The genus Nabidomiris (Hemiptera: Heteroptera: Miridae): review of the species and description of a new species from South Africa
Full text
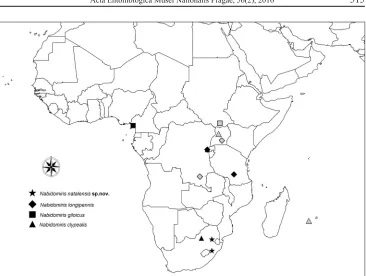
Figure
Related documents
• The development of a model named the image based feature space (IBFS) model for linking image regions or segments with text labels, as well as for automatic image
National Conference on Technical Vocational Education, Training and Skills Development: A Roadmap for Empowerment (Dec. 2008): Ministry of Human Resource Development, Department
Negative association Table I: Student engagement questionnaire factor analysis: statements most closely associated with item on consumerism This suggests that students who
Marie Laure Suites (Self Catering) Self Catering 14 Mr. Richard Naya Mahe Belombre 2516591 info@marielauresuites.com 61 Metcalfe Villas Self Catering 6 Ms Loulou Metcalfe
In 2005, the Montana legislature unanimously repealed Mon- tana Code Annotated section 28-2-722, which made employment contracts unenforceable by the employer beyond
sulla base della nostra esperien- za di cinque anni la presenza di un medico (spesso come co-conduttore) è molto utile per discutere ad esempio dei sintomi
• Follow up with your employer each reporting period to ensure your hours are reported on a regular basis?. • Discuss your progress with
Targeting the computation of NNMs for high-dimensional systems such as those encountered in industry, we propose to solve the set of PDEs using the finite