University of Windsor University of Windsor
Scholarship at UWindsor
Scholarship at UWindsor
Electronic Theses and Dissertations Theses, Dissertations, and Major Papers
1-1-1973
A study of the EPR spectrum of the Ca(OH)
A study of the EPR spectrum of the Ca(OH)
22
:Gd 3+ system as a
:Gd 3+ system as a
function of temperature.
function of temperature.
Gérald E. Roberge
University of Windsor
Follow this and additional works at: https://scholar.uwindsor.ca/etd
Recommended Citation Recommended Citation
Roberge, Gérald E., "A study of the EPR spectrum of the Ca(OH)2:Gd 3+ system as a function of temperature." (1973). Electronic Theses and Dissertations. 6887.
https://scholar.uwindsor.ca/etd/6887
This online database contains the full-text of PhD dissertations and Masters’ theses of University of Windsor students from 1954 forward. These documents are made available for personal study and research purposes only, in accordance with the Canadian Copyright Act and the Creative Commons license—CC BY-NC-ND (Attribution, Non-Commercial, No Derivative Works). Under this license, works must always be attributed to the copyright holder (original author), cannot be used for any commercial purposes, and may not be altered. Any other use would require the permission of the copyright holder. Students may inquire about withdrawing their dissertation and/or thesis from this database. For additional inquiries, please contact the repository administrator via email
N A TIO N A L LIB R A R Y
jWk
BIB U O TH ^C fU E N A TIO N A LE
O t t a w a A 8 8 B O t t a w a
Oanam
' - ■
NAME OF
TITLE OF THESIS.. .A.
W*.
.aP.e.c.t™?f.
PK8. p & ( P H k ;Gd3+system. aa^ a fu n c tio n of_ tempejrp tujre._
UNIVERSITY.... Ppiypr??-?Y. pf Windsor.Ifcpdppr,.
OvtZ-tfgJ...
DEGREE FOR WHICH THESIS WAS PRESENTED... ...
L
YEAR THIS DEGREE GRANTED ; ... . 3-973... .*
Permission'is hereby granted to THE NATIONAL LIBRARY
f •
OF CANADA to microfilm this thesis and to lend or sell copies
of the film.
The author reserves other publication Tights, and
neither the thesis nor extensive extracts from it may be
printed or otherwise reproduced without the author's
written permission.
(Signed).
PERMANENT ADDRESS:
f . r . u 7j c - y - , s o % ,
'cn
A STUDY OF THE EPR SPECTRUM OF THE Ca(OH)2 :Gd3+
SYSTEM AS A FUNCTION OF TEMPERATURE
V
by
Gerald E. Roberge
A Thesis
Submitted to the Faculty of Graduate Studies through the Department of Physics in Partial
- Fulfillment of the Requirements for the Degree of Master of Science at
\ the University erf Windsor
Windsor, Ontario
’ 1973
(ey Gerald E* Roberge 1973
447000
^ 'f
APPROVED BY:
7 T
T
ABSTRACT
. ■■**. • *
The temperature dependence of the C a ^ H ) ^ G d ^ +
» # ^
system was studied in the 77°K to 375°K range by means of
the electron paramagnetic resonance method at K band ■.
frequency. '
The trigonal-crystal field parameters were fitted
to the \spin-tlairfi'4ta>ni^o
£ B T / '
n 0 . Anm nm • n=0,2,4,6
nr=
0
, +3
, +6
(~t |m 1 s n
^ /
using a multi-dimcnsional least squares method. The.
■ ' . ' v
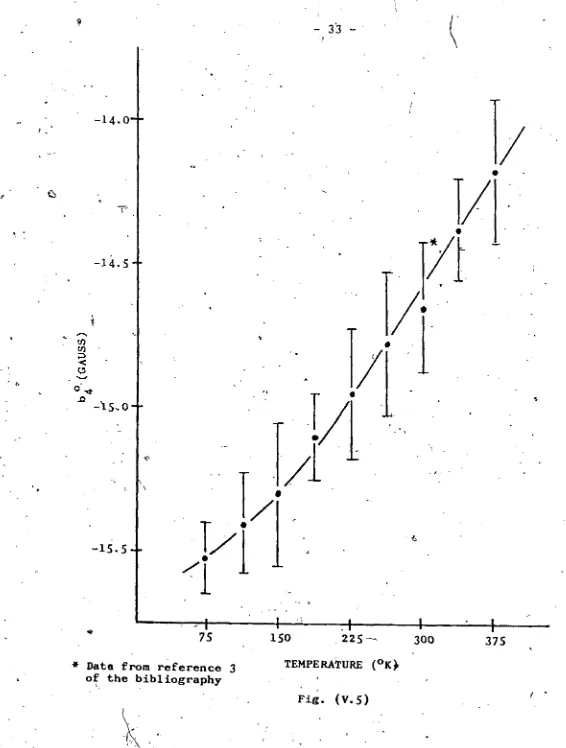
p^araeters which showed temperature dependence were the \ixi 1
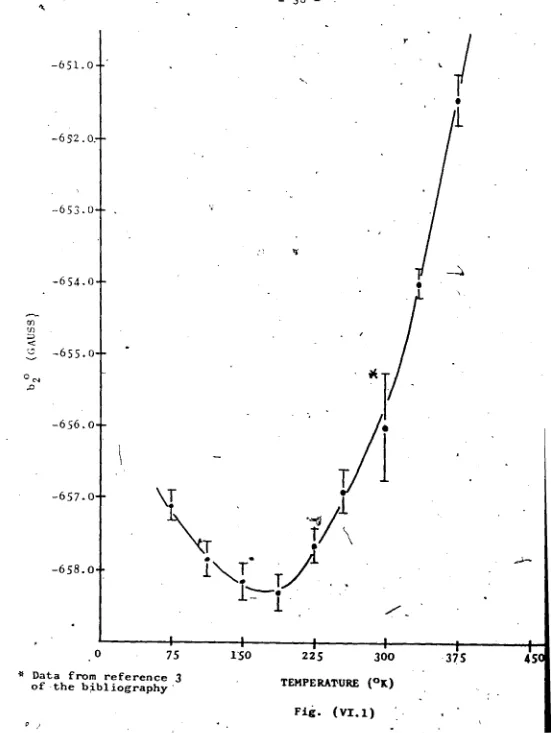
parameters 1*20 anc^ ^4 0’ while the b sbowed a very rapid
change at around room Cemperaturc.
£
The values of thectcmporaturc independent
parameters turned out to be:
Rx — Sy — g z = 1.9920 + 0 .0006
. b 6° - (-1.2 + .2) Gauss ^
bfi3 - = (27 + 4) Gauss
b 66 - ( - 1 9 + 2) Gauss
Nq hyperfine stiiicturc was observed,, although the nuclear s
of Gd^~* and G d ^ Y each occurring with — 1 5 abundance, ha
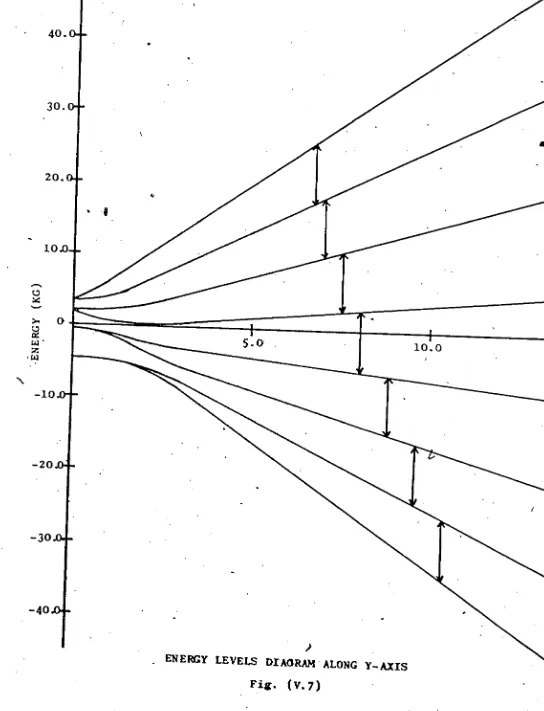
values of ~. Energy level diagrams were drawn, ^showing
perfect linearity for a magnetic field >2 Kgauss and up to
1 5 Kguussr. . ^
iii
\
ACKNOWLEDGEMENTS
.
^ ' 1,
I
The author wishes to thank Dr. F. Holuj for
first proposing this study and for his guidance daring this
work. Thanks are also due to Dr. T. Kwan for his help'
concerning the experimental work' and to my wife, Paulette,
for her assistance in^ recording of ‘data and preparation
p of diagrams. ' * _
IV
TABLE OF CONTENTS
ABSTRACT «
)
ACKNOWLEDGEMENTS
LIST OF TABLES
LIST OF TIGURES
CHAPTERS
« •
I.
II.
III.
IV.
V.
INTRODUCTION
CRYSTAL STRUCTURE OF Ca'(OH).
THEORY
A) Electron Paramagnetic Resonance
1. Free Paramagnetic Ion
• 5
-%. Crystal Field
3. Trlvalent Gadolinium Ion
B) Hamiltonian
1. General Form {
^,
2. Spin-Hamiltonian Operator
3- Matrix Elements
INSTRUMENTATION
A) Microwave Circuit
B) Electronic Circuit /
C) Magnetic Field
i
EXPERIMENTAL PROCEDURES AND RESULTS
A) Angular Variation
B) Temperature Variation
C) Results
page
iii
iv
vii-viii
I-2
4
« 4
4
4
4^
5
5
7
8
14
14
16
17
19
19
30
30
page
VI. DISCUSSION AND CONCLUSIONS p 37
A) Crystal Orientation . - : 37
B) Temperature Dependence 37
C) Charge Compensator 40
BIBLIOGRAPHY " ’ 42
APPENDICES ' 42
A. - Magnetic Field Calibration 44
B. Best Fit Data of Spin-Hamiltonian 45 Parameters
VITA AUCTORIS 4 6.
r
\
V
LIST OF TABLES
\
page
II.1 Structural Data for Ca(0H)2 ' . 21
111.1 Table of Tensor Operator Equivalents 11
S' ^ •
111.2 Relationship Between b m and B 11
n nm
111.3 Matrix Elements of Spin-Hamiltonian 12
111.4 Values of Matrix Elements of the t
Tensor Operator Equivalents 13
V.l(a-h) Spectrum Measurements ' ' 22
.. \
-V
.
■
* x
1 '
*
*
vii
Reproduced with permission
7 * *
*
OF FIGURES
page
II.1 C a f O H ^ crystallography 3
IV.1 Block Diagram of Apparatus 15
V.l Angular Variation in XY Plane 20
V. 2 Angular Variation in ZX Plane 21
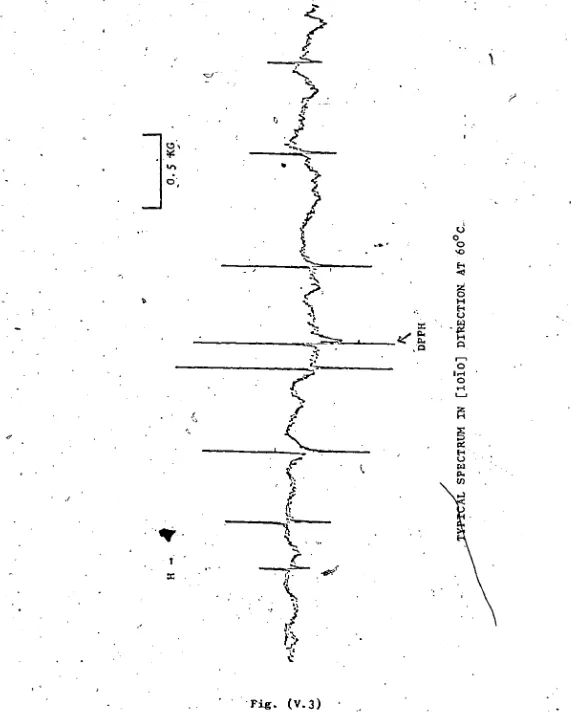
V. 3 iTypical Spectrum in [1010] Direction
at 60°C - < 31
V,4 Temperature Dependence o f , b^5 Parameter 3.2
V. 5 . Temperature Dependence of Parameter 33
V.6 Temperature Dependence of b Parameter3
4 34
V. 7 Enprgy Levels Diagram Along Y-Axis 35
V. 8 Energy Levels Diagram Along' Z-Axis 36
VI. 1 ModifiefKPlot of Temperature Dependence of b,°/Parameter
Z/ \
38
viii
CHAPTER I
INTRODUCTION
The purpose o f this experiment was to measure the
' *) 3+
temperature dependence of the Ca(0H)2:Gd system from liquid
nitrogen (77°K). to above room temperature (375°K) using the
methods of Electron Paramagnetic Resonance Spectroscopy. The
choice of CatOH)^ was motivated by the fact that it proved to
be a suitable host in which to study temperature effects on
* 1 ?*
the EP& spectra.of (Cu^+ ) and (Mn“'+ ) .
A Jahn-Teller effect having a relatively higl
transition' temperature from the static to the dynamic state
■ 2+
was encountered in the Cu spectrum. Moreover, the dynamic
Jahn-Teller spectruqi turned out to be anisotropic. In the
4
case of the-Mn EPR spedtrum, the spin-Hami 1 tonian
o ^
parameter reversed its sign at 4 5 0-K, while the value of the
ipotropic hyperfine structure tensor component decreased
sizeably with the increase of temfSerature.
* The room temperature investigation of the G d . ' s p e d
had been carried out earlier in this laboratory' . A computer
program, which diagonalizes the Hamiltonian, f?ts the crystal
C'. t
field parameters through a multidimensional Newtoq-Raphson
least square minimization method. The form of live program us
in this investigation is a revised version of that routine.'
- 1
-\
\
s
CHAPTER II
f
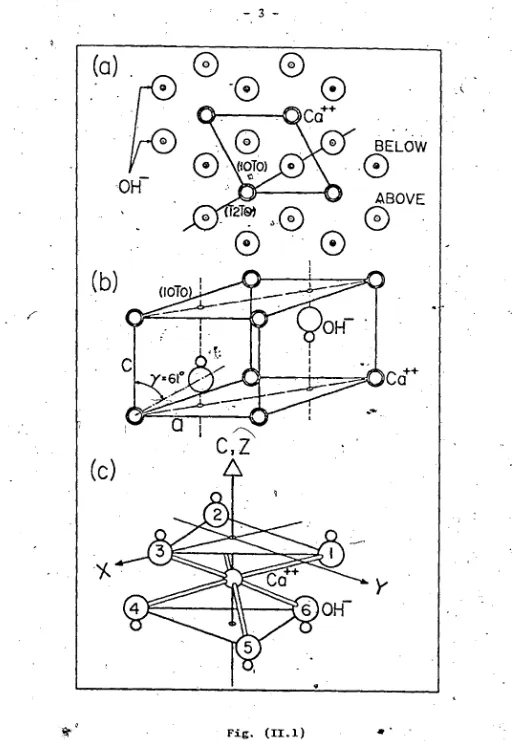
-CRYSTAL STRUCTURE OF,Ca(OH>2
' % _
Calcium hydroxide crystal belongs t<? the hexagonal
— 2 1
system (Cdl9 type) with space group P3 — (D,,). There is
■t- m Ja
t
one formula unit per unit cell. The calcium ion occupies
the invariant position (0,0,0) with point symmetry 3m while
the OH complex occupies positions with symmetry 3-rji.
Fig- (II.la) is a projection of the lattice in the
,-.(0001) plane, showing the^ three-fold symmetry axis passing
2+
through the Cn“ sites, while Fig. (II.lb) shows the hexagonal
unit cell. The choice of coordinate system is defined in
Fig. (II.lc). The X, Y and Z axes are parallel to the [ lOlO],
)
[1210] and [0001] di rcctions respectively; the hyuhoxyl ions
thus occupy the two adjacent layers normal to the hexagonal
2+
axis and are sandwiched on either side of the Ca layer.
The folloVing table gives structural data of the
Ca(0il) 9 crystal at 20°C. ^
TAM.E ( IT. 1 ) 4
Unit cell parameter a = 3-591'8 A’
> Unit cell parameter c = 4-9063 A
Structure parameter = 0.2341 A
Structure parameter = O.4248 A
- 2
\
3
-S'
©
B E L O W
ABOVE
'ST
Fig. (II.1)
•'
A '
CHAPTER III
. * THEORY
>
Et^CTRON/PARAMAGNETIC RESONANCE
Free paramagnetic ion
- e. 0
If a free ion-with a resultant electronic angular
momentum J is.placed in a magnetic f i e l d H , the energy levels
. \ ;
of this magnetic dipole are g^ven by
✓ • ,
E .= mjg/^H ' (III.1)
To produce magnetic dipole transitions, such that Anij = + 1»
an alternating R.F. field of frequency v .satisfying the
' • v; , . ‘ " '*
resonance Condition •
hv = g/^H (III. 2)
^ r
is applied at right angles to H. - ,
r * " * *
2 * Crystal Field '
When the paramagnetic ion is placed iri a solid, it
will form bonds with the diamagnetic ligands. These ligands
are charged ions which not only set up internal electric fields,
considered to a good approximation to be static, but also form
chemical bonds. The overall effect on the energy levels is a
splitting of the ground state into a number of components. ‘ As
a result, the orbital and spin degeneracy of the free iori is
partly removed.
3. * Tr^. valent Gadolinium -Ion
1
•
'
7
Gadolinium’ ion has the electronic structure (Xe)4f
so that the ground state o f ^ h e free trivalc^llt iorfvis the
4
-r
-l '
8 7
orbital singlet state ^7 /2 *'*le ^ configuration. When the ion is introduced in C a C O H ^ crystal as an impurity, the
8
ground state becomes since the spin degeneracy is
unaffected by the crystal field.
There is no simple explanation of the
zero-field-splitting, although qualitatively, it is no doubt caused by
high-order processes. Wybourne^ enumerated several, and in
the specific case of ethylsulfate host, two contributed
significantly to the ^ 2 ° P arameber: the fourth-order, involvin
the crystal field linearly, and a second-order, also linear in 7
crystal field but involving the excited 4 f P7/9 state. These,
two mechanisms give" a contribution having an opposite sign to
the experimental b^0 parameter.
B) HAMILTONIAN
1. General form ^
The general form of the Hamiltonian describing the
various interaction^' of each unpaired electron of the free. t
•paramagnetic ion with its surroundings tnay be written
as:-w h e r e :
JC= T + vc + VSL + vss + VN + v2 . . (III.3),
. t»K2 . ■ *' •
• T =/* E -r— is the kinetic energy; sum is over all
7 r f « *
K *
s
K electrons.
^ Z e 2 ' e2
s= - S + --- is the interaction of each C .. r., . s,. v. .
h h xj
i '
electron with the Coulomb field of the-nucleus,
f *
modified _by the repulsive field' of the other
electrons, and summed over all electrons. ^
-VSL = ■ Z \ l . 's^ is the spin orbit coupling which
i •
<• , reduces to X L ' S for Russell-Saunders scheme . V „ _ *=•£ Si ’?.i 3(ri.i‘S i ){ri.i‘SK )
SS ,• — o— ~--- -" c -7— --- : s p m - s p i n
,J -rA r A
ij 13
coupling. , .
Vjj, is the hyperfine term while the electrostatic interaction '
with the quadrupole moment Q of the nucleus is described by
V
follows:
The order of magnitude of these interactions is as
t.
Vr ~ 105 cm 1
c ■ CJ
3 - 1
10 cm for rare earths '
v ss~
1
cm
V.. ~ 10" 1 - 10“ 3 cm" 1 ..
N
V ~ 10"3 cm" 1
Now, when the ion is placed in the crystalline field,
the interaction between the eldctrons of the paramagnetic ion '
and the ligands adds.an -additional term V to the Hamiltonian:
A
V = £ e $(r ) : depends on position of each electron.
X K K •
* — * 1
Finally, if the ion is acted upon by a magnetic field H,
additional interactions arc: *
VH = £ K + electronic interaction
K
Vh = : nUc^ear interaction
7
-2. Spin Hamiltonian Operator
Since the general Hamiltonian is complicated and
avgcward to work, with* it has become general practice to
define a "spin Hamiltonian" in the following way: the EPR
spectrum is looked upon as belonging to a fictitious spin S
and having a degeneracy of 2S + 1. Now, since the crystalline
field is, in general anisotropic, its contributions to the
splitting of the ground state varies with the orientation of
the crystal with respect to the applied external magnetic
field.
'The Hamiltonian may thus *be written:
3C = /ifiS-g-H + £ Bnm Tm + S-3C-I + I* Q-1 - AjgjjI'.H-
(HI. 4)
run0 *
where g,; A and Q are second rank tensors
"^nm are *'ensor operator equivalents. They are functions
*
of the effective spin operator S.
Bnm are empi rical parameters. These quantities are fitted
to experimental data.
The first term in Eq. (III.4) is the electronic Zeeman tens,
while the second describes zero-field splitting by the crystal
field. The third term is the hyperfine term, the fourth
being the quadrupole interaction and finally, the last t^rm
describes the nuclear Zeeman splitting.
'' ' ^ f .. * *
" > In the case of C a ^ H ^ t G d , no hyperfine splitting or nuclear Zeeman transitions are observed at the microwave
P-frequency and in the range of magnetic field used.
The electrostatic potential in which the Gd^+ ion
with
finds itself arises from the ligand ions. These are
regarded as point charges such that the potential is a
solution of the Laplace’s equation AV = 0, and is given by
the spherical' harmonics : ».
V = E E E A® r11 = E ; V® (III. 5)
. n ^ n
n X n,m ■
Ct
where: . *
K relates to the electron in the unfilled shell
Y ® (0k„ O is the angular part of the spherical harmonics, n k k
Symmetry considerations define the values that m and n can
t a k e :
— n can only take on values up to 6 since f electrons
(3) """
transform like D of the rotation group and when the
matrix elements V® are computed, the direct product
(3) (3)
D x D involves no representation higher than 6.
- also n can take on even values only since the parity of
,t matrix elements of V m must be even. n
— n = 0 gives \'^ contribution which is an additive constant.that we chose to ignore.
- m can only takb on values m — 0, + 3} + 6 since the
crystal f^ield at the paramagnetic ion possesses axial,
three-fold and six-fold symmetries.
3 - Matrix Elements
We express the crystal field contribution in terms
of angular momentum operator equivalents as follows. In
genera], any tensor may be decomposed into components which
transform independently under rotation of the coordinate
systefti. An*' "irreducible tensor operator" T , with ,nm
components (m = -n, -n + 1, ... , n -- 1, n) additionally
satisfies the commutation relations with the angular
momentum operator J:
[Ji> Tnm] = ia+in 1/2Tnny.1 •> . (III.6)
p > Tnn.J = mTnm '• ’"here J ± r J x ± *■
3y,
Eq. (III.6), in its inverse form, thus generates all of the v
tensor operators. The ones of importance in this case are
listed in Table (III.1). We thus have, substituting in
Eq. (III.4): / ■
" “ % hn ho + V
t
! T1-1 \7J Tn>
Ulhn
m=0, + 3,6
mlfin
/
wh e r e ;
hi - g xHx ± igy 11/
h (| = g Z HZ
The Zeeman term is represented in a frame of reference in
which the g tensor is diagonal; the hyperfine, as well as the
quadrupole and nuclear Zeeman terms have not been included.
The matrix' element's of the crystal field tensor
operators are given by <m,lT |m,»>, where T transform as
J nm J ' nm
the spherical harmonics Y m , h m1 beihg the Z component of
n J
the angular momentum. The non zero terms are the ones for
which**
m = m J - m j.
■ . .. ■ ' ■ . i
- 1 0
-The matrix elements of the spin-Hamiltonian operator are
given in Table (ill.3)* The numerical values of the matrix
elements of the tensor operators are given in Table (XXI.4),
/*
while Table (III.2) gives the relationship between the fitted
parameters B and the more conventional b m parameters.•
nm v n .
"N
11
-•TABLE (III.1)
Table of Tensor Operator Equivalents
T10 - Jz
Tl+1
J±
T20 = / J ^3JZ2 - J (J + ^
T40 =
J I^b
t35Jz4 - ,5{6J(J+l)-5}Jz2+ 3 fJ2(J+l)2-2J(J+l)}]T4+3 “
^2
II
£J+ Jz +^60 ~ ~4 / ^3l - 105 C3J( J + 1) - 71Jz^
+ 2l[5J^(J + 1)Z - 25J(J + 1) + 1 4}j z
-5Cj3 (J + l)3 - 8j 2 (j + l)2 + 1 2j (j + 1)}]
t6±3 = ? I / I T tJ+ (llJz2 ' 0 <2 + 1) + 59) Jz 3
~ ■
\
+ {11JZ3 - Q3J(J + 1) +59) J2 )J+]
T = 1 T6
6+6 = S J+
TABLE (III. 2). . .
Relationship Between b m and B
n nm
h °=(l)1/ 2B ■ K h O 315
2 V 20 4.'’ 7 40 6 - ( ~31~ l/ 2 60
\ 3= - F6 0 B43 • b63= -630(|r )1/2B63 b66= 3 1 S 'B66
M a t r i x E l e m e n t s of S p i n -H a m i l t o n i a n
- YZ
-K
t\
L sj OQ* £ *m **
4 . -J t*5 $>Q
■ ^ i s R
■sfi §
-* *
i t ? « o
r > *
£
sr fn
^ ° 1 '
+ I
i - c
t n
3 0
L. ^Hi< Xl" ^
» i i
4-■ -C
t w
« i ' o A
l * “ 5 ®
3 ^
• * *
-t t 4,
\ k
£ *>
* * cS
1 1
J - C
, § 5 2 IjjjH tO W
.* 1 ^,w>
1 4, »
i g ,
-«** *
V tj}*
l y x
I ® ^ >4 <*
+ -V
n» ^ vfi
f t ?
-a ^
« i
i l l
l * j a » W
- c *- w - w
+
o
«
m tf\
* * rvf
00
1 4 *
* - r
•
\
- 9 o
0 £
f c t i *
> ■ *
__ ^ n
* - v i
x +
/
*
ta t*
f c u [ * ^
*.«
4.
2 *
^ N»
50 <0
f $
•
-o
• o
£
2
k \ *
..
V'
V
. . .:
r t ? ^
i & s :
» i
f. a >* * o (S*
K
"SsA
OIcQ VReproduced with permission of the copyright owner. Further reproduction prohibited without permission.
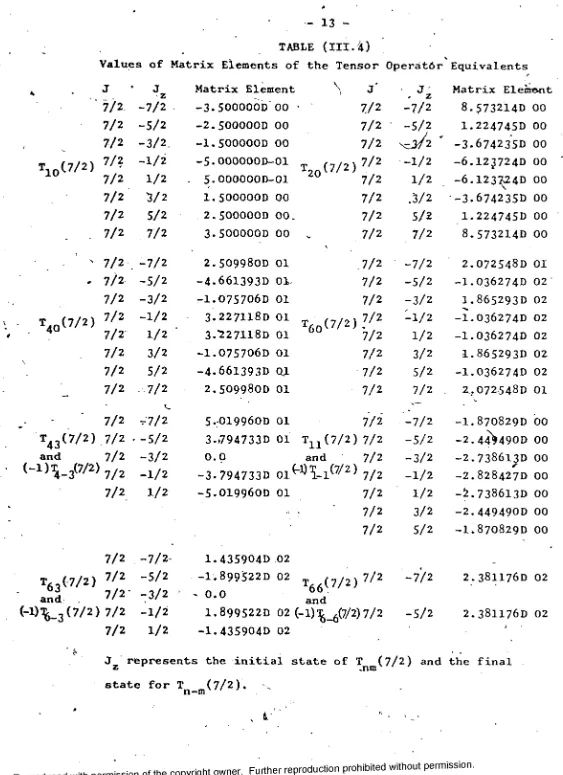
13
-T1 0(7/2)
t40(7/2)
Values
J
‘ 7/2
7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2 7/2
TABLE (III.4)
of Matrix Elements of the Tensor 0perat6r Equivalents
' 7/2
t4 3( 7 / 2 ) .7/2
and 7 /2
(-1) W 7/2) 7/2
7/2 J z -7/2 -5/2 -3/2. -1/2 1/2 3/2 5/2 7/2 -7/2 -5/2 -3/2 -1/2 1/2 3/2 5/2 7/2 r7/2 -5/2 -3/2 -1/2 1/2
7/2 -7/2
7/2 -5/2
and . 7/2- -3 /2 (-l)^_3(7/2) 7/2 -1 /2
7 /2 1 /2
T6 3(7/2)
Matrix Element
-3.500000D 00
-2.500000D 00
-1.500000D 00
-5.OOOOOOD-Ol
5•OOOOOOD-Ol
1. 500000D 00 2.500000D 00.
3.500000D 00
2.5 0 9 9 8 0D 01
-4.661393D 01
-1.075706D 01
3.227118D 01
3 . 7 2 7 H 8 D 01
-1.075706D 01
-4.661393D Q1
2.5 0 9 9 8 0D 01
5.0 1 9 9 6 0D 01
3-»794733D 01 0 . 0
-3-794733D 01^
-5.019960D 01
\
T20(7/2)
60
" 1 1 v and
1.435904D 02
-1.899522D 02
< 0.0
66
and (7/2)
J J
* z Matrix Element
7 /2 -7/2 8.573214D 00
7/2 -5/2 1.224745D 00
7 /2 -3.674235D 00
7/2 -1 /2 -6 .1 2 3 72 4 0 00
7 /2 1 /2 -6.123^24D 00 7 /2 .3 /2 -3.674235D 00
7 /2 5 /2 1.2247450 00
7 /2 7 /2 8.573214D 00
7 /2 -7/2 2.072548D 01
7/2 -5/2 -1.0362740 02
7 /2 -3/2 1.865293D 02 7/2 - 1 / 2 -1.036274D 02 7/2 1 /2 -1.036274D 02
7/2 3/2 1.865293D 02
7/2 5/2 -1.036274D 02
7/2 7/2 2..072548D 01
7/2 -7/2 -1.870829D 00
7/2 -5/2 -2.44^490D 00 7/2 -3/2 -2.7386lpD 00
7/2 -1 /2 - z. 8284270 00
7/2 1 /2 -2.738613D 00
7 /2 3/2 -2.4494900 00
7/2 5/2 -1.870829D 00
7/2 -7/2 2.381176D 02
1.899522D 02 (-1)^(7/2) 7/2 -S/2
-1.435904D 02
2.381176D 02
J represents the initial state of T (7/2) and the final
z .nm ‘ '
state for T m (7/2).
CHAPTER IV
f '
INSTRUMENTATION
The ESR spectrometer used in this experiment was ,
of the balanced bridge .type with the microwave frequency ^
\
stabilized against the sample cavity. A block diagram
describing\the system is shown in Fig. (IV.1). It operated
at a n ominal>frequency of 24GHz. A 1 2 n Varian magnet, o f x
which the field was stabilized against a Hall-effect probe,
provided the magnetic field. The resohant fields were
measured using a proton magnetometer oscillator.
' The following is a brief description of the
principal components of the apparatus used in the experiment.
A) Microwave Circuit:
- Klystron is the microwave source (Varian model VA98M), of
reflex type with a nominal power output of 30mw. The
power supply w a d a Hewlett-Packard (#7l6B). The klystron
was immersed in a) water-cooled oil bath.
1
' '-Isolator is a microwave ferrite device which makes use of
’ \ ^ ' '
the Faraday effect)to permit transmission of microwaves
in one direction^only.
-Wavemeter cavity is a calibrated, tunable cylindrical' cavity
used as a frequency meter.
- Power attenuator is used to monitor power iricidepb on loaded
cavity in order to avoid saturation.
*
%
* *r
-Circulator permits transmission of power from one terminal to
the next in sequence. Here, the microwave is transmitted
B
l
o
c
k
D
i
a
g
r
a
m
of
A
p
p
a
r
a
t
u
s
15
-^4. M
w S'
u
u-p* O 41 4J C u
§ 1
m
c JC « «cw ui o i f
H O
Li
hfl •H
e c & 1 X »
*4 « « U 9>
d o
u
>> U U h« • o. >,J a
u
•3 41 U
*
s
• < 1> > « 41 c
H O P
X (Xe V) 5 2
1 8 >8 •
m
Reproduced with permission of the copyright owner. Further reproduction prohibited without permission.
LO
O
kH
i
F
i
e
l
d
M
o
d
u
l
a
t
i
o
— 16
-^ . *
to the resonant cavity arm and then to the crystal
detector arm.
-Slide-screw tuner serves to match the impedance of the
klystron arm with the cavity arm. Usually, the cavity ♦
is slightly mismatched in order to bias the detector
crystal.
N
- Resonant cavity was made of a glass tubing whose inner
surface was sputtered with gold. The cavity is coupled
to the microwave arm by a thin copper iris ( ~4mm
diameter)* since the waveguide was' rectangular.
A heater was made of chromel wire and embedded in Sauer
Eisen on the o^ts^de of the bottom plate of the cavity.
The crystal, glued to a hollow quartz pin, could be ^
rotated about an horizontal axis by means of a
gonio-meter. Any orientation of the crystal, relative to
the magnetic field, could thus be attained since the
magnet could be rotated about a vertical axis. Copper as
constantan thermocouple wires were inserted in the pin
so that the junction made contact with the crystal. The
cavity operated in the mode.
v.
\ ‘ N
B) Electronic Circuit: ^
-Klystron frequency control is used to stabilize the klystron”^ )
frequency. The principle of operation is the following:
by frequency modulating the reflector, the signal
detected by the. AFC (automatic frequency control) crystal
detector contains a component of the modulating signal;
whose phase and amplitude depends on the difference
between the-klystron and cavity frequencies. This AFC
signal is then fed to a phase sentitive detector (P.S.D.)
The resulting D.C. error voltage is applied to the
reflector of the klystron-.
The klystron stabilizer used was a Teltronic Model KSLP,
the modulating frequency being 70 Khz.
-LQCk-in amplifier compares the phase and frequency of a
\ ■
resonant signal, from the signal crystal detector, with
the original 100 Khz modulation signal. This modulation
signal is generated by an oscillator incorporated in the
lock-in amplifier and used to modulate the magnetic field
by means of two Helmholtz coils placed along side the
cavity, and parallel to the pole faces of the magnet.. The
result is a derivative signal proportional to the
resonant signal, which is displayed on the oscilloscope or
chart recorder as a function of magnetic field. The
Variac is used to regulate 60 Hz’ modulation in oscilloscope
display. The lock-in amplifier used was a Princeton
" * • "
Applied Res^Arch, Model JB-6.
C) Magnetic Field
The magnetic field was produced by a water-cooled
12 inch Yar ian electromagnet with a 3 .5 inch gap and rotating
^ ^ A ,
base. The field was stabilized to 1 part in 10 by'means of
o Fieldial model V-FR2503 (Varian) control unit. The 100 KKf"
signal, originating from the lock-in amplifier, is first
c
power»amplified before being applied to the two Helmholtz
coils-connected in series.
The magnetic field strength was measured by means
of a protdn magnetic resonance the probe of which
consists’ of a marginal oscillator. ' The source of* proton
was a small piece of rubber and the probe was placed at the
center and near the pole-face.
. The correction for difference in field strength
at the polle face and in the center-of the gap, where this
crystal is located, was obtairted by making corresponding 0 field measurements at both positions; these are tabulated
in Appendix•A. Since the differences involved were-less t
than the reproductibility deviations of the E.P.R. lines, the v
J • - $ ‘
average difference was calculated and added as the correction
i - ■ * •
to the measured fields.
CHAPTER V
EXPERIMENTAL PROCEDURE AND RESULTS
A) Angular Variation
The orientation of the crystal was arrived at by
determining the'.Z-axis (C-axis) and the Y-axisj these were
4
-defined by observing the turning point of the J— 5, | > line
in'each case. The coordinates of each axis was plotted on a
Wulff net, thus determining the thre^p'rincipal planes. The
angular variation of the spectrum in the basal (X-Y)
plane-* I .
was accurately measured, using the P.M.R. magnetometer at
intervals of 10 degrees. The results, for the j ~ > — ]-r >
apd | — ] — rg > transitions are given in Fig. (V.l). Next, the angular variation in the X-Z.plane, at
liquid nitrogen temperature, was determined from the spectra
traced-out on a chart ^recorder at intervals of 10 degrees.
This result is given in Fig. (V.2). A comparison with the
room temperature results^ reveals no qualitative difference.
The spectrum was measured accurately at different
orientations and temperatures, always using the same set of
ten orientations at each temperature. These measurements
are given in Tables (V.la) - (V.lh), along with the oricntatio
angles. THETA and PHI, the D.PPH ^standard (free radical a,
a.'-dipheryl (3-picrylhydrazyl) and the temperature (°C). Note
o t '
that the fields are given in Mhz.
V .
a) 19
-V
A
«H|<M iH
t
A
' m
A
i—4
JO
O
\Ti
t
LO LO
o
rn IA
O X sP
o o
•Ql<-( • Ov O
i— i w
z
<
>* X
z o
M M < H
a.
< > D£ < £3
a
H >
tB
U«
m vO
O.
w->
o
oo
N
O
I— I
o o
O ' >— I
1_I
o
oo
%
Reproduced with permission of the copyright owner. Further reproduction prohibited without permission.
A
N
G
U
L
A
R
'
V
A
R
I
A
T
I
O
N
I
N
’
ZX
P
L
A
N
<N> fxl UJ 3 X U J XI o X <z X L U •3 i i *4 X 3 m (M < r\l X a < i o •+ m x a ~r> X U J »— L U ■X < a c X c z D Z U1 > 2 O u \: LTV O' I U UJ X 3 f-<1 X U J n. x U J a IS) 2 3 tt. O DC UJ C D X < X U. c U J IM ftM in 3 r-3 3 iT k
r° '
o o1
•
«
ftin o 3
r
c m
r-F °
X
x O 3
, • • •
in o o
to IM >
r— « 3 X m O '
o > : x >j; O' r\j
X i; M O' M , n
co > 3 3 4 3 ! :m
• •j • • • • •
> . r- u i 3
•o m m .■n cr\' :0
X\ JO 3D 4 n 1
n > — i rvj "M o
4 X 1 JO ?*•} •O ■o
X ! 4 ^o: JO ~~4
• • ' « • « : • m
x! —4 o| -}• X
f\J f 1
ro co ■4- x f
o 1
x ; 2) O CM ^ in X»
CM o , r- in > X
xj-30 w-t 1 r- m 3 o
!. X
C7-% • •. • « . « •
■O' 2) ' ■ 4 >0 O:'
'J-C M m to ^ ; »»■
N
b- ] « 4 o r\i m i o m
r - <NI 3D D > CM
r -3 &#-34;
in > in CM m o
m 3 ; r-- 2) X in X
' % • • • • • « • • •
Vn o o O ' X r - in ret « i O'
m r- o m ra f<^ m cn c n C M
U J »~4 ■tb <c 3 0 •— V o m n
-1— X • & « »4 > < n r - j ' C M
U J X C M P-* C M n- n r ^ C M
X X ■ o o o r - o O
< • « • *vj • ♦ • • • % %
cc in o o X >T J O C M o 2D
< c a > X *4- m m cn C M
U .
3
X
U J
m
— X
»- 3
2 3 2C in r - O
r — 1— r o C M in o - 3 D C O
O u 2 > 2 3 * ■ 4 r O
in o o U J c n C O O 2 ) O *— »
« • X • • • • • •
s\ o o V3 <r r 4 2 3 4 * C M O
m > C M c O m • C O c O
&
ft
<t
> 2 O —
a n in in 2 O lf\ X
-- 3 “'
•
X)
<x Z 4- f t CM 3 ^ 4 CM
X C O r- rM C O o • X 4
U J x O C O C O in 23 * 4 O'
»— in o o C O
4 O P ^ 3 xO
9-* • • • • ' • • • • « ♦
in o in 4 , ® 4 CM o 23
O
F if\ • ro
to a* . 4 4 4 co ro C O C M
X ■0 & 4 C O C O 3 ’ O'
U J f- 23 4 23 i'- (St CCl
2: r- O C M C M C M <3 O
X n o O x O 4 O > C O <
* U J • • * • • « • • ft 9
2 D tn o o in •-* in o •o 4
C O O' «->4 CM C O ro <x 4 in
>- \ Is- O <» X O' C M
< CD N ftft rvj c- o o < X
» - X 1 CO 4 o X o « <»
2 It X o o 10 2 ) ftft 10 ( O «>ft f
-U J X • » ft • • • ft ft ' • ft
X o o 4 s ft- N ' o X
or a m o 4 <» I O o n, m tvj
IS)
r
x
-- <
X K
X U J —
-ft* X I o t- a
i o
! X
> M
X 3
. 33 3
* « ^ (M m » jtM UJ
iu 3
in _i m <
3 >
<
3 3
C 2
t- 3
X
rxj
X «->
**i UJ 3
Z U J
3
»— UJ
O u j
X X
x
u-i
in
in 3
< ’ I? O O
t ■< O X c w^ in r\j m CM
X X X X X to 2) 2) Q
ffi O' I- l'< 4 in »—* n
X o O n <o —• ® . o •-4 2) «
ft • • • « • ft • ft % O
X 3 3 rH xt 4 CM o 2)
n O l> a <» n CO *o. fO CM n o
2 O o o
t - O
u O
UJ *
a °*
gfi £ 5 J o UJ I _J a c a a D Qt f-t > « a < H
I
u>
* ! jj I x j
2 1
3 C ■ 1
< 3t tu 0 1 I CVJ X 3 co fM M !>
X 3 3
m «
n 3 3
ro
p-IM PO
3> "V k> a O i • % •
In r 3 r\l 3 o
*o o 3
I ft • ft
in 3 3
ro rvj tP
co o < cm X
o
< o + CO o 3 at 3 u. i/i X UJ X < X <1a
z
o z . UJ > z o u UJ X 3 h-< X UJ X X UJ ■ l/l XUJ o ►- » z>
uj in
X in
«< »
X X
< m
X 5 6 5 3 O 3 • •
in o o
CO 3*
o O'
o
u_ W-* ro 3 X)
in
n in o o
*-/i X • • ft
z UJ in \3 3
3 X m > cm
►* X
H 3
< Z >
or ro i
UJ in
»— in O O
*—t » ft •
in o in
u m CO tp
a •
ro
X N
UJ X
XI X
X u X o o
3 UJ * ft •
Z 3 ■ X - o o
3 ro o
<
>
■<»
z JO
c ft* X
a
X . o o■ in . • ft «
X X 3 3
z ro CP o
o
ft* j
*- «
< X
J- »
z ■ - X o o
UJ A ft ft • *-• ft* X o o a
o r*a
<
JO t
o n
X X p—•
o
X Isl
X
a G X UJ ■— <.
c UJ X t-X P. a X •-*
«— a X
i
z
XD
I-a
1
4 nj | N o JO. rsi X i i XL 3 ■ r- X X 3 3
< 4 > ; X 3 O >i i 4 M ; x> X , m . CM i , ' « •; • ft ft ft j * > 2* n m r * 4 3
i i
cO ro | rO rO ro m CO I
1
i
i O ro _« 4 n ^ X I * 30 1 O > ■o
>-ro m , o X r^* m 3 i ft>4 « o 4 ^0 30 *“< i
ft • i ft • «> • ft j > x i :\l X O ' 4 30 t
CVJ , j
ro ro 4 i j
4 4
i I c m! W CM CO , in
i
i
; CM g tM 30
i > CM X X O r^ CM
r- 3* 4 f- r- O'
ft ft • • • « ft
o X —4 O sj
r-; oa CO CO 4 M- 4 i O
i < !' o
i i CM
o X o >o r*- X CM J- nj
X S\ X X n ro B "4 X 3
cm ro > «—* X O' n 0
35 Ch r- 4 « * • • • « » « S? CM O' X m -fO ro u ro CO CO ro CO rO CM CM ii i
‘ / » 3
* / IX _J
« X X m 3^ CM 4 CO / to <X * O F‘< o in 4 CM -J / 3 >
ro X v 4 - -O 4 n o < ro o r- ^4 3 X -5 J NJ • • ft • ft • •
4 X 4 (M C 30 C 2
£ 4 ro ro ro ro CM *“ 3
— at
< tvi i—
ac X o r\j ro CM wO r*- X o CM in O 4 30 > Z LU
o r-- X :— o O — « >» _I LU ro ro o XI O 4V in z UJ
• • • ft « 1 ft • 3
iS) 4 X 4 CM 3 x> *— M
* ’4- ro CO fO to CM 3 LU
* at jt
. ft * '. a- u. ft X C •S^ X •H X >
4 X ■4“ o tn X 4 tO CO 4 “ N- > ro O N O o in
• ft » • ft ft • >r 2) vj <M o
CO CO CO CO CM
o ■0 cO » * J) i/i
t> ro X 4 ■ * O' lO
ro in 30 O' Wft ro 4 3
<0 ro o X X -a
ft ♦ ft ft ft • % o N <n o X o X 4
eg CO CO 4 m o
O '
•—*
^*ft *
® p- o ® c CM
< 10 o X o O
« m o •hU to O JO to •—s X o ® «
♦ * • ft ft • • r o s <> nj 3 3)
<» JO JO n IO (V “ o
1 2 O
• • 1 O X
N N <t N X O o JO
X n 0> <J X <r > f, O ft o N {P <VJ X 4 O o ft o X CO N X .
ft • ft • ft ft ~ CM
ft GO N 9 * n
A’ <» n ro ro to C%J' O m
u j < u > < o :*5 D a J3 i-i • > W vJ
R
e
p
ro
d
u
c
e
d
w
ith
p
e
rm
is
s
io
n
of
th
e
c
o
p
y
ri
g
h
t
o
w
n
e
r.
F
u
rt
h
e
r
re
p
ro
d
u
c
ti
o
n
p
ro
h
ib
it
e
d
w
it
h
o
u
t
p
e
rm
is
s
io
n
.
. 24
-C O N V E N T I O N A L P A R A M E T E R S F O K Cil53 + I N C A ( O H ) 2 A T 2 3 G H Z — G E K A L O K 0 B 9 K G E
NUMRRR OF O R I E N T A T I O N S a l O N U M B E R °OF I T E «A7IUNS = 3 T E M P E K A T U K E (C ) =-122.0
SIZE OF M AT R IX = B _ SPIN VALUE* 3.5 NUMBER OF P A K A M E T E K S = 9
DPPH(MMZ)'” 35.6689 3 5 . 5 4 4 7 3 5 . 6 7 8 8 35. 5465 35.5491 35 .5502 35 . 5729 ' 3 5 7 6 7 8 8 -- 3 3 T 6 9 5 8 - ^ 3 7
“6975---THETA 9 0 . 0 ' ^ 9 0 . 0 0.0 90.0 90.0 90 .0 70.0 20.0 20.0 Vu.O
PHI 0.0 90 . 0 90.0 45.0 20.0 70.0 90.0 90.0 0.0 . 0.0
♦
S P E C T K A f M H Z ) ***
4 4 . 3 9 9 8 4 4 . 3 B 3 0 17.6352 44.3357 4 4 . 3 7 0 2 4 4 . 3 2 5 8 3 9 . 86bO 1 9 . 2 5 7 9 19.1163 3 9 . 3 5 4 7
-j » /
_________ ______ 41 .64231 4 1 . 3 4 2 2 2 5 . 3 4 0 9 41 .4304 41. 3782 "4 1.4639 38.9 7 3 0 2 6 . 7 5 7 2 _ 2 6 .6289 39 . 2 1 7 1____
'38.1022 3 8 . 0 9 1 9 30.9661 38.0352 3 8 . 0 6 4 6 3 B . 0 0 4 8 ’37.7283 3 2 . 0 1 8 9 3 1 . 9 7 4 2 3 7 . 9152
34.8671 3 4 , 8 0 2 0 3 5 . 8 8 8 4 34.7731 3 4 . 7892 3 4 . 7 6 0 6 35.8481 3 6 . 4 8 6 3 36.4832 3 5 . 9192
* i
______________ 3 2 . 2 9 5 3 3 2 . 0 1 1 7 4 0 . 8 1 0 b 32.0928 32.0577 3 2 . 1 4 1 5 33.4992 4 0 . 7 0 9 V 4 U . 6 9 8 7 _ 33.6 > 9 7
30 .1277 30. 1 269 4 6 . 4 3 0 3 30.0693 30. 1058 3 0 . 0 5 1 5 31 .5463 44 . 7 5 1 3 44 . 9 1 22 31 .4127
i
•2 B . 7223 2 8 . 5 5 7 2 5 4 . 1 2 7 8 . 2 8 . 5 8 0 4 2 8 . 5 6 6 9 2 8 . 5 8 7 2 2 9 . 8 6 2 4 4 7 . 6 9 0 0 4 8 . 2 7 1 9 3 0 . 2 6 1 4
7 ">0Ue^F'ACE “C O R R E C T I ON- 0 . 0 0 2 1 0 0 GAUSS" " P-*1)T0N MHZ TO G A U S S * 2 3 4 . 8 6 9 “--- -- ---- — — ---D P P H G V A L U E * ? . 00 3 6 0 0 FREE E L E C T K U N G V A L U E « 2 .002 2 9 0
i
♦ ,
UJ •o X UJ x o X < x i i *vj I o m c m < c m I C < o ' + U-!
CM CS
3
* X
a u. LO X LU (— LU X < a < a z UJ >
z
o
u o r-o rsi 3 o o 3 m to• 3 o
o ! * • V
x in 3 3
ro
t CM O'
a Ii
in
o
f-— o
UJ in 3 3
3C O' • • «
3 in O C
I— M ro r- O'
< LO
ac X
UJ UJ vo
u ►- O'
2: LU x
UJ X in 3 3
h- < • « *
X in c O
< ro O'
X u. •© m o. lA t X tA
• © .• o•
z LU in 3 3
a X) ro 0> CM
ftft X
)— 3
< Z 3
a O
in
i— m O O
ft • •
in o m
u. m m O' X
o *
ro
X ro
UJ X
tc
n-X ii <0 o o
3 UJ • • •
Z 3 in o o
3 to O'
< >
•j
Z CM
o « <n
H a in o o
u . in • ’ ♦ •
in' in 3 3
z ro o> O
o i
•■A
*- * ft*
< B to
>- in
z ■ >0 o o
UJ A. • • ft
M ft* in.. o o
or a to O'
o
r-< i
u. X
o N
u. X
a c X
UJ -—
-<E ui * X H
.X K
a
ttj ft*3
• •a
X XZ
m
Q 1-a
-4 ■ CM X XI
n j x ; X V x . O' -A
-h . Aft* 1 ,■>» X -o X
ro -A . X X 'O; to rvi
• » ’ ft ft • i ft •
O' O' i xv ro i ■ ^ o
to r o ; ro ro ro ro ft*n
XI X O' in * a. » 3 , 3 •O « ' ■o;
c m 1
Vfl • O' ro x O' m ifi r» in x • •o m a r'• x ' 3 f'--O • 3 ’
X ■
in o o X O' 30 • x x
J
z> X\ JNI rvj • 31 X -O rorg o r - in rg ■ 4
eg >3- r^ ^4
• • « • • « <o —i -0 o O'
* ^ rg ro ro * X
<2 'X rvj X & D
o ro roT fNJ rg > 3
in X
r - r^ X <r X
• * • * • • • •
o X r - m ro
CO to ro rO rO ro rg
* ro X f\J in rg X
r-* ft—4 > > rg in ro
ro o n X
fO r- •—< o in
NJ • « * • • • •
z r— rg o X
z 'f CO CO to rv . cv
X r - r~ O r - in ■O
►— . > —4 CM o o ■4“ IM
o o X ■O > ' X o -o
UJ ro , to O r^ o w 4 m
X • • • • • • •
CO 4- •“4 X eg 3 'J X ro CO m co CM
CM 3 ft* o> ro
O CO ft* 30 . O' CO in
fti- v1 4 ro N O' N
ro ft* O fft 3 o in
* « » » • • «
a* ft-4» ft* CM O 33
ft* ft* ro ro ro ro tM
O' C M o >o N >0 to
to < C M <* in O'
m <* O' tv to in
to to o 33 o »-4 • in
• • ft • ft • •
« - H X < * , M O X
to to to to tVJ
N N •fti to - to in V *
ftl r- to (V ■* •■ * . in
X 3 0 in o
to o o ® N N
ft • ft « * • , ft
® ■* CVJ o 9
•<* to to to ro O l
3 O'
ru
O' CM
■o 3
X> o
• •
X CM
fCl u
CM uj
tt
VO _i
y* <
3 >
<
C Z *“ 3 X
tvi ►—
X O X iu
3
X UJ
3
— LU
3 xi X X
X U_
!> A- > in co 3 r- CA
O. >* in o X r- o *<Si
rO in X o CM X
-o ro o X X A* o <l
ft * « • ft * » O
f - m o in o •o <3
tM CO CO * 4 to o CM
c c
; o Z O
O o
n
*- o
O o
UJ .
jrw
a a
s §
uj <
o >
<
“■
a
3 1 r a 5? a
a o
- \ X} H • > CO u