iQ)bcr
~"P
tndian
i.
Plant Phys/oi.,Vol.
XXXtrt.
No.1.
pp. ~19-22$.(Sept.,
t~)DEVELOPMENT OF MITOCHONDRIA IN HORSE GRAM COTYLEDONS DURING GERMINAnON
D.KARUNAGARAN·AND~RAM~SHNARAO
Department of Biochemistry, Sri Krishnadevaraya University Anantapur-SIS 003
Received on 18 June, 1990
SUMMARY
Time-course of changes in activities of mitochondrial enzymes in cotyledons of germinating horse gram seeds showed an upward trend during the initial stages and a decline during the later stages of germination. While succinate dehydrogenase artd malate dehydrogenase activities enhanced upto day I, activities of cytochrome-c oxidase, 2 oxog)utarate dehydrogenase, isocitrate dehydrogenase and glutamate dehydrogenase increased upto day 3. Treatment with cycloheximide or chloramphenicol had no inhibitory effect on appearance of the enzyme activity, Mitochondrial maturation in horse gram is probably m=diated by activation of pre-existing enzymes rather than by their de novo synthesis. Influence of axis was indicated by the restricted development of enzyme activity in the absence of axis.
INTRODUCTION
Mitochondria in the cotyledons of dry seeds of legumes are structurally and enzymatically underdeveloped. During germination mitochondria become well developed to meet the energy-requirement for utilization of the mobiJized food reserves. Mitochondrial development takes place by the repair and activation of pre-existing immature mitochondria or biogenesis of new mitochondria (Bewley and Black, 1978; Morohashi, 1986). As the cotyledonary reserves get depleted, mito
chondria become disorganized and gradually Ios~ their respiratory efficiency, enzyme
complement and activity (Bewley and Black, 1978). Earlier studies on mitochon drial development have been restricted to a few leguminous seeds with no detailed information on the biochemical processes. Therefore, the present study is aimed at understanding the mode of development of mitochondria in cotyledons of germinat ing horse gram see.ds in terms of enzymatic changes. Time-course of changes in
220
...~. . . ~
..
'.~r~-~:;"~;'·
,
activities of a few mitochondrial enzymes were followed for a period of 4 days. Effects of cytosolic and mitochondrial protein synthesis inhibitors on activities of succinate dehydrogenase and cytochrome-c oxidase were also investigated. Since embryonic axis is suggested to influence the development of mitochondria (Morohashi and Bewley, 1980) and hydrolases (Morohashi. 1982; Sharma, 1988) during germination, possible role of axis was examined as well.
MATERIALS AND METHODS
Uniform-sized seeds of Co 1 variety of horse gram (Macrotyloma uniflorum Lam.) used in this study were procured from Tamil Nadu Agricultural University,
Coimbatore. Seeds were surface sterilized with 0.1
%
mercuric chloride for 5 minand rinsed thoroughly with distilled water. Seeds were then soaked in water for 4 hrs and a bafch of 20 seeds was spread on two layers of filter paper, in a 10 cm Petri dish, moistened with 8 ml of water. These were placoo in a wooden tray, covered tightly with polythene sheets and left for germination in dark at 28±2°C. Sterile conditions were maintained by including 20 ppm of streptomycin sulfate in water and test solutions. To evaluate the influence of axis, seed coats were removed and cotyledons separated from each other, so that the axis remained attached to one of them. The cotyledons with (attached) or without (detached) axis were incubated under the conditions described, for the specified length of time. The time, when the
plant materials were transferred to the Petri dishes was considered as day O.
Preparation of mitochondrial fraction : Mitochondria were isolated according to the method of Bonner (1967). Cotyledons were ground in a mortar and pestle
at 0-4°C using a solution containing 0.3 M mannitol, 0.1
%
BSA, 0.05~1,. cysteineand 1 mM EDTA (tissue; medium=l : 2). The pH was maintained at 7.2 by adding a few drops of 1 N KOH and the homogenate was filtered through cheese cloth, centrifuged at 1000 X g for 15 min and the supernatant was centrifuged at 10,000 X g. for 15 min in a refrigerated centrifuge Remi K24A model. The mitochondrial pellet was washed. resedimented and suspended in K-Pi buffer (0.1 M. j>H 7.2) for use.
MITOCHONDRIA IN GERMINATING HORSE GRAM 221
RESULTS AND DISCUSSION
Time-course of changes in mitochondrial enzyme activity: Mitochondria are important centres of energy production and changes in metabolic state of cotyledons
may be reflected through the pattern of mitochondrial enzyme activity. Mitochondri~l
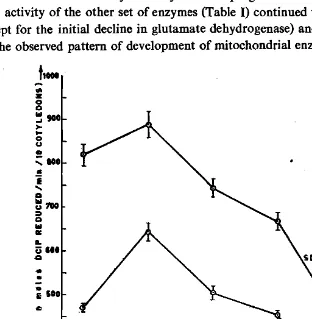
enzyme activities assayed in the cotyledons of horse gram during germination exhibited two discernible patterns of development over a 4-day period. The activities of succinate dehydrogenase (SDH) and malate dehydrogenase (MDH) showed (Fig. 1) an initial increase from day 0 to day 1 with a progressive decline thereafter. How ever, activity of the other set of enzymes (Table I) continued to increase upto day 3 (except for the initial decline in glutamate dehydrogenase) and then declined on day 4. The observed pattern of development of mitochondrial enzymes in horse gram is
t,...
OIl
z 0
~ 100
..
a 0 ~ U !! ...0 C i ... D ~,.. a ~ III•
L iiltt Q•
-
•
i
sooc
>
~-0:: u c :300 a OIl !to 0.t
. 'J.t
t
-
OIla
•
a
III
1.2
...
.
;;
u ~ ... 2·'•
I ......
2·. I! ) ( c <l 2.0
..
:; ~1.1 u ~
c :z: Cl z 1-2 0.' 0.0
t 2 3 4
liME (DAYS) __
-222
D. ICARUNAGARAN AND P. RAMAKlUSHNA RAOTable I. Changes in activity of some mitochondrial enzymes in the cotyledons of horse gram during germination
Time (days)
2.()xoglutarate
dehydrogenasea dehydrogenaseIsocitrate b dehydrogenasec Glutamate
Cytocbrome-c oxidased
o
4.26 ±0.08 26 ±0.73 2.84 ±0.06 1.69 ±0.041 4.57 ±0.07 96*+1.93 1.12*+0.04 3.11*+0.09
2 8.33*+0.20 153*+3.67 3.41*+0.11 3.85*+0.08
3 11.79*+0.22 177 +4.27 5.22*+0.18 4.68*+0.07
4 2.44*+0.04 38*+0.83 1.39*+0.04 2.76*+0.03
Each value is the mean ± SE of six values.
&. Activity expressed as nmoles of ferrocyanide Hberated/hr/l0 cotyledons.
b. Activity expressed as nmoles 'of products (2, 4-DNP hydrazones of 2-oxoglutarate and NADPH) formed X lo-'/min/l0 cotyledons.
c. Activity expressed as nmoles of oxoglutarate formed/hr /10 cotyledons. d. Activity expreSSed as 6. A X 10.../min/10 cotyledons.
*Significant difference at each day interval (p
<
0.001).Table
n.
Effects of cyclohe,ximide and chloramphenicol on mitochondrial succinatedehydrogenase and cytochrome-c oxidase activities
Succinate dehydrogenase activity (nmoles of Cytochrome-c oxidase activity (6. A x 10-a/
dichlorophenol indophenol reduced/min/lO min/lO cotyledons) cotyledons)
Time Control 0.1 mMCH 2mMCP Control 0.1 mMCH 2mMCP
(Days) treated treated treated treated
89Cl+29 900+31 910+32 3.11+0.09 3.20+0.10 3.19+0.13
2 745+23 750+25 740+26 3.85+0,08 3.90+0.12 3.87+0.11
3 669+21 680+23 670+24 4.68+0.07 4.71+0.16 .4.70+0.12
4 433+16 440+15 450+18 2.76+0.03 2.63+0.08 2.68+0.06
Each value is the mean±SE of six values.
No significant difference between control and treated.
consistent with that in starch-storing legumes (Morohashi, 1986). Nawa et al. (1973), reported a decrease in mitochondrial respiratory activity in pea cotyledons at the later stages of germination accompanied by a loss of mitochondrial enzymes.
Effect ofprotein synthesis inhibitors: Seed germination was completely inhibit ed by cycloheximide (CH) at a cone. of 0.1 mM (no protrusion of radicle occurred
J
MITOCHONDRIA IN GERMINATING HORSE GRAM
223
radicle on day 1 as compared to 1.2 cm in control). However, both CH and CP
failed to inhibit the development of SDH and cytochrome oxidase (Table II) in
horse gram suggesting tbat the de novo syntbesis of proteins may not be involved in
the development of mitochondrial enzymes. These results indicate that tbe develop ment of mitochondria is achieved by incorporation of pre-existing cytoplasmic pro teins into tbe immature mitochondria as is the case with peas and soybeans (Malhotra et al., 1973; Nawa and Asahi, 1973; Singh et al., 1987).
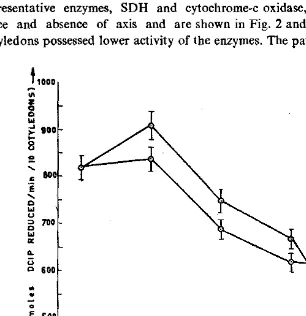
Effect ofaxis excision on enzyme activity: The changes in the activities of two representative enzymes, SDH and cytochrome-c oxidase, were followed in the pre sence and absence of axis and are shown in Fig. 2 and 3, respectively. Detached cotyledons possessed lower activity of tbe enzymes. The pattern of enzyme activities
t,ooo
....
I
11'1'"
100 ...I§
!!
...
,,800 i
...
c
u
'"
E100
DC
'"
Q.
u
c 600..
•
-
0Iii 500
c
>
...
400:>
...
e.,):z:
'"
10011'1
oT
!a
0 Z '3 4
TIME fDAYS1_
Fig. 2. Effect of axis excision on mitO<?hondri~1 succi~ate dehydrogen~ activity in the cotyledoQs
224
• =•
~~~':~.'
. . ..,.-, : . ' .
.
.
D. KARrJNAOARAN AND P. RAMAKRISHNA RAO
~·l
r;
t~
:>:
:·2
v
Q
....
>
u
,.8
'.6
o.oT !
0 2 4
TIME (DAYS) __
Fig. 3. Effect of axis excision on mitochondrial cytocbrome-c oxidase activity in borse gram
cotyledons
was similar in attached and detached cotyledons for both enzymes upto day 3. From day 3 to day 4 no perceptible changes were noticed in detached cotyledons. In agreement with the present work, excision of axis resulted in a relatively lesser development of mitochondrial enzyme activities in peanut cotyledons (Morohashi
et 01., 1981) and cucumber (Morohashi and Matsushima, 1988). However, in peas lack of embryonic axis control over mitochondrial activity has been reported
REFERENCES
Bergmeyer, H.U. (1974). Methods of Enzymatic Analysis. Ed. 2. Academic Press, New York. Bernath, P. and Singer, T.P. (1962). Succinic dehydrogenase. Methods Enzymol., 5 : 597-614. Bewley, J.D. and Black, M. (1978). Physiology and Biochemhtry of seeds in relation to germina
tion. 1. Development, Germination and Growth. Springer-Verlag, Heidelberg. Bonner, W.O. Jr. (1967). A general method for the preparation of plant mitochondria. Methods
Enzymol., 10 : 126-133.
Kalra, S.K. and Brooks, J.L. (1979). Behaviour of some mitochondrial enzymes in ripening tomato fruit. Phytochemistry., 18: 2017-2019.
King, J. (1967). Thesis, Institute of Medical Laboratory Technology, London.
Malhotra, S.S., Solomos, T. and Spencer, M. (1973). Effects of cycloheximide,D-threo chloramphenicol, erythromycin and actinomycin D.on de novo synthesis of cytoplasmic
and mitochondrial proteins in the cotyledons of germinating pea seeds. Planta, 114 :
169-184.
Marbach, I. and Mayer, A.M. (1982). Lack of embryonic axis control in Pisum cotyledon
mitochondria. Phytochemistry, 21: 823-826.
Morohashi, Y. (1980). Development of mitochondrial activity in pea cotyledons followmg imbibition. Influence of the embryonic axis. J. Exp. Bot., 31: 805-812.
Morohashi, Y. (1982). Control of development of amylolytic and proteolytic activities of germinating blackgram seeds. Physiol. Plant., 56 : ]89-193.
Morohashi, Y. (1986), Patterns of mitochondrial development in reserve tissues of germinated seeds. A survey. Physiol. Plant., 66 : 653-658.
Morohashi, Y. and Bewley. J.D. (1980). Development of mitochondrial activities in pea cotyle· dons during and following germination of the axis. Plant Physiol., 66 : 70-73.
Morohashi, Y., Bewley, J.D. and Yeung, E.C. (1981). Biogenesis of mitochondria in imbibed peanut cotyledons: Influence of the axis. J. Exp. Bot., 32: 605-613.
Morohashi, Y. and Matsushima, H. (1988). Effect of embryonic axis on the development of glyoxysomes, plastids and mitochondria in cotyledons of germinated cucumber seeds.
J. Plant Physiol., 132 : 279-283.
Nawa, Y and Asahi, T. (1973). Biochemical studies on development of mitochondria in pea cotyledons during the early stage of germination. Effects of antibiotics on the develop ment. Plant Physiol., 51 : 833-838.
Nawa, Y., lzawa, Y. and Asahi, T. (1973). Effect of pea embryo on formation and degeneration of the mitochondrial membrane in cotyledons during germination. Plant Cell Physiol.,
14 : 1073-1080.
Ochoa, S. (1955). Malic enzyme. MPlhods Enzymol., 1 : 739-741.
Pearl, W., Cascarano, J. and Zweifach, B.W. (1963). Microdetermination of cytochrome c oxidase in rat tissue by the oxidation of N-pbenyl-p-phenylene diamine or ascorbic acid. J. Histochem. Cytochem., 11 : 102-107.
Reed, L.J. and Mukherjee, B.B. (1969). «-Ketoglutarate dehydrogenase complex from &::herichia coli. Methods Enzymol., 13 : 55-61.
Sharma, S.G. (1988). Regulation of reserve protein mobilization during germination and early seedling establishment in chick pea. Indian J. Plant Physiol., 31 : 193-196.
Singh, V.K., Misra, H.S. and Misra, D.P. (1987). Behaviour of mitochondria in soy bean
(Glycine max L. var Bragg) seeds during early stage of germination: Indian J. Exp.