Effect of the gene transformer of Anastrepha on the somatic sexual development of Drosophila
Full text
Figure
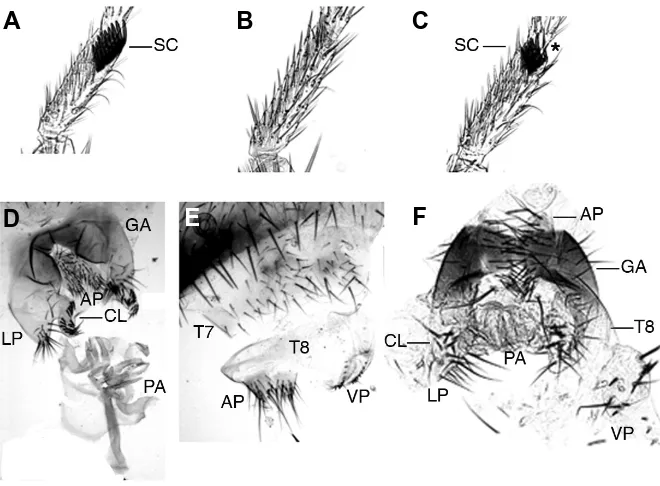
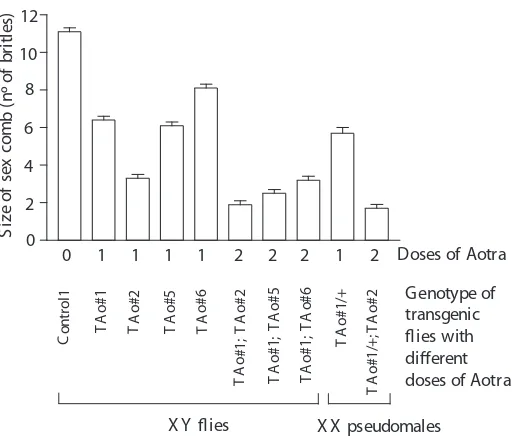
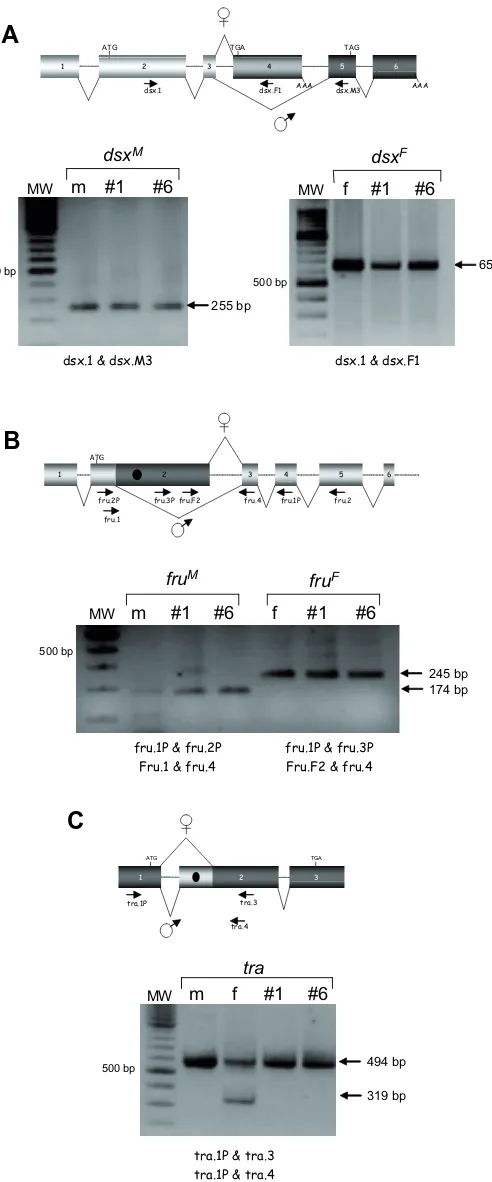
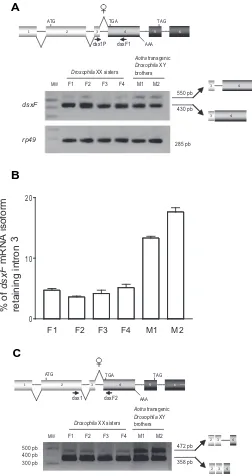
Related documents
Biofilm production by selected strains of bacteria (a) and yeasts (b) in the presence of Textus bioactiv, Textus multi dressings, and culture broth without dressing.. The figures
Screening of cytotoxic activities using WiDr and Vero cell lines of ethyl acetate extracts of fungi-derived from the marine sponge
Subdivision of the tumors suggested a difference in the distribution pattern of expression of the two TACC pro- teins, with TACC3 loss being more common in serous papillary
Antihypertensive therapy in hypertensive patients imme- diately post stroke may be effective and cost-effective compared with placebo from the acute hospital perspec- tive at
Abstract: This study examines the unique experience of participants who during their reintegration back into the community, following a conviction for sexual offending, re-
Keywords— Turn around Time (TAT), Communication Distance, Requester, Resolver, Information Distance, Communication Bounce, Information Bounce, Information Technology
Implementing binary matrices with as few XOR operations as possible is also known as the problem of finding the shortest linear straight-line program [BMP13; FS10] over.. the