organic papers
Acta Cryst.(2006). E62, o2099–o2101 doi:10.1107/S1600536806015029 Gainsfordet al. C
34H38O8P22.5H2O
o2099
Acta Crystallographica Section EStructure Reports
Online
ISSN 1600-5368
1
D-1,2,5,6-Tetra-
O
-methyl-3,4-di-
O
-phosphinoyl-chiro
-inositol 2.5-hydrate
Graeme J. Gainsford,* Cornelis Lensink and Andrew Falshaw
Industrial Research Limited, PO Box 31-310, Lower Hutt, New Zealand
Correspondence e-mail: g.gainsford@irl.cri.nz
Key indicators
Single-crystal X-ray study T= 168 K
Mean(C–C) = 0.018 A˚ Rfactor = 0.066 wRfactor = 0.186
Data-to-parameter ratio = 10.7
For details of how these key indicators were automatically derived from the article, see http://journals.iucr.org/e.
Received 21 April 2006 Accepted 25 April 2006
#2006 International Union of Crystallography All rights reserved
In the title compound, C34H38O8P22.5H2O, intermolecular
interactions consist of strong O—H(water) O hydrogen bonds, as well as C—H O interactions.
Comment
The title compound, (I), was prepared in research aimed at generating new hydrogenation catalyst ligands from inositols (Falshawet al., 1999, 2006; Gainsfordet al., 2000).
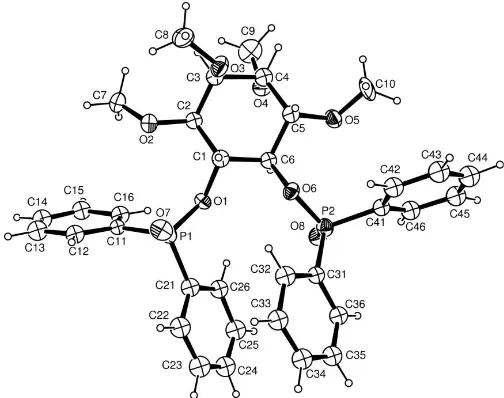
The asymmetric unit contains the independent molecule shown in Fig. 1 and three water molecules, one of which (O3W) is on a twofold rotation axis. Based on four units per cell, the composition is 2C34H38O8P25H2O; one of the water
[image:1.610.205.457.524.723.2]H atoms was not located (or refined). The absolute config-uration was known from the synthesis and is confirmed here by the anomalous dispersion effects.
Figure 1
The inositol ring adopts the chair form, with Cremer & Pople (1975) puckering parameters Q = 0.569 (12) A˚ , = 3.3 (12) and ’ = 91 (17). There are few related diphenyl-phosphinate structures reported [CSD (Version 5.27; Allen, 2002) refcodes CESJIT (Mazhar-ul-Haque et al., 1984), HANBUU (Griceet al., 2004), HOFLOD (Falshawet al., 1999) and VANQUW (Francio et al., 1998)]. No significant devia-tions in geometry are found from the ranges reported.
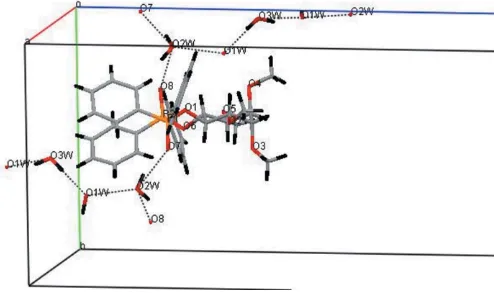
Crystal packing is dominated by linking hydrogen bonds utilizing the water molecules via two O(inositol), two O—H(water) O(water), two P O H(water) and weaker C—H O types (most are shown in Table 2). Fig. 2 illustrates how the chains of water molecules link the organic molecules running approximately along thec
axis. Note that the (expected) interaction O2W O1W
[2.774 (10) A˚ ] is shown in Fig. 2 but not in Table 2 as the H atom on O1Wwas not located.
Experimental
Crystals of (I) settled out of a solution of 1d-3,4-bis(o
-diphenyl-phosphino)-1,2,5,6-tetra-O-methyl-chiro-inositol (Falshaw et al., 2006) in CDCl3after exposure to air for several days.
Crystal data
C34H38O8P22.5H2O
Mr= 680.62
Tetragonal,P41212
a= 13.115 (5) A˚ c= 39.441 (15) A˚ V= 6784 (4) A˚3
Z= 8
Dx= 1.333 Mg m
3
MoKradiation = 0.19 mm1
T= 168 (2) K Plate, colourless 0.400.370.19 mm
Data collection
Siemens SMART CCD area-detector diffractometer ’and!scans
Absorption correction: multi-scan
(SADABS; Blessing, 1995;
Sheldrick, 1996)
22268 measured reflections 4694 independent reflections 1666 reflections withI> 2(I) Rint= 0.245
max= 26.1
Refinement
Refinement onF2 R[F2> 2(F2)] = 0.066
wR(F2) = 0.186 S= 0.85 4694 reflections 437 parameters
H atoms treated by a mixture of independent and constrained refinement
w= 1/[2
(Fo2) + (0.0828P)2]
whereP= (Fo2+ 2Fc2)/3
(/)max< 0.001 max= 0.36 e A˚
3 min=0.31 e A˚
3
Extinction correction:SHELXL97 Extinction coefficient: 0.061 (3) Absolute structure: Flack (1983),
[image:2.610.47.294.74.223.2]783 Friedel pairs Flack parameter: 0.0 (2)
Table 1
Selected geometric parameters (A˚ ,).
P1—O7 1.478 (6)
P1—O1 1.577 (6)
P2—O8 1.485 (6)
P2—O6 1.576 (6)
P2—C31 1.753 (10)
P2—C41 1.769 (10)
O7—P1—O1 116.4 (4)
O7—P1—C11 112.4 (5)
O1—P1—C11 105.1 (5)
O7—P1—C21 113.9 (5)
C1—O1—P1 125.3 (6)
C6—O6—P2 121.5 (6)
Table 2
Hydrogen-bond geometry (A˚ ,).
D—H A D—H H A D A D—H A
O1W—H1W1 O5i
0.87 (6) 2.29 (6) 3.123 (8) 160 (11) O1W—H1W1 O4i
0.87 (6) 2.55 (12) 3.136 (9) 126 (9) O2W—H2W1 O7 0.87 (7) 1.95 (9) 2.732 (10) 149 (9) O2W—H2W2 O8i
0.90 (8) 1.91 (8) 2.678 (10) 142 (7) O3W—H3W O1Wii
0.91 (12) 1.92 (12) 2.791 (10) 160 (12)
C23—H23 O3Wiii 0.95 2.52 3.385 (19) 152
C44—H44 O2Wiv
0.95 2.47 3.406 (15) 168
Symmetry codes: (i) xþ1 2;yþ
1 2;zþ
1
4; (ii) yþ 1 2;x
1 2;zþ
1 4; (iii)
yþ1 2;xþ
1 2;z
1
4; (iv)x1;y;z.
H atoms on C atoms were constrained to their expected geome-tries with C—H = 0.95–1.0 A˚ . Only one of the two H atoms on water atom O1Wcould be located in difference maps; the water H atoms on O2Wand O1Wwere restrained to O—H = 0.85 (3) A˚ and H H = 1.3 (1) A˚ . For all H atoms, Uiso(H) = 1.2Ueq(parent atom).
Exam-ination of the full sphere of collected data showed that a section of data taken late in the collection was badly measured, probably from crystal shifting (from consideration of equivalent reflections and very high statistical errors). These data were the major contributors to the high value forRint.
Data collection:SMART(Siemens, 1996); cell refinement:SAINT
(Siemens, 1996); data reduction:SAINT; program(s) used to solve structure:SHELXS97(Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics:
ORTEP-3(Farrugia, 1997); software used to prepare material for publication:SHELXL97andPLATON(Spek, 2003).
We thank Professor Ward T. Robinson & Dr J. Wikaira of the University of Canterbury, New Zealand, for their assis-tance.
References
Figure 2
Blessing, R. H. (1995).Acta Cryst.A51, 33–38.
Bruno, I. J., Cole, J. C., Edgington, P. R., Kessler, M., Macrae, C. F., McCabe, P., Pearson, J. & Taylor, R. (2002).Acta Cryst.B58, 389–397.
Cremer, D. & Pople, J. A. (1975).J. Am. Chem. Soc.97, 1354–1358. Falshaw, A., Gainsford, G. J. & Lensink, C. (1999).Acta Cryst.C55, 1353–
1355.
Falshaw, A., Gainsford, G. J., Lensink, C., Slade, A. T. & Wright, L. J. (2006).
Polyhedron. Submitted.
Farrugia, L. J. (1997).J. Appl. Cryst.30, 565. Flack, H. D. (1983).Acta Cryst.A39, 876–881.
Francio, G., Arena, C. G., Panzalorto, M., Bruno, G. & Faraone, F. (1998).
Inorg. Chim. Acta,277, 119–126.
Gainsford, G. J., Lensink, C., Hart, J. B. & Falshaw, A. (2000).Acta Cryst.C56, 1396–1398.
Grice, I. D., Jenkins, I. D., Busfield, W. K., Byriel, K. A. & Kennard, C. H. L. (2004).Acta Cryst.E60, o2384–o2385.
Ul-Haque, M., Ahmed, J. & Horne, W. (1984).J. Crystallogr. Spectrosc. Res.
14, 169–177.
Sheldrick, G. M. (1996).SADABS. University of Go¨ttingen, Germany. Sheldrick, G. M. (1997). SHELXL97 and SHELXS97. University of
Go¨ttingen, Germany.
Siemens (1996).SMARTandSAINT. Versions 4.0. Siemens Analytical X-ray Instruments Inc., Madison, Wisconsin, USA.
Spek, A. L. (2003).J. Appl. Cryst.36, 7–13.
organic papers
Acta Cryst.(2006). E62, o2099–o2101 Gainsfordet al. C
supporting information
Acta Cryst. (2006). E62, o2099–o2101 [https://doi.org/10.1107/S1600536806015029]
1
D-1,2,5,6-Tetra-
O
-methyl-3,4-di-
O
-phosphinoyl-
chiro
-inositol 2.5-hydrate
Graeme J. Gainsford, Cornelis Lensink and Andrew Falshaw
1D-1,2,5,6-Tetra-O-methyl-3,4-di-O-phosphinoyl-chiro-inositol 2.5-hydrate
Crystal data
C34H38O8P2·2.5H2O Mr = 680.62 Tetragonal, P41212
Hall symbol: P4abw 2nw
a = 13.115 (5) Å
c = 39.441 (15) Å
V = 6784 (4) Å3
Z = 8
F(000) = 2888
Dx = 1.333 Mg m−3
Mo Kα radiation, λ = 0.71073 Å
Cell parameters from 6000 reflections
θ = 2.8–25.4°
µ = 0.19 mm−1
T = 168 K
Plate, colourless 0.40 × 0.37 × 0.19 mm
Data collection
Siemens SMART CCD area-detector diffractometer
Radiation source: fine-focus sealed tube Graphite monochromator
Detector resolution: 8.192 pixels mm-1
φ and ω scans
Absorption correction: multi-scan
(SADABS; Blessing, 1995; Sheldrick, 1996)
Tmin = 0.629, Tmax = 0.965
22268 measured reflections 4694 independent reflections 1666 reflections with I > 2σ(I)
Rint = 0.245
θmax = 26.1°, θmin = 2.2°
h = −16→13
k = −15→14
l = −34→35
Refinement
Refinement on F2
Least-squares matrix: full
R[F2 > 2σ(F2)] = 0.066 wR(F2) = 0.186
S = 0.85
4694 reflections 437 parameters 4 restraints
Primary atom site location: structure-invariant direct methods
Secondary atom site location: difference Fourier map
Hydrogen site location: inferred from neighbouring sites
H atoms treated by a mixture of independent and constrained refinement
w = 1/[σ2(Fo2) + (0.0828P)2]
where P = (Fo2 + 2Fc2)/3
(Δ/σ)max < 0.001
Δρmax = 0.36 e Å−3
Δρmin = −0.31 e Å−3
Extinction correction: SHELXL97, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4
Extinction coefficient: 0.061 (3)
Absolute structure: Flack (1983), 783 Friedel pairs
supporting information
sup-2
Acta Cryst. (2006). E62, o2099–o2101 Special details
Experimental. Crystal decay was monitored by repeating the initial 10 frames at the end of the data collection and analyzing duplicate reflections. The standard 0.8 mm diameter collimator was used.
Examination of the full sphere of collected data showed that a section of data taken late in the collection (with 28=<l<40) was badly measured probably from crystal shifting (from consideration of equivalent reflections & VERY high statistical
errors). This data was the major contributor to the high R(int) value of 0.44. Removal of this block of data gave an R(int)
of 0.25. It was also noted at the time that an excessive buildup of ice occurred (a fault in the gas supply).
Apart from one low angle reflection (0,1,2), 11 other outlier relections were identified in the final Fo/Fc table statistics
with Fo >> Fc by factors of 1.5–2.0. In these cases, equivalent reflections for these data generally agreed indicating either
an ice scattering effect, or possibly a partial crystal fragment. With these reflections excluded, anisotropic thermal
parameter refinement was stable, even though the change in R1 factor is only from 0.0703 (1676 data) to 0.0665 (1666).
The outlier data are: (0,1,2); (0,2,0,); (0,2,1); (1,1,12);(0,9,19); (-1,3,4); (1,3,4); (0,1,3); (-1,3,2); (1,3,2); (1,1,9) & (2,3,0).
One of the water H atoms could not be refined. It is probable that some data is still affected by overlap; however (apart
from improving the R factors), the chosen conservative ratio was considered a reasonable if rather arbitrary compromise.
The full dataset is available from the authors on request.
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes.
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2,
conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used
only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2
are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger.
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
x y z Uiso*/Ueq
P1 0.4353 (2) 0.3997 (2) 0.15576 (9) 0.0595 (10)
P2 0.0957 (2) 0.4327 (2) 0.12525 (8) 0.0588 (10)
O1 0.3315 (4) 0.3670 (4) 0.17279 (16) 0.0534 (19)
O2 0.4008 (5) 0.4088 (5) 0.2389 (2) 0.072 (2)
O3 0.2345 (5) 0.5383 (6) 0.25888 (18) 0.074 (2)
O4 0.1025 (5) 0.3019 (6) 0.24559 (19) 0.066 (2)
O5 −0.0034 (5) 0.4226 (5) 0.20206 (17) 0.066 (2)
O6 0.1539 (4) 0.4722 (4) 0.15765 (18) 0.0536 (19)
O7 0.4622 (5) 0.5090 (5) 0.15799 (18) 0.069 (2)
O8 0.0903 (5) 0.3202 (5) 0.12167 (16) 0.066 (2)
O1W 0.5591 (6) 0.6912 (5) 0.0500 (2) 0.091 (3)
H1W1 0.543 (9) 0.753 (4) 0.044 (3) 0.109*
O2W 0.5387 (6) 0.6642 (6) 0.1194 (2) 0.086 (3)
H2W1 0.500 (7) 0.613 (4) 0.125 (3) 0.103*
H2W2 0.504 (7) 0.710 (5) 0.132 (2) 0.103*
O3W −0.0416 (7) 0.0416 (7) 0.2500 0.108 (5)
H3W −0.084 (9) 0.062 (9) 0.267 (3) 0.130*
C1 0.2743 (7) 0.4274 (8) 0.1974 (3) 0.057 (3)
H1 0.2925 0.5013 0.1959 0.068*
C2 0.2969 (8) 0.3843 (8) 0.2323 (3) 0.059 (3)
H2 0.2878 0.3086 0.2322 0.071*
H3 0.2448 0.4017 0.2809 0.069*
C4 0.1186 (8) 0.4095 (9) 0.2485 (3) 0.065 (3)
H4 0.0716 0.4393 0.2658 0.078*
C5 0.0940 (8) 0.4498 (7) 0.2142 (3) 0.053 (3)
H5 0.0992 0.5259 0.2147 0.064*
C6 0.1664 (7) 0.4092 (8) 0.1871 (3) 0.056 (3)
H6 0.1534 0.3357 0.1820 0.068*
C7 0.4449 (8) 0.3473 (10) 0.2632 (3) 0.097 (4)
H7A 0.4063 0.3530 0.2844 0.116*
H7B 0.5154 0.3690 0.2671 0.116*
H7C 0.4441 0.2764 0.2555 0.116*
C8 0.3005 (8) 0.5782 (9) 0.2843 (3) 0.095 (4)
H8A 0.2747 0.5591 0.3068 0.114*
H8B 0.3029 0.6526 0.2825 0.114*
H8C 0.3691 0.5502 0.2813 0.114*
C9 0.0942 (9) 0.2570 (8) 0.2771 (4) 0.095 (4)
H9A 0.1593 0.2628 0.2891 0.114*
H9B 0.0765 0.1849 0.2744 0.114*
H9C 0.0407 0.2915 0.2902 0.114*
C10 −0.0829 (8) 0.4886 (9) 0.2134 (3) 0.096 (4)
H10A −0.1018 0.4707 0.2367 0.115*
H10B −0.1424 0.4810 0.1986 0.115*
H10C −0.0591 0.5594 0.2127 0.115*
C11 0.5288 (8) 0.3200 (7) 0.1744 (3) 0.050 (3)
C12 0.6278 (10) 0.3495 (8) 0.1776 (3) 0.069 (3)
H12 0.6464 0.4161 0.1704 0.083*
C13 0.7012 (10) 0.2878 (13) 0.1905 (3) 0.090 (4)
H13 0.7696 0.3109 0.1924 0.108*
C14 0.6752 (12) 0.1927 (11) 0.2007 (3) 0.078 (4)
H14 0.7259 0.1484 0.2096 0.094*
C15 0.5769 (11) 0.1603 (10) 0.1983 (3) 0.077 (4)
H15 0.5592 0.0939 0.2059 0.092*
C16 0.5026 (8) 0.2233 (9) 0.1849 (2) 0.060 (3)
H16 0.4342 0.2002 0.1830 0.073*
C21 0.4182 (10) 0.3551 (9) 0.1137 (3) 0.065 (4)
C22 0.4731 (10) 0.3995 (11) 0.0880 (5) 0.092 (4)
H22 0.5213 0.4519 0.0924 0.111*
C23 0.4552 (12) 0.3644 (12) 0.0554 (5) 0.093 (5)
H23 0.4931 0.3937 0.0374 0.111*
C24 0.3861 (13) 0.2900 (13) 0.0479 (4) 0.087 (5)
H24 0.3772 0.2674 0.0252 0.104*
C25 0.3322 (11) 0.2504 (11) 0.0725 (5) 0.089 (5)
H25 0.2833 0.1994 0.0673 0.107*
C26 0.3444 (9) 0.2808 (9) 0.1065 (4) 0.064 (3)
H26 0.3040 0.2519 0.1240 0.077*
C31 0.1639 (8) 0.4929 (7) 0.0927 (4) 0.054 (3)
C32 0.2448 (9) 0.5589 (8) 0.0984 (4) 0.075 (4)
supporting information
sup-4
Acta Cryst. (2006). E62, o2099–o2101
C33 0.2979 (9) 0.6008 (10) 0.0714 (5) 0.085 (5)
H33 0.3550 0.6436 0.0755 0.102*
C34 0.2683 (10) 0.5806 (11) 0.0392 (4) 0.093 (5)
H34 0.3026 0.6118 0.0207 0.111*
C35 0.1900 (11) 0.5164 (11) 0.0334 (3) 0.075 (4)
H35 0.1705 0.5035 0.0106 0.090*
C36 0.1369 (9) 0.4685 (9) 0.0591 (4) 0.069 (4)
H36 0.0842 0.4210 0.0543 0.083*
C41 −0.0279 (8) 0.4867 (9) 0.1261 (3) 0.061 (3)
C42 −0.0417 (8) 0.5879 (9) 0.1362 (3) 0.071 (3)
H42 0.0150 0.6272 0.1436 0.086*
C43 −0.1351 (10) 0.6296 (10) 0.1355 (3) 0.088 (4)
H43 −0.1432 0.7000 0.1405 0.105*
C44 −0.2202 (9) 0.5709 (14) 0.1276 (3) 0.092 (5)
H44 −0.2866 0.5996 0.1288 0.111*
C45 −0.2072 (10) 0.4715 (12) 0.1180 (3) 0.084 (4)
H45 −0.2640 0.4311 0.1114 0.101*
C46 −0.1079 (9) 0.4301 (9) 0.1181 (2) 0.070 (3)
H46 −0.0983 0.3605 0.1122 0.084*
Atomic displacement parameters (Å2)
U11 U22 U33 U12 U13 U23
P1 0.055 (2) 0.053 (2) 0.070 (3) 0.0045 (15) 0.0070 (18) 0.0010 (18)
P2 0.0490 (18) 0.059 (2) 0.069 (3) −0.0059 (15) 0.0008 (18) 0.0051 (19)
O1 0.048 (4) 0.053 (4) 0.059 (5) 0.000 (3) 0.010 (4) −0.003 (4)
O2 0.051 (5) 0.094 (6) 0.072 (6) 0.008 (5) −0.008 (4) −0.001 (5)
O3 0.074 (5) 0.071 (6) 0.076 (6) 0.004 (4) −0.017 (5) −0.014 (5)
O4 0.086 (6) 0.068 (6) 0.045 (6) 0.002 (4) 0.000 (5) 0.010 (4)
O5 0.052 (5) 0.075 (5) 0.071 (6) 0.001 (4) 0.010 (4) 0.001 (4)
O6 0.055 (4) 0.049 (4) 0.057 (6) −0.001 (3) −0.012 (4) −0.011 (4)
O7 0.092 (6) 0.037 (4) 0.080 (6) 0.001 (4) 0.003 (4) −0.004 (4)
O8 0.078 (5) 0.049 (5) 0.071 (6) 0.002 (4) −0.001 (4) 0.002 (4)
O1W 0.076 (6) 0.085 (6) 0.112 (7) 0.007 (5) −0.006 (5) 0.011 (6)
O2W 0.078 (6) 0.059 (5) 0.121 (8) 0.009 (4) 0.025 (5) 0.002 (5)
O3W 0.114 (7) 0.114 (7) 0.098 (13) 0.032 (8) 0.010 (7) 0.010 (7)
C1 0.057 (8) 0.064 (8) 0.049 (10) 0.015 (6) 0.000 (7) −0.013 (7)
C2 0.056 (8) 0.072 (8) 0.050 (10) −0.001 (6) 0.004 (7) −0.001 (7)
C3 0.044 (7) 0.088 (10) 0.040 (9) 0.010 (7) −0.008 (6) 0.003 (7)
C4 0.076 (9) 0.060 (9) 0.061 (11) 0.004 (7) −0.009 (7) 0.002 (7)
C5 0.047 (7) 0.056 (7) 0.058 (10) 0.001 (6) 0.002 (7) −0.017 (7)
C6 0.046 (7) 0.048 (7) 0.075 (10) 0.014 (6) 0.013 (7) 0.017 (7)
C7 0.065 (8) 0.141 (12) 0.084 (11) 0.028 (8) −0.026 (8) −0.015 (10)
C8 0.078 (8) 0.123 (11) 0.085 (10) −0.027 (8) −0.008 (8) −0.056 (9)
C9 0.098 (11) 0.081 (9) 0.106 (13) 0.003 (8) 0.001 (9) 0.008 (9)
C10 0.068 (8) 0.119 (11) 0.101 (11) 0.036 (9) 0.001 (8) −0.034 (9)
C11 0.051 (8) 0.027 (7) 0.074 (9) 0.009 (5) −0.004 (6) −0.002 (6)
C13 0.058 (9) 0.114 (13) 0.097 (12) 0.018 (10) −0.015 (8) −0.033 (10)
C14 0.101 (13) 0.082 (11) 0.053 (10) 0.036 (9) −0.008 (8) 0.007 (8)
C15 0.067 (9) 0.084 (9) 0.079 (11) 0.011 (9) −0.004 (8) −0.005 (8)
C16 0.056 (8) 0.071 (9) 0.055 (9) 0.018 (7) 0.004 (7) −0.009 (7)
C21 0.073 (9) 0.053 (8) 0.070 (12) 0.006 (7) 0.007 (8) −0.006 (8)
C22 0.091 (10) 0.102 (11) 0.085 (14) 0.009 (9) 0.025 (11) 0.015 (12)
C23 0.100 (12) 0.092 (12) 0.086 (17) 0.023 (9) 0.004 (11) 0.017 (10)
C24 0.097 (13) 0.119 (14) 0.044 (13) 0.047 (10) 0.002 (11) −0.008 (11)
C25 0.084 (11) 0.130 (13) 0.054 (14) 0.010 (9) −0.013 (10) −0.034 (11)
C26 0.065 (9) 0.066 (8) 0.062 (13) 0.018 (7) −0.012 (7) 0.005 (7)
C31 0.048 (7) 0.041 (7) 0.073 (11) 0.000 (5) 0.017 (7) 0.012 (7)
C32 0.063 (8) 0.052 (8) 0.110 (13) 0.003 (7) −0.013 (9) −0.006 (8)
C33 0.056 (8) 0.096 (10) 0.103 (14) −0.020 (7) 0.004 (10) 0.039 (11)
C34 0.061 (10) 0.111 (13) 0.106 (16) 0.016 (10) 0.018 (10) 0.032 (11)
C35 0.081 (10) 0.091 (10) 0.052 (11) 0.020 (8) −0.002 (9) 0.006 (9)
C36 0.065 (8) 0.081 (9) 0.062 (12) 0.017 (7) −0.010 (9) −0.011 (9)
C41 0.045 (8) 0.060 (8) 0.077 (10) 0.007 (6) 0.005 (7) −0.001 (7)
C42 0.053 (8) 0.083 (10) 0.078 (10) 0.012 (7) 0.006 (6) 0.018 (8)
C43 0.072 (10) 0.100 (11) 0.090 (12) 0.024 (9) 0.016 (8) 0.038 (8)
C44 0.053 (9) 0.160 (16) 0.063 (11) −0.007 (11) 0.000 (7) 0.064 (11)
C45 0.078 (11) 0.118 (12) 0.057 (10) 0.004 (9) 0.012 (7) 0.025 (9)
C46 0.066 (9) 0.099 (9) 0.044 (9) −0.015 (9) −0.001 (7) 0.000 (7)
Geometric parameters (Å, º)
P1—O7 1.478 (6) C10—H10C 0.9800
P1—O1 1.577 (6) C11—C12 1.360 (13)
P1—C11 1.771 (9) C11—C16 1.378 (12)
P1—C21 1.773 (12) C12—C13 1.357 (14)
P2—O8 1.485 (6) C12—H12 0.9500
P2—O6 1.576 (6) C13—C14 1.353 (16)
P2—C31 1.753 (10) C13—H13 0.9500
P2—C41 1.769 (10) C14—C15 1.361 (14)
O1—C1 1.460 (10) C14—H14 0.9500
O2—C7 1.381 (11) C15—C16 1.383 (13)
O2—C2 1.424 (11) C15—H15 0.9500
O3—C3 1.415 (11) C16—H16 0.9500
O3—C8 1.425 (11) C21—C22 1.371 (14)
O4—C9 1.380 (12) C21—C26 1.402 (14)
O4—C4 1.432 (11) C22—C23 1.387 (15)
O5—C5 1.410 (10) C22—H22 0.9500
O5—C10 1.426 (10) C23—C24 1.365 (17)
O6—C6 1.436 (10) C23—H23 0.9500
O1W—H1W1 0.86 (3) C24—C25 1.310 (16)
O2W—H2W1 0.87 (7) C24—H24 0.9500
O2W—H2W2 0.90 (8) C25—C26 1.407 (15)
O3W—H3W 0.92 (11) C25—H25 0.9500
supporting information
sup-6
Acta Cryst. (2006). E62, o2099–o2101
C1—C2 1.518 (12) C31—C32 1.388 (13)
C1—H1 1.0000 C31—C36 1.408 (13)
C2—C3 1.474 (12) C32—C33 1.386 (14)
C2—H2 1.0000 C32—H32 0.9500
C3—C4 1.526 (12) C33—C34 1.354 (15)
C3—H3 1.0000 C33—H33 0.9500
C4—C5 1.490 (12) C34—C35 1.347 (16)
C4—H4 1.0000 C34—H34 0.9500
C5—C6 1.524 (12) C35—C36 1.380 (14)
C5—H5 1.0000 C35—H35 0.9500
C6—H6 1.0000 C36—H36 0.9500
C7—H7A 0.9800 C41—C46 1.324 (12)
C7—H7B 0.9800 C41—C42 1.398 (13)
C7—H7C 0.9800 C42—C43 1.341 (13)
C8—H8A 0.9800 C42—H42 0.9500
C8—H8B 0.9800 C43—C44 1.392 (15)
C8—H8C 0.9800 C43—H43 0.9500
C9—H9A 0.9800 C44—C45 1.367 (15)
C9—H9B 0.9800 C44—H44 0.9500
C9—H9C 0.9800 C45—C46 1.411 (14)
C10—H10A 0.9800 C45—H45 0.9500
C10—H10B 0.9800 C46—H46 0.9500
O7—P1—O1 116.4 (4) O5—C10—H10C 109.5
O7—P1—C11 112.4 (5) H10A—C10—H10C 109.5
O1—P1—C11 105.1 (5) H10B—C10—H10C 109.5
O7—P1—C21 113.9 (5) C12—C11—C16 118.2 (10)
O1—P1—C21 101.5 (6) C12—C11—P1 122.2 (10)
C11—P1—C21 106.3 (5) C16—C11—P1 119.6 (9)
O8—P2—O6 115.3 (4) C13—C12—C11 122.8 (11)
O8—P2—C31 113.7 (5) C13—C12—H12 118.6
O6—P2—C31 101.5 (6) C11—C12—H12 118.6
O8—P2—C41 110.8 (5) C14—C13—C12 118.9 (13)
O6—P2—C41 107.3 (5) C14—C13—H13 120.6
C31—P2—C41 107.5 (5) C12—C13—H13 120.6
C1—O1—P1 125.3 (6) C13—C14—C15 120.3 (13)
C7—O2—C2 113.3 (9) C13—C14—H14 119.8
C3—O3—C8 114.4 (9) C15—C14—H14 119.8
C9—O4—C4 111.1 (9) C14—C15—C16 120.5 (12)
C5—O5—C10 113.7 (7) C14—C15—H15 119.7
C6—O6—P2 121.5 (6) C16—C15—H15 119.7
H2W1—O2W—H2W2 95 (8) C11—C16—C15 119.3 (10)
O1—C1—C6 102.7 (8) C11—C16—H16 120.4
O1—C1—C2 107.5 (8) C15—C16—H16 120.4
C6—C1—C2 111.8 (9) C22—C21—C26 120.5 (12)
O1—C1—H1 111.5 C22—C21—P1 119.0 (13)
C6—C1—H1 111.5 C26—C21—P1 120.4 (12)
O2—C2—C3 111.0 (9) C21—C22—H22 121.5
O2—C2—C1 105.5 (9) C23—C22—H22 121.5
C3—C2—C1 110.6 (9) C24—C23—C22 123.5 (16)
O2—C2—H2 109.9 C24—C23—H23 118.2
C3—C2—H2 109.9 C22—C23—H23 118.2
C1—C2—H2 109.9 C25—C24—C23 118.7 (16)
O3—C3—C2 113.6 (9) C25—C24—H24 120.7
O3—C3—C4 103.4 (8) C23—C24—H24 120.7
C2—C3—C4 109.3 (9) C24—C25—C26 122.3 (15)
O3—C3—H3 110.1 C24—C25—H25 118.9
C2—C3—H3 110.1 C26—C25—H25 118.9
C4—C3—H3 110.1 C21—C26—C25 117.9 (12)
O4—C4—C5 104.2 (9) C21—C26—H26 121.0
O4—C4—C3 109.8 (9) C25—C26—H26 121.0
C5—C4—C3 111.3 (9) C32—C31—C36 119.2 (11)
O4—C4—H4 110.5 C32—C31—P2 123.5 (13)
C5—C4—H4 110.5 C36—C31—P2 117.3 (11)
C3—C4—H4 110.5 C33—C32—C31 120.3 (12)
O5—C5—C4 114.5 (9) C33—C32—H32 119.8
O5—C5—C6 103.9 (8) C31—C32—H32 119.8
C4—C5—C6 112.2 (9) C34—C33—C32 120.0 (13)
O5—C5—H5 108.7 C34—C33—H33 120.0
C4—C5—H5 108.7 C32—C33—H33 120.0
C6—C5—H5 108.7 C35—C34—C33 119.9 (14)
O6—C6—C1 103.7 (8) C35—C34—H34 120.0
O6—C6—C5 107.1 (8) C33—C34—H34 120.0
C1—C6—C5 110.2 (9) C34—C35—C36 123.1 (14)
O6—C6—H6 111.8 C34—C35—H35 118.5
C1—C6—H6 111.8 C36—C35—H35 118.5
C5—C6—H6 111.8 C35—C36—C31 117.3 (12)
O2—C7—H7A 109.5 C35—C36—H36 121.3
O2—C7—H7B 109.5 C31—C36—H36 121.3
H7A—C7—H7B 109.5 C46—C41—C42 119.9 (10)
O2—C7—H7C 109.5 C46—C41—P2 119.8 (10)
H7A—C7—H7C 109.5 C42—C41—P2 120.3 (9)
H7B—C7—H7C 109.5 C43—C42—C41 119.9 (11)
O3—C8—H8A 109.5 C43—C42—H42 120.0
O3—C8—H8B 109.5 C41—C42—H42 120.0
H8A—C8—H8B 109.5 C42—C43—C44 120.8 (13)
O3—C8—H8C 109.5 C42—C43—H43 119.6
H8A—C8—H8C 109.5 C44—C43—H43 119.6
H8B—C8—H8C 109.5 C45—C44—C43 119.3 (14)
O4—C9—H9A 109.5 C45—C44—H44 120.4
O4—C9—H9B 109.5 C43—C44—H44 120.4
H9A—C9—H9B 109.5 C44—C45—C46 118.8 (13)
O4—C9—H9C 109.5 C44—C45—H45 120.6
H9A—C9—H9C 109.5 C46—C45—H45 120.6
supporting information
sup-8
Acta Cryst. (2006). E62, o2099–o2101
O5—C10—H10A 109.5 C41—C46—H46 119.5
O5—C10—H10B 109.5 C45—C46—H46 119.5
H10A—C10—H10B 109.5
O7—P1—O1—C1 −16.7 (9) P1—C11—C12—C13 −177.3 (9)
C11—P1—O1—C1 108.5 (8) C11—C12—C13—C14 0.1 (18)
C21—P1—O1—C1 −140.9 (8) C12—C13—C14—C15 −0.7 (19)
O8—P2—O6—C6 −22.3 (8) C13—C14—C15—C16 1.1 (18)
C31—P2—O6—C6 −145.7 (7) C12—C11—C16—C15 0.2 (15)
C41—P2—O6—C6 101.7 (7) P1—C11—C16—C15 177.7 (8)
P1—O1—C1—C6 141.2 (7) C14—C15—C16—C11 −0.8 (16)
P1—O1—C1—C2 −100.7 (9) O7—P1—C21—C22 30.6 (11)
C7—O2—C2—C3 81.4 (11) O1—P1—C21—C22 156.5 (9)
C7—O2—C2—C1 −158.7 (9) C11—P1—C21—C22 −93.8 (10)
O1—C1—C2—O2 69.0 (9) O7—P1—C21—C26 −145.0 (8)
C6—C1—C2—O2 −179.0 (8) O1—P1—C21—C26 −19.1 (9)
O1—C1—C2—C3 −170.9 (8) C11—P1—C21—C26 90.6 (9)
C6—C1—C2—C3 −58.9 (11) C26—C21—C22—C23 −2.6 (17)
C8—O3—C3—C2 −97.2 (10) P1—C21—C22—C23 −178.2 (9)
C8—O3—C3—C4 144.5 (9) C21—C22—C23—C24 0.7 (19)
O2—C2—C3—O3 60.8 (12) C22—C23—C24—C25 1 (2)
C1—C2—C3—O3 −56.0 (11) C23—C24—C25—C26 −1 (2)
O2—C2—C3—C4 175.6 (9) C22—C21—C26—C25 2.8 (16)
C1—C2—C3—C4 58.9 (12) P1—C21—C26—C25 178.2 (8)
C9—O4—C4—C5 −163.1 (8) C24—C25—C26—C21 −0.9 (18)
C9—O4—C4—C3 77.6 (11) O8—P2—C31—C32 −126.1 (8)
O3—C3—C4—O4 178.3 (8) O6—P2—C31—C32 −1.7 (9)
C2—C3—C4—O4 57.0 (12) C41—P2—C31—C32 110.7 (9)
O3—C3—C4—C5 63.5 (11) O8—P2—C31—C36 51.0 (9)
C2—C3—C4—C5 −57.8 (12) O6—P2—C31—C36 175.4 (7)
C10—O5—C5—C4 84.6 (11) C41—P2—C31—C36 −72.1 (9)
C10—O5—C5—C6 −152.8 (9) C36—C31—C32—C33 0.4 (15)
O4—C4—C5—O5 54.6 (10) P2—C31—C32—C33 177.5 (9)
C3—C4—C5—O5 172.8 (9) C31—C32—C33—C34 2.7 (18)
O4—C4—C5—C6 −63.4 (10) C32—C33—C34—C35 −3 (2)
C3—C4—C5—C6 54.8 (12) C33—C34—C35—C36 −0.1 (19)
P2—O6—C6—C1 137.3 (7) C34—C35—C36—C31 3.1 (17)
P2—O6—C6—C5 −106.2 (8) C32—C31—C36—C35 −3.2 (14)
O1—C1—C6—O6 −76.7 (9) P2—C31—C36—C35 179.5 (8)
C2—C1—C6—O6 168.3 (8) O8—P2—C41—C46 −11.3 (11)
O1—C1—C6—C5 169.0 (8) O6—P2—C41—C46 −137.9 (9)
C2—C1—C6—C5 54.0 (12) C31—P2—C41—C46 113.6 (10)
O5—C5—C6—O6 71.0 (9) O8—P2—C41—C42 166.8 (8)
C4—C5—C6—O6 −164.9 (8) O6—P2—C41—C42 40.2 (10)
O5—C5—C6—C1 −176.9 (8) C31—P2—C41—C42 −68.3 (11)
C4—C5—C6—C1 −52.8 (12) C46—C41—C42—C43 −4.5 (17)
O7—P1—C11—C12 −24.5 (10) P2—C41—C42—C43 177.4 (8)
C21—P1—C11—C12 100.8 (10) C42—C43—C44—C45 −4.7 (19)
O7—P1—C11—C16 158.0 (8) C43—C44—C45—C46 3.0 (19)
O1—P1—C11—C16 30.4 (9) C42—C41—C46—C45 2.9 (17)
C21—P1—C11—C16 −76.7 (10) P2—C41—C46—C45 −179.0 (8)
C16—C11—C12—C13 0.2 (16) C44—C45—C46—C41 −2.2 (18)
Hydrogen-bond geometry (Å, º)
D—H···A D—H H···A D···A D—H···A
O1W—H1W1···O5i 0.87 (6) 2.29 (6) 3.123 (8) 160 (11)
O1W—H1W1···O4i 0.87 (6) 2.55 (12) 3.136 (9) 126 (9)
O2W—H2W1···O7 0.87 (7) 1.95 (9) 2.732 (10) 149 (9)
O2W—H2W2···O8i 0.90 (8) 1.91 (8) 2.678 (10) 142 (7)
O3W—H3W···O1Wii 0.91 (12) 1.92 (12) 2.791 (10) 160 (12)
C23—H23···O3Wiii 0.95 2.52 3.385 (19) 152
C44—H44···O2Wiv 0.95 2.47 3.406 (15) 168