Serum
Immunoglobulins
in Preterm
Infants:
Comparison
of Human
Milk and Formula
Feeding
Erkki
Savilahti,
MD, Anna-Liisa
Jarvenpaa,
MD, and
Niels C. R. R#{228}ih#{228},
MD
From the Children’s Hospital, University of Helsinki, Finland
ABSTRACT.
Serum concentrations of immunoglobulins(IgA, IgG, and 1gM) were studied in 64 preterm infants with gestational age of 31 to 36 weeks (mean 33.2 weeks), between 1 week and 4 months after birth. Infants were fed solely human milk or formula based on cow’s milk. Infants fed formula exhibited significantly higher IgA levels at the age of 9 to 13 weeks than infants fed human milk. Infants given human milk who received more than 60% of their feeding in the hospital from their own mother had significantly higher IgA levels at the age of 3 weeks than did those receiving less than 30% of their feeding from their own mother. At 1 week of age, formula-fed infants with gestational age of 31 to 33 weeks had a significantly lower mean IgG concentration than those with gestational age of 34 to 36 weeks. However, the two term subgroups of infants receiving human milk, had similar mean concentrations of IgG and the value for infants of 31 to 33 weeks of gestation was significantly higher than that for formula-fed infants at the same gestational age. Serum 1gM concentrations were similar whether infants were receiving human milk or formula. The formula feeding caused a more rapid maturation of IgA than did human milk. Results suggest that preterm infants may absorb IgA from the milk of their own mothers and that preterm infants with gestational age of 31 to 33 weeks may absorb IgG from human milk. Pedi-atrics 1983; 72:312-316; preterm infants, serum immuno-globulins, formula feeding, human milk feeding.
The preterm newborn infant is capable of specific immune responses at the time of birth,”2 but he is born with a lower serum immunoglobulin G (IgG) concentration than the full-term infant.3 It is
Received for publication June 28, 1982; accepted Dec 20, 1982. Dr R#{228}ih#{224}’spresent address: Department of Pediatrics,
Univer-sity of Lund, Malm#{246}, Sweden.
Reprint requests to (E.S.) Children’s Hospital, University of Helsinki, SF-00290 Helsinki 29, Finland.
PEDIATRICS (ISSN 0031 4005). Copyright © 1983 by the American Academy of Pediatrics.
widely thought that the newborn infant does not
absorb colostral antibodies.7’8 Proteins and their hydrolytes and microorganisms in the gut are the major antigenic stimuli for the newborn infant. These stimuli may have different effects, depending
on whether the infant receives breast milk or cow’s milk formula.9 Levels of serum IgA rise more rap-idly in formula-fed term infants than in breast-fed term infants, while 1gM values show no difference.’#{176} The effect of feeding on the development of serum immunoglobulins in preterm infants has not been studied. In the present investigation we have
meas-ured serum immunoglobulins in preterm infants who have been fed from birth with either human milk or adapted formulas.
METHODS
Patients
The study included 68 preterm infants (33 girls and 35 boys) whose gestational age ranged from 31 to 36 weeks (mean 33.2 weeks) as calculated from the last menstrual period of the mother and deter-mined clinically.” The patient data are described in detail elsewhere.’2”3 For purposes of statistical analysis two groups were formed on the basis of gestational age (Table). The infants’ birth weight was appropriate for gestational age as assessed by Finnish intrauterine growth charts.’4 None of the infants had major medical problems.
The infants were followed in the hospital until they attained a weight of 2,400 g (at a mean age of 5 weeks). Venous blood samples were taken bi-weekly during the hospital stay. After discharge, the infants were examined at the outpatient clinic
IGA mg/
1 3 5 7 9 11 131517
AGE (weeks)
Fig 1.
Mean levels of serum IgA in infants receivingformula (diamond) or human milk (circle). Standard error of the mean is indicated. Star indicates that differ-ence between groups is statistically significant (P < .05).
TABLE.
Birth Weight (g) of Studytional Age and Feeding Group*
Infants by
Gesta-Gestational Human Milk Age
Formula
31-33 wk 1,792 ± 210 (19)
34-36 wk 1,835 ± 150 (12) Total (31-36 wk) 1,809 ± 187 (31)
1,753 ± 197 (20) 1,926 ± 128 (17) 1,832 ± 188 (37)
* Values are means ±SD; the number of infants is shown in parentheses.
Feeding
The infants were fed solely with human milk or formula throughout the study. In the hospital, ap-proximately 55% of human milk was pooled from donors and frozen; it was pasteurized for 30 minutes at 62.5#{176}Cbefore feeding. Additionally, 35% of the human milk consumption was expressed milk from the individual infants mother; this milk was stored at 4#{176}C,fed within two days, and pasteurized before being fed. The remainder of the total milk intake
(approximately 10%) was given by breast-feeding. The proportion of the human milk delivered by the infant’s own mother was calculated for each child and for statistical analysis three groups were formed: those receiving less than 30%, those receiv-ing between 30% and 60%, and those receiving more than 60% of their total milk consumption from their own mother. After discharge from the hospital the infants were nursed directly by their mother.
Formula feeding was started at the age of 2 to 3 days. Before the formula was introduced, infants were given pooled, pasteurized human milk at vol-umes of 60 to 80 mL/kg/d. The formula had a protein concentration of 1.45 g/dL and a whey!
casein ratio of 60:40.12 The formula was manufac-tured as powder (Wyeth Laboratories), and it was heat-sterilized (62.5#{176}Cfor 30 minutes) after recon-stitution before being fed.
Measurements
Serum immunoglobulins were measured by single radial immunodiffusion and compared with a serum standard of known immunoglobulin content, which had earlier been compared with the WHO standard 67/97.’ Commercial antiserea (Hyland Laborato-ries) to IgA, IgG, and 1gM were used.
The infants were divided according to their diet, their gestational age (31 to 33 weeks and 34 to 36 weeks), and the origin of human milk (less than
30%,
30% to 60%, and more than 60% from their own mother). Immunoglobulin concentrations in the different subgroups were compared by Student’st test with a computer. Means, standard deviations, and correlations of the measurements were calcu-lated.
RESULTS
lmmunoglobulin
A
Only four infants had a measurable IgA concen-tration at the age of 1 week (>0.5 mg/dL). At 3, 5, and 7 weeks of age there were no significant
differ-ences betwen infants fed human milk and those who were formula fed (Fig 1). The subsequent in-crease in IgA was faster in the infants receiving formula than in those receiving human milk, and
the difference became statistically significant at the age of 9 weeks (5.6 v 2.8 mg/dL; P < .05) and 13 weeks (7.3 v 3.5 mg/dL; P < .05) (Fig 1). In infants receiving human milk, a significant decrease in
serum IgA values took place between ages 6 weeks and 8 weeks (4.2 v 2.8 mg/dL; P < .05) (Fig 1).
The increase in IgA values in infants receiving human milk at gestational age of 31 to 33 weeks was similar to that seen in infants receiving human milk at gestational ages 34 to 36 weeks. In contrast,
formula-fed infants with gestational ages of 34 to 36 weeks had higher IgA values after the age of 3 weeks than did formula-fed infants with gestational ages of 31 to 33 weeks. The difference between these gestational age groups became significant at
age 11 to 13 weeks (9.4 v 4.9 mg/dL; P < .05). Those infants who were fed human milk and got most of it (more than 60%) from their own mother had a significantly higher mean IgG concentration at the age of 3 weeks than those getting less than 30% of their total milk consumption from their own mother (2.1 v 0.7 mg/dL, P < .05). This difference decreased after age 3 weeks.
Immunoglobulin
G
Up to age 9 weeks, there were no differences in mean IgG concentration between the two feeding
IGG /di
1000
900 800
700
600 500 400 300
200 100
1 3 5 7 911131517
AGE(vk)
Fig 4.
Mean serum IgG level in infants receiving humanmilk at gestational ages 31 to 33 weeks (solid circle) and at gestional ages of 34 to 36 weeks (open circle with dot).
1000 900 800
700 600
500
400
300
200
100
1 3 5 7 911131517
AGE (w..ks)
Fig 2.
Mean serum IgG level in infants fed formula(diamond) and human milk (circle).
IGG % 1200
1100 Iv
1000 900 800
700 tN 600
500
.1
400 ‘l
300
200
100
1 3 5 7 911131517
AGE (weeks)
Fig 3.
Mean serum IgG level in infants receivingfor-mula at gestational ages 31 to 33 weeks (solid diamond) and at gestational ages 34 to 36 weeks (open diamond with dot). Star indicates that difference between groups is statistically significant (P < .05).
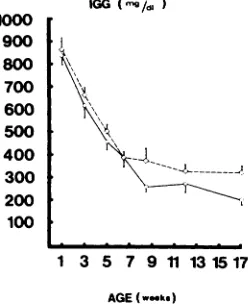
groups (Fig 2). Beginning at 11 weeks, the IgG
values for infants fed human milk was lower. IgG levels at 1 and 3 weeks showed a significant positive correlation with the gestational age of the infants (R .323 and .317). In the formula-fed in-fants, IgG values were significantly lower in the
group of infants at 31 to 33 weeks of gestation than in infants at 34 to 36 weeks of gestation (at age 1 week, 700 v 1,O4Omg/dL; P < .05, Fig 3) until the age of 5 weeks. Thereafter the statistical difference
disappeared (Fig 3).
At 1 week of age, preterm infants fed human milk
had similar IgG levels whether their gestational age was 31 to 33 weeks or 34 to 36 weeks (Fig 4). The mean IgG values of infants receiving human milk with gestational age of 31 to 33 weeks was signifi-cantly higher than values for formula-fed infants of the same gestational age (840 v 700 mg/dL; P <
.05).
The less mature infants receiving human mikcontinued to have higher IgG than the more mature infants throughout the follow-up period.
Immunoglobulin
M
The change in 1gM concentration was similar in the two feeding groups throughout the study (Fig
5).
IGG
(s/)
Correlations
Between
Immunoglobulin
Values
There was a positive correlation between the concentrations of IgA, IgG, and 1gM in each mdi-vidual infant, measured at the same time. 1gM values showed a positive corrrelation with IgG
con-centrations measured on the following two visits in 7/11 occasions.
DISCUSSION
The human fetus receives passive humoral im-munity from the mother via placental transport of the IgG class of antibodies; this is an active process during the last 20 weeks of gestation.3’8 It is widely accepted that full-term infants do not absorb any of the antibodies of colostrum or mature milk.7’8 These antibodies are predominantly secretory IgA.’6 The milk of mothers of preterm infants con-tains an increased concentration of IgA.’7 Human milk reduces the antigenic stimuli of the newborn baby by offering species-specific protein and a pas-sive immunity to intestinal pathogens.7”6 Its pro-tective effects are, therefore, particularly important for preterm infants. In this study we report the different effects of formula and human milk diets on the quantitative development of immunoglobu-lins in preterm infants.
We found a more rapid increase in serum IgA
in-1GM “#{176}/. I
I
35 30 25 20 15 10 51 3 5 7 911131517
REFERENCES
1. Pabst HF, Kreth HW: Ontogeny ofthe immune response as a basis of childhood disease. J Pedio,tr 198097:519
2. Gitlin D, Biasucci A: Development of ‘yG, yA, ‘yM, 91C,
/filA, C’l esterase inhibitor, ceruloplasmin, transferrin,
hemopexin, haptoglobin, fibronogen, plasminogen, al-anti-trypsin, orosumucoid, j-lipoprotein, a2-macroglobulin, and prealbumin in the human conceptus. J Clin Invest l9648:l433
3. HObbs JR, Davis JA: Serum -yG-globulin levels and gesta-tional age in premature babies. Lancet 1967;l:757
4. Rothberg RM: linmunoglobulin and specific antibody syn-thesis during the first weeks of life of premature infants. J
Pediatr 1969;75:391
5. Berg T: Immunoglobulin levels in infants with low birth weights Acta Paediatr Scand 1968;57:369
6. Pilgrim U, Fontanellaz HP, Evers H, et al: Normal values of immunoglobulins in premature and in full-term infants, calculated as percentiles. Helv PoediatrActa 1975;30:121
7. Butler JE: Lmmmunologic aspects of breast feeding,
antiin-AGE (ees)
Fig
5.
Mean serum level of 1gM in infants fed formula (diamond) or human milk (circle).fants probably reflects a faster maturation of the
local IgA-producmg system in the intestine, a
re-sponse that may also be more rapid in the more mature infants. The developmental pattern of serum and intestinal IgA are correlated in healthy
infants.’8 The claim that colostral IgA is not ab-sorbed has not found complete acceptance.19 We found a more rapid increase in IgA values during the first three weeks of life in the infants whose major source of human milk was their own mother, relative to those receiving mostly banked human
milk. This observation may indicate that a greater amount of IgA is absrobed from the colostrum of the preterm infant’s own mother than from pooled, banked human milk. This difference may be due to the higher IgA content of milk from mothers deliv-ering preterm infants compared with milk from mothers delivering full-term infants.’7
We
observeda significant
decrese in serum IgA values in infantsreceiving human milk from ages 6 to 8 weeks. Preterm infants have been shown to absorb bovine
jI-lactoglobulin more than term infants during the first days of life.#{176}Therefore, the decrease in IgA
values could be related to gut closure of the preterm infants to IgA in human milk.
Umbilical cord IgG values have been found to be higher in mature newborn infants than in the
pre-term infant. The positive correlation with
gesta-tional age is linear from 27 weeks on.3 In the
present study,
we found
this
samepositive
correla-tion between gestational age and IgG concentration at 1 and 3 weeks of age. However, the infants receiving human milk at gestational ages of 31 to 33 weeks had higher mean IgG concentration than babies of the same gestational age who were fed
formula. This may be due to the small preterm infant’s greater ability to absorb colostral IgG com-pared with the full-term infant. Feeding of fresh human milk to preterm babies has been reported to result in higher IgG levels relative to the values of
infants who received heated (3 minutes at 100#{176}C) human milk.2’
At
5 weeks of age the correlation betwen serum IgG values and gestational age was lost with therapidly
decreasing
level
of IgG.
Later
on, infants
fed human milk had lower IgG levels than infants fed formula. This lower concentration may be due to the decreaed antigenic stimuli of human milk, as
is also reflected in the lower IgA content. The protein concentration of the infant’s diet has also been shown to affect the IgG concentration: Zoppi et al found lower IgG levels in infants who had a protein intake of 2.5 g/kg/d than in infants receiv-ing 4 g/kg/d. In our study, the difference in protein intake was small (1.8 g/kg/d in the infants receiving human milk and 2.2 gfkg/d in infants receiving formula).
A positive
correlation
between
IgG and IgA levelshas been demonstrated in adults.’ We found a
positive correlation between all three immunoglob-ulin classes measured in preterm infants. We found a frequent correlation between 1gM levels and IgG levels measured at follow-up. This finding is in agreement with the assumed sequence of 1gM re-sponse and subsequent production of IgG antibod-ies.2
Our findings in this study are in agreement with previous studies on the general development of IgA, IgG and 1gM in preterm infants. Additionally, we found a more rapid increase in IgA in formula-fed preterm infants. Our results indicate that
absorp-tion of colostral IgG in preterm infants of gesta-tional age 31 to 33 weeks and absorption of IgA from the infant’s own mother’s breast milk may take place.
ACKNOWLEDGMENT
fectious activity of breast milk. Sem Perinatol 1979;3:255 8. Gitlin D: Development and metabolism of immunoglobulins,
in Kagan BM, Stiehm ER (eds): Immunologic Incompetence.
Chicago, Year Book Medical Publisher, mc, 1971, p 3 9. Long 55, Swenson RM: Development of anaerobic fecal
flora in healthy newborn infants. J Pediatr 1977;91:298 10. Saarinen UM, Pelkonen P, Siimes MA: Serum
immunoglob-ulin A in healthy infants: An accelerated postnatal increase in formula-fed compared to breast-fed infants. J Pediatr 1979;95:410
11. Dubowitz LMS, Dubowitz V, Goidger C: Clinical assessment of gestational age in the newborn infant. J Pediatr 1970;77:1 12. J#{227}rvenp#{227}AA-L, R#{228}ih#{228}NCR, Rassin DK, et al: Feeding the
low-birth-weight infant: 1. Taurine and cholesterol supple-mentation of formula does not affect growth and metabo-lism. Pediatrics 1983;71:171-178
13. J#{228}rvenp#{225}#{228}A-L, RAih#{228}NCR, Rassin DK, et al: Preterm infants fed human milk attain intrauterine weight gain. Acta
Paediatr Scand 1983;72:239
14. B#{224}ckstr#{246}mL, Kauppinen MA: Lapsen siki#{246}naikainen pai-nonkasvu. Suom LO,dkdril 1968;23:1553
15. Savilahti E: IgA deficiency in children: Immunoglobulin-containing cells in the intestinal mucosa, immunoglobulins in secretions and serum IgA levels. Clin Exp Immunol
1973;13:395
16. Hanson LA, Carlsson B, Dahlgren U, et al: The secretory IgA system in the neonatal period. Ciba Found Symp
1980;77:187
17. Gross SJ, Buckley RH, Wakil SS, et al: Elevated IgA con-centration in milk produced by mothers delivered of preterm infants. J Pediatr 1981;99:389
18. Savilahti E: Immunoglobulin-containing cells in the intes-tinal mucosa and immunoglobulins in the intestinal juice in children. Clin Exp Immunol 1972;11:415
19. Iyengar L, Selvaraj RJ: Intestinal absorption of immuno-globulins by newborn infants. Arch Dis Child 1972;47:41 1 20. Roberton DM, Paganelli R, Dinwiddie R, et al: Milk antigen
absorption in the preterm and term neonate. Arch Dis Child
1982;57:369
21. Gribakin SG, Kuvaeva IB: Blood serum immunoglobulins in premature infants and their dependence on the method of feeding. Vopr Okhr Materin 1979;24:30
22. Zoppi G, Zamboni G, Siviero M, et al: -y-Globulin level and dietary protein intake during the first year of life. Pediatrics
1978;62:1O1O
23. Allansmith M, McClellan B, Butterworth M: Evidence for the independence of human immunoglobulin class levels.
Immunology 1967;13:483
COUGH VARIANT
Hannaway et al evaluated 32 children who had been referred to them because of chronic coughing; the children were followed over a five-year period. The mean duration of cough was 20 months. The cough was nocturnal (23/32
patients) and exercise induced (25/32), and in all patients the coughing was triggered by upper respiratory tract infections. In all cases the cough was unresponsive to antitussives, antibiotics, and antihistamine-decongestant com-binations. All 32 patients had a significant response to a trial of oral theophylline in appropriate dosage; 18 of the 24 patients ultimately developed other symp-toms compatible with asthma.
Comment: Even though this study was small and unsophisticated, it
empha-sized several points: (1) the astute clinician in practice can add much to the
understanding of various disease processes, and (2) the article “blows the whistle” on unnecessary and inappropriate use of antitussives, antihistamine, antibiotics, etc, to control the annoying “diehard” cough. The term “variant
asthma” has been used by Carrao (N Er&gl J Med
1979;300:633).
I am not sure whether this variant asthma is a separate entity, or perhaps an unrecognized slow surfacing of the full-blown entity we know as asthma. I think the messageto be conveyed is that in the absence of clinical evidence of other causes of chronic coughing, a therapeutic trial of theophylline should be considered. (M. Barton, MD)