Trabeculectomy versus Ahmed Glaucoma Valve implantation in neovascular glaucoma
Full text
Figure
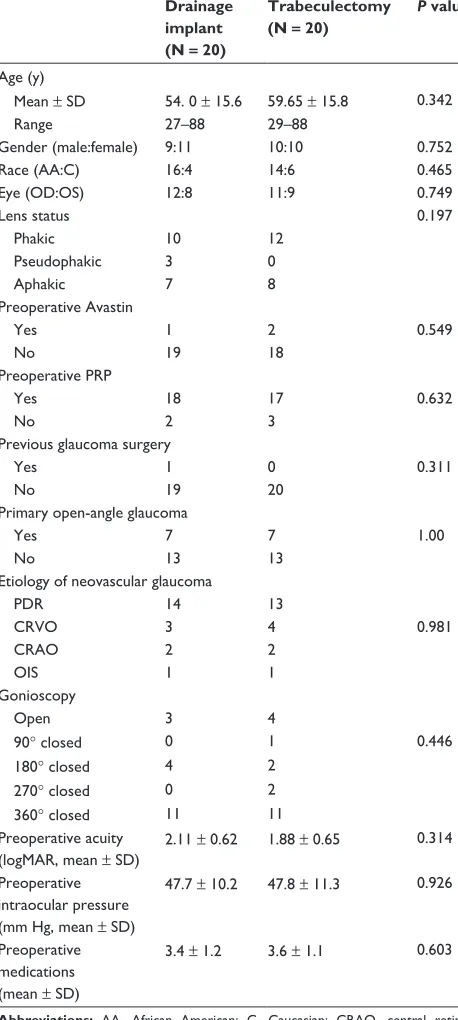
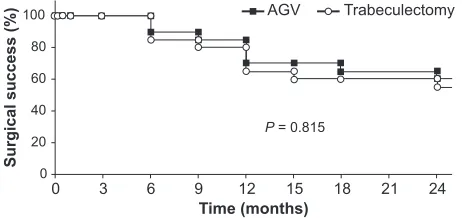
Related documents
© The Math Learning Center | mathlearningcenter.org Bridges in Mathematics Grade 4 Home Connections 29. Base Ten
Natasa Miteva, Ph.D., Goce Delcev University of Stip, Faculty of Tourism and Business logistics, Stip, North Macedonia.. Aneta Stojanovska - Stefanova, M.Sc., Teaching
It is restricted to patients with a history of recurrent cyst infection , recurrent major bleeding ,complicated lithiasis , severe Hypertension ,or massive renal
extended zero" method will be used for Gauss elimination algorithm in setion
While addressing the effectiveness of CBL and PBL as learning vehicles in comparison to traditional didactic lectures, the study also compares CBL and PBL as an
Findings of the present study indicated that the mean score of the dimension of qualitative research was different between the study groups, so that the scores obtained
Included in this study were nine patients with the same documented diagnosis in both eyes of either ocular hypertension, glaucoma suspect, or mild open-angle glaucoma; one eye of