Fundamentals of Nuclear Science & Engineering pdf
Full text
Figure

![Table 3.2. Nuclear density parameters for three spherical nuclei.From Cottingham and Greenwood [1986] using data from Barrettand Jackson [1977].](https://thumb-us.123doks.com/thumbv2/123dok_us/933039.606155/70.612.57.411.262.477/nuclear-density-parameters-spherical-cottingham-greenwood-barrettand-jackson.webp)

Related documents
Jorgensen, B.B., and Revsbech, N.P. Diffusive boundary layers and the oxygen uptake o f sediments and detritus. Role o f the organic layer on stones in detrital metabolism in
Maximum DC output voltage of 1 V is obtained from input amplitude level of 0.16 V using 7-stage charge pump rectifier6. In the other hand maximum power efficiency of 23 % is
86. Define and briefly explain what is meant by each of the following terms. a) strategic vision b) stretch objectives c) strategic objective d) balanced scorecard e)
In 2005 and 2006 winter and spring subplots, total percent plant cover, cover crop or cotton cover, and Palmer amaranth (Amaranthus palmeri Wats.) and common purslane
XTensions for multiple apps including super resolution, cell tracking, filament tracing, object detection, GPU deconvolution, inter object relationships, batch
In health care facilities, mercury can be found in medical instruments (such as thermometers, blood pressure instruments, gastrointestinal tubes, dilation and feeding tubes),
These latter effects may be due to a RESPIRATORY DYSKINESIA, involving choreiform movement of the muscles involved in respiration or abnormalities of ventilatory pattern
To return to the language of the glorious first draft of your Arcades project: if the dialectical image is nothing but the way in which the fetish character is perceived in
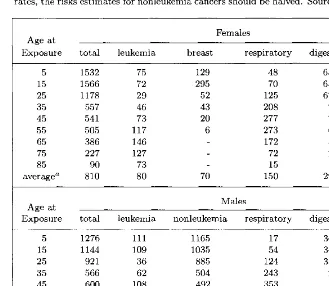
![Table 9.9.UNSCEAR committees. Source: Natural incidence of hereditary illness as estimated by the BEIR and NAS [1990] and UN](https://thumb-us.123doks.com/thumbv2/123dok_us/933039.606155/256.612.84.398.72.214/unscear-committees-source-natural-incidence-hereditary-illness-estimated.webp)