Long term efficacy and safety of antitumour necrosis factor alpha treatment in rhupus: an open label study of 15 patients
Full text
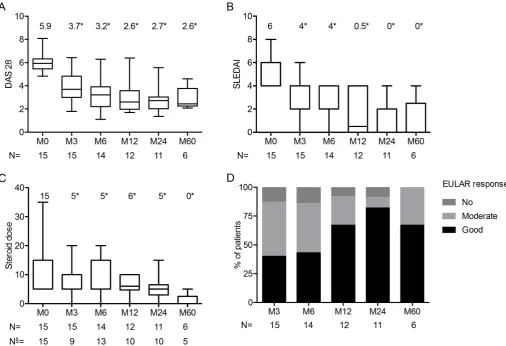
Figure



Related documents
The open-label extension study will evaluate the long-term safety of subcutaneously administered lanadelumab (over 12–18 months of exposure across both studies) and its
Efficacy and Safety of Asfotase Alfa in Infants and Young Children With Hypophosphatasia: A Phase 2 Open-Label Study..
This is a repository copy of Combined inhibition of tumour necrosis factor-alpha and interleukin-12/23 for long-standing, refractory psoriatic disease: a differential role for
As data on the efficacy and safety of long-term vonoprazan use in EE are limited, the Vonoprazan study In patients with eroSIve esophagitis to evaluate lONg-term safety (VISION)
This open-label extension study evaluated the long-term efficacy, safety and immunogenicity of LBEC0101 in Ko- rean patients with RA who were previously treated with ETN-RP or
SELECTED is an ongoing, single-arm, open-label exten- sion study to evaluate the long-term safety and efficacy of daclizumab 150 mg SC every 4 weeks for up to 6.5 years from
Efficacy and safety of anti- epidermal growth factor receptor agents for the treatment of oesophageal cancer: a systematic review and meta- analysis.. To view these files,
Long-term efficacy and safety of adalimumab in patients with moderate to severe psoriasis treated continuously over 3 years: results from an open-label extension study for patients