-mented in the dialysis units.
Patients, families, and caregivers should be educated on infectious risks through raised general public awareness and awareness
within the CKD community. More meticulous patient management by medical professionals and increased attention to infections and their prevention may reduce at least 1 source of inflammatory stimulation in the CKD popula-tion, which may affect CVD and high mortal-ity rates.
References
1. National Kidney Foundation: K/DOQI clinical prac-tice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Kidney Disease Out-come Quality Initiative. Am J Kidney Dis 39:S1-S246, 2002
2. Collins AJ, Li S, Gilbertson DT, et al: Chronic kidney disease and cardiovascular disease in the Medicare population. Kidney Int 64:S24-S31, 2003 (suppl 87) 3. Ross R: Atherosclerosis—An inflammatory disease.
N Engl J Med 340:115-126, 1999
4. Ridker PM, Cushman M, Stampfer MJ, et al: Inflam-mation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med 336:973-979, 1997
5. Ishani A, Collins AJ, Herzog CA, et al: Septicemia, access and cardiovascular disease in dialysis patients: The USRDS Wave 2 study. Kidney Int 68:311-318, 2005
6. Kessler M, Hoen B, Mayeux D, et al: Bacteremia in patients on chronic hemodialysis. A multicenter pro-spective survey. Nephron 64:95-100, 1993
7. Johnson DW, Fleming SJ: The use of vaccines in renal failure. Clin Pharmacokinet 22:434-446, 1992 8. Smith PS: Management of end-stage renal disease in
children. Ann Pharmacother 32:929-939, 1998 9. Appel GB, Radhakrishnan J, D’Agati V. Secondary
glomerular disease; and Asplin JR, Favus MJ, Coe FL. Nephrolithias. In: Brenner BM, ed. Brenner & Rector’s The Kidney Volume II. 6th ed. Philadelphia: W.B.
Saunders; 2000:1350-1448, 1774-1819.
10. Sarnak MJ, Jaber BL: Mortality caused by sepsis in patients with end-stage renal disease compared with the general population. Kidney Int 58:1754-1764, 2000 11. D’Agata EM, Mount DB, Thayer V, et al: Hospital-acquired infections among chronic hemodialysis pa-tients. Am J Kidney Dis 35:1083-1088, 2000
12. U.S. Renal Data System: USRDS 1997 Annual Data Report. Bethesda, Maryland, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 1997
13. Powe NR, Jaar B, Furth SL, et al: Septicemia in dial-ysis patients: Incidence, risk factors, and prognosis. Kidney Int 55:1081-1090, 1999
14. Nassar GM, Ayus JC: Infectious complications of the hemodialysis access. Kidney Int 60:1-13, 2001 15. Doulton T, Sabharwal N, Cairns HS, et al: Infective
endocarditis in dialysis patients: New challenges and old. Kidney Int 64:720-727, 2003
16. Strom BL, Abrutyn E, Berlin JA, et al: Risk factors for
infective endocarditis: Oral hygiene and nondental exposures. Circulation 102:2842-2848, 2000
17. U.S. Renal Data System: USRDS 2005 Annual Data Report. Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kid-ney Diseases, 2005
18. von Graevenitz A, Amsterdam D: Microbiological aspects of peritonitis associated with continuous am-bulatory peritoneal dialysis. Clin Microbiol Rev 5:36-48, 1992
19. U.S. Renal Data System: USRDS 2004 Annual Data Report. Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kid-ney Diseases, 2004
20. U.S. Renal Data System: USRDS 2003 Annual Data Report. Bethesda MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kid-ney Diseases, 2003
21. Gilbertson DT, Unruh M, McBean AM, et al: Influenza vaccine delivery and effectiveness in end-stage renal disease. Kidney Int 63:738-743, 2003
22. Eickhoff TC, Sherman IL, Serfling RE: Observations on excess mortality associated with epidemic influ-enza. JAMA 176:776-782, 1961
23. Zhou W, Pool V, Iskander JK, et al: Surveillance for safety after immunization: Vaccine Adverse Event Reporting System (VAERS)—United States, 1991-2001. MMWR Surveill Summ 52:1-24, 2003
24. Szmuness W, Prince AM, Grady GF, et al: Hepatitis B infection: A point prevalence study in 15 US hemodi-alysis centers. JAMA 227:901-906, 1974
25. Szmuness W, Neurath AR, Stevens CE, et al: Preva-lence of Hepatitis B “e” antigen and its antibody in various HBsAg carrier populations. Am J Epidemiol 113:113-121, 1981
26. Tokars JI, Miller ER, Alter MJ, et al: National Surveil-lance of Dialysis-Associated Diseases in the United States, 1997. Atlanta, GA, Centers for Disease Control, 1998
27. Vercaigne LM, Zelenitsky SA, Findlay I, et al: An in vitro evaluation of the antibiotic/heparin lock to ster-ilize central venous haemodialysis catheters. J Anti-microb Chemother 49:693-696, 2002
28. Shinefield H, Black S, Fattom A, et al: Use of a Staphylococcus aureus conjugate vaccine in patients receiving hemodialysis. N Engl J Med 346:491-496, 2002
29. Polderman KH, Girbes AR: Central venous catheter use—Infectious complications. Intensive Care Med 28:18-28, 2002
30. Khan IH, Catto GR: Long-term complications of dial-ysis: Infection. Kidney Int 41:S143-S148, 1993 (suppl) 31. Advisory Committee on Immunization Practices: Pre-vention and control of influenza: Recommendations of the Advisory Committee on Immunization Prac-tices (ACIP). MMWR Recomm Rep 48:1-28, 1999 32. Johnson DW, MacGinley R, Kay TD, et al: A
random-ized controlled trial of topical exit site mupirocin application in patients with tunnelled, cuffed haemo-dialysis catheters. Nephrol Dial Transplant 17:1802-1807, 2002
![Fig 1. Congestive heart failure after septicemia. (From American Society of Nephrology [ASN] 2003 United States Renal Data System [USRDS] One Hour Talk, available at www.usrds.org.)](https://thumb-us.123doks.com/thumbv2/123dok_us/9619388.2840845/2.891.130.765.115.450/congestive-failure-septicemia-american-society-nephrology-united-available.webp)