Acta Crystallographica Section E
Structure Reports
Online
ISSN 1600-5368
(+)-(4a
R
,9a
S
)-
O
-(4-Bromobenzoyl)-
N
-(3-methyl-anthracen-9-ylidene)hydroxylamine
Shigeru Ohba,a* Naoyuki Ishida,b Dai-ichiro Kato,bKenji Miyamotoband Hiromichi Ohtab
aDepartment of Chemistry, Faculty of Letters,
Keio University, Hiyoshi 4-1-1, Kohoku-ku, Yokohama 223-8521, Japan, andbDepartment
of Biosciences and Informatics, Faculty of Science and Technology, Keio University, Hiyoshi 3-14-1, Kohoku-ku, Yokohama 223-8522, Japan
Correspondence e-mail: ohba@flet.keio.ac.jp
Key indicators
Single-crystal X-ray study
T= 298 K
Mean(C–C) = 0.035 A˚
Rfactor = 0.063
wRfactor = 0.231 Data-to-parameter ratio = 7.6
For details of how these key indicators were automatically derived from the article, see http://journals.iucr.org/e.
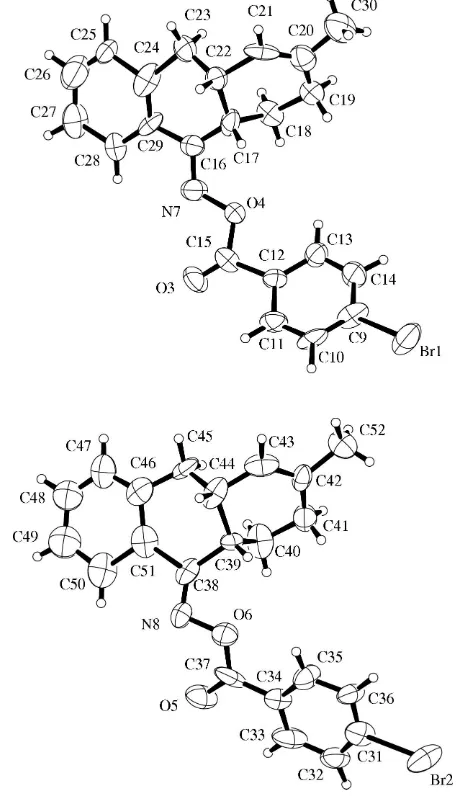
In the title compound, C22H20BrNO2, there are two indepen-dent molecules having the same absolute configuration, although the crystal structure has a pseudo-center of symmetry. The oxime group is anti to the benzene ring of the 6–6–6 fused ring system.
Comment
In the research developing an entirely new asymmetric reac-tion, optically active 8.9-benzobicyclo[4.4.0]-2-decen-7-one, (1), is one of the expected target molecules of the reaction. In order to determine the absolute configuration of (1), one enantiomer was converted to the corresponding oxime (2), to which thep-bromobenzoyl group was introduced. The chiral C atoms of (1) are retained in the title compound, (3). The X-ray structure analysis of (3) has been carried out to determine the absolute configuration based on the strong anomalous scat-tering of Br.
In the crystal structure of (3), there are two independent molecules (Fig. 1). For both molecules, the ring junction iscis, and the oxime group is antito the benzene ring of the ring system. Fig. 2 shows the crystal structure. The two molecules containing Br1 and Br2 are related by a pseudo-center of symmetry at nearly (1
4, 0, 1
4), except for the chiral atoms C17/ C22 and C39/C44.
Experimental
A racemic mixture of (1R,6S)-(1) and its enantiomer was prepared starting fromo-bromophenylacetic acid. Details of the synthesis will be published elsewhere. The optical resolution of (1) was carried out
via preparative high-pressure liquid chromatography using a chiral AD column (Daicel). One of the isolated optical isomers was converted to the corresponding oxime (2) by hydroxylamine. Compound (2) was then acylated withp-bromobenzoyl chloride to give (+)-(3), which was purified by repeated recrystallization from a methanol/diethyl ether mixture [yield 65% from (1)]. Crystals of (+)-(3) suitable for X-ray study were grown from a diethyl ether solution by slow evaporation [m.p. 353 K (decomposition)]. The
Crystal data
C22H20BrNO2
Mr= 410.31 Monoclinic,P21
a= 10.536 (2) A˚
b= 19.225 (5) A˚
c= 9.500 (2) A˚ = 97.67 (2) V= 1907.1 (7) A˚3
Z= 4
Dx= 1.429 Mg m
3 MoKradiation Cell parameters from 25
reflections = 10.0–11.6
= 2.18 mm1
T= 298 K Prism, colorless 0.500.450.30 mm
Data collection
Rigaku AFC-7Rdiffractometer !–2scans
Absorption correction: integration (Higashi, 1999)
Tmin= 0.389,Tmax= 0.552 3670 measured reflections 3466 independent reflections 1224 reflections withI> 2(I)
Rint= 0.098
max= 25.0
h=12!0
k= 0!22
l=11!11 3 standard reflections
every 150 reflections intensity decay: 1.4%
Refinement
Refinement onF2 R[F2> 2(F2)] = 0.063
wR(F2) = 0.231
S= 0.98 3466 reflections 459 parameters
H-atom parameters constrained
w= 1/[2 (Fo
2
) + (0.1051P)2 + 1.0533P]
whereP= (Fo2+ 2Fc2)/3 (/)max= 0.018
max= 0.24 e A˚
3 min=0.43 e A˚
3
Absolute structure: (Flack, 1983), no Friedel pairs
Flack parameter:0.02 (3)
Table 1
Selected torsion angles ().
O4—N7—C16—C17 2 (2) O6—N8—C38—C39 2 (2) C16—C17—C22—C23 51 (2)
C18—C17—C22—C21 35 (2) C38—C39—C44—C45 62 (2) C40—C39—C44—C43 47 (2)
Atoms C48 and C49 were refined isotropically to avoid abnormal deformation of the benzene ring from ideal geometry. The highUeq
values of C26 and C33, compared with those of their neighbors, and the large variation in the C—C distances of the benzene rings may be attributable to the large correlations in the atomic parameters because of the pseudo-centrosymmetry. All H atoms were positioned geometrically and refined as riding, with C—H distances of 0.95 A˚ and withUiso(H) = 1.2Ueq(C).
Data collection: WinAFC Diffractometer Control Software
(Rigaku, 1999); cell refinement: WinAFC Diffractometer Control Software; data reduction:TEXSAN(Molecular Structure Corpora-tion, 2001); program(s) used to solve structure:SIR92(Altomareet al., 1994) andDIRDIF94(Beurskenset al., 1994); program(s) used to refine structure:SHELXL97(Sheldrick, 1997); molecular graphics:
ORTEPII (Johnson, 1976); software used to prepare material for publication:TEXSAN.
References
Altomare, A., Cascarano, G., Giacovazzo, C., Guagliardi, A., Burla, M. C., Polidori, G. & Camalli, M. (1994).J. Appl. Cryst.27, 435.
Beurskens, P. T., Admiraal, G., Beurskens, G., Bosman, W. P., de Gelder, R., Israel, R. & Smits, J. M. M. (1994). The DIRDIF94 Program System. Technical Report of the Crystallography Laboratory, University of Nijmegen, The Netherlands.
Flack, H. D. (1983).Acta Cryst.A39, 876–881.
Higashi, T. (1999).ABSCOR. Rigaku Corporation, Tokyo, Japan.
Johnson, C. K. (1976).ORTEPII. Report ORNL-5138. Oak Ridge National Laboratory, Tennessee, USA.
Molecular Structure Corporation (2001).TEXSAN. Version 1.11. MSC, 9009 New Trails Drive, The Woodlands, TX 77381-5209, USA.
Rigaku (1999).WinAFC Diffractometer Control Software. Rigaku Corpora-tion, Tokyo, Japan.
Sheldrick, G. M. (1997).SHELXL97. University of Go¨ttingen, Germany.
organic papers
Acta Cryst.(2005). E61, o1620–o1621 Ohbaet al. C
22H20BrNO2
o1621
Figure 2
[image:2.610.325.553.77.475.2] [image:2.610.312.567.523.630.2]The projection of the crystal structure of (3) along theaaxis. H atoms have been omitted, and non-H atoms are drawn as small spheres for clarity.
Figure 1
supporting information
Acta Cryst. (2005). E61, o1620–o1621 [https://doi.org/10.1107/S1600536805013966]
(+)-(4a
R
,9a
S
)-
O
-(4-Bromobenzoyl)-
N
-(3-methylanthracen-9-ylidene)hydroxyl-amine
Shigeru Ohba, Naoyuki Ishida, Dai-ichiro Kato, Kenji Miyamoto and Hiromichi Ohta
(+)-(4aR,9aS)-O-(4-Bromobenzoyl)-N-(3-methylanthracen-9-ylidene)hydroxylamine
Crystal data
C22H20BrNO2 Mr = 410.31
Monoclinic, P21 a = 10.536 (2) Å
b = 19.225 (5) Å
c = 9.500 (2) Å
β = 97.67 (2)°
V = 1907.1 (7) Å3 Z = 4
F(000) = 840
Dx = 1.429 Mg m−3
Mo Kα radiation, λ = 0.71073 Å Cell parameters from 25 reflections
θ = 10.0–11.6°
µ = 2.18 mm−1 T = 298 K Prism, colorless 0.50 × 0.45 × 0.30 mm
Data collection
Rigaku AFC-7R diffractometer
ω–2θ scans
Absorption correction: integration (Higashi, 1999)
Tmin = 0.389, Tmax = 0.552
3670 measured reflections 3466 independent reflections
1224 reflections with I > 2σ(I)
Rint = 0.098 θmax = 25.0° h = −12→0
k = 0→22
l = −11→11
3 standard reflections every 150 reflections intensity decay: 1.4%
Refinement
Refinement on F2 R[F2 > 2σ(F2)] = 0.063 wR(F2) = 0.231 S = 0.98 3466 reflections 459 parameters
H-atom parameters constrained
w = 1/[σ2(F
o2) + (0.1051P)2 + 1.0533P]
where P = (Fo2 + 2Fc2)/3
(Δ/σ)max = 0.018
Δρmax = 0.24 e Å−3
Δρmin = −0.43 e Å−3
Absolute structure: (Flack, 1983), no Friedel pairs
Absolute structure parameter: −0.02 (3)
Special details
Refinement. Refinement using reflections with F2 > 0.0 σ(F2). The weighted R-factor (wR), goodness of fit (S) and R
-factor (gt) are based on F, with F set to zero for negative F. The threshold expression of F2 > 2.0 σ(F2) is used only for
supporting information
sup-2
Acta Cryst. (2005). E61, o1620–o1621
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
x y z Uiso*/Ueq
Br1 0.9823 (3) 0.2998 (2) 0.2312 (3) 0.150 (1)
Br2 −0.4885 (3) −0.2109 (2) 0.2557 (2) 0.141 (1)
O3 0.336 (1) 0.2528 (9) 0.222 (1) 0.106 (5)
O4 0.422 (1) 0.1793 (7) 0.389 (1) 0.071 (4)
O5 0.160 (1) −0.1554 (9) 0.315 (1) 0.118 (5)
O6 0.091 (1) −0.0899 (9) 0.123 (1) 0.092 (5)
N7 0.296 (2) 0.155 (1) 0.405 (2) 0.094 (6)
N8 0.219 (2) −0.069 (1) 0.125 (2) 0.081 (5)
C9 0.810 (2) 0.282 (1) 0.253 (2) 0.095 (8)
C10 0.713 (3) 0.302 (1) 0.156 (2) 0.101 (8)
C11 0.590 (2) 0.287 (1) 0.169 (2) 0.092 (6)
C12 0.560 (2) 0.253 (1) 0.294 (2) 0.070 (5)
C13 0.662 (2) 0.232 (1) 0.392 (2) 0.088 (7)
C14 0.782 (3) 0.250 (1) 0.373 (2) 0.116 (9)
C15 0.422 (2) 0.229 (1) 0.283 (2) 0.070 (6)
C16 0.287 (2) 0.095 (1) 0.463 (2) 0.077 (6)
C17 0.399 (2) 0.058 (1) 0.514 (2) 0.092 (7)
C18 0.472 (2) 0.0774 (9) 0.645 (2) 0.086 (5)
C19 0.600 (2) 0.040 (1) 0.683 (2) 0.114 (7)
C20 0.603 (2) −0.032 (2) 0.656 (2) 0.102 (8)
C21 0.513 (3) −0.062 (1) 0.571 (2) 0.099 (7)
C22 0.384 (2) −0.024 (1) 0.500 (2) 0.081 (5)
C23 0.270 (2) −0.047 (1) 0.562 (2) 0.105 (8)
C24 0.158 (2) 0.001 (2) 0.503 (2) 0.105 (8)
C25 0.031 (3) −0.029 (2) 0.528 (3) 0.12 (1)
C26 −0.066 (3) 0.006 (2) 0.492 (4) 0.17 (1)
C27 −0.064 (2) 0.073 (2) 0.432 (3) 0.123 (8)
C28 0.055 (2) 0.107 (1) 0.426 (2) 0.076 (6)
C29 0.160 (2) 0.070 (1) 0.471 (2) 0.070 (6)
C30 0.707 (2) −0.074 (1) 0.726 (3) 0.136 (9)
C31 −0.306 (2) −0.192 (1) 0.233 (3) 0.112 (8)
C32 −0.227 (2) −0.211 (1) 0.354 (2) 0.107 (7)
C33 −0.095 (3) −0.193 (1) 0.343 (2) 0.124 (9)
C34 −0.065 (2) −0.157 (1) 0.234 (2) 0.074 (6)
C35 −0.155 (2) −0.139 (1) 0.118 (2) 0.100 (8)
C36 −0.281 (2) −0.153 (1) 0.127 (2) 0.091 (7)
C37 0.073 (3) −0.137 (1) 0.220 (3) 0.116 (9)
C38 0.213 (2) −0.010 (1) 0.063 (2) 0.078 (7)
C39 0.097 (2) 0.0365 (10) 0.000 (2) 0.063 (5)
C40 0.040 (2) 0.065 (1) 0.095 (2) 0.109 (7)
C41 −0.084 (2) 0.098 (1) 0.020 (2) 0.104 (6)
C42 −0.080 (2) 0.135 (1) −0.104 (3) 0.083 (6)
C43 0.020 (3) 0.128 (1) −0.180 (2) 0.105 (7)
C44 0.142 (2) 0.083 (1) −0.127 (2) 0.094 (6)
C46 0.371 (3) 0.091 (2) −0.011 (2) 0.098 (8)
C47 0.486 (3) 0.121 (1) 0.019 (2) 0.109 (8)
C48 0.583 (3) 0.087 (2) 0.085 (3) 0.1297 (1)*
C49 0.578 (3) 0.024 (2) 0.145 (3) 0.1531 (1)*
C50 0.459 (2) −0.015 (2) 0.136 (2) 0.124 (9)
C51 0.346 (2) 0.028 (2) 0.065 (2) 0.106 (8)
C52 −0.183 (2) 0.187 (1) −0.159 (2) 0.104 (7)
H10 0.7310 0.3271 0.0748 0.1217*
H11 0.5240 0.2995 0.0955 0.1097*
H13 0.6468 0.2044 0.4721 0.1048*
H14 0.8497 0.2400 0.4467 0.1398*
H17 0.4542 0.0694 0.4463 0.1104*
H18A 0.4890 0.1259 0.6416 0.1034*
H18B 0.4211 0.0685 0.7187 0.1034*
H19A 0.6606 0.0612 0.6310 0.1368*
H19B 0.6265 0.0462 0.7820 0.1368*
H21 0.5237 −0.1094 0.5499 0.1187*
H22 0.3714 −0.0355 0.4019 0.0973*
H23A 0.2502 −0.0934 0.5368 0.1257*
H23B 0.2858 −0.0426 0.6629 0.1257*
H25 0.0263 −0.0742 0.5685 0.1476*
H26 −0.1461 −0.0133 0.5055 0.2024*
H27 −0.1420 0.0955 0.3953 0.1475*
H28 0.0596 0.1536 0.3926 0.0909*
H30A 0.6733 −0.1141 0.7655 0.1630*
H30B 0.7611 −0.0878 0.6580 0.1630*
H30C 0.7557 −0.0478 0.7989 0.1630*
H32 −0.2550 −0.2341 0.4328 0.1281*
H33 −0.0293 −0.2067 0.4163 0.1496*
H35 −0.1294 −0.1185 0.0357 0.1202*
H36 −0.3470 −0.1355 0.0592 0.1091*
H39 0.0363 0.0050 −0.0463 0.0758*
H40A 0.0201 0.0309 0.1609 0.1314*
H40B 0.0933 0.0997 0.1432 0.1314*
H41A −0.1439 0.0617 −0.0014 0.1244*
H41B −0.1139 0.1291 0.0865 0.1244*
H43 0.0162 0.1516 −0.2692 0.1266*
H44 0.1660 0.0543 −0.2009 0.1132*
H45A 0.2673 0.1588 −0.1407 0.1111*
H45B 0.2215 0.1542 0.0076 0.1111*
H47 0.4976 0.1681 −0.0088 0.1308*
H48 0.6643 0.1087 0.0922 0.1552*
H49 0.6542 0.0036 0.1931 0.1842*
H50 0.4522 −0.0604 0.1708 0.1486*
H52A −0.1773 0.2270 −0.0988 0.1247*
H52B −0.1716 0.2013 −0.2526 0.1247*
supporting information
sup-4
Acta Cryst. (2005). E61, o1620–o1621
Atomic displacement parameters (Å2)
U11 U22 U33 U12 U13 U23
Br1 0.144 (2) 0.187 (4) 0.121 (2) −0.090 (3) 0.024 (1) −0.006 (2) Br2 0.147 (2) 0.158 (3) 0.114 (2) −0.081 (2) 0.009 (1) 0.017 (2)
O3 0.073 (10) 0.12 (1) 0.12 (1) 0.033 (9) 0.006 (8) 0.037 (9)
O4 0.066 (9) 0.077 (10) 0.067 (8) −0.003 (7) 0.002 (6) 0.010 (7)
O5 0.14 (1) 0.11 (1) 0.093 (9) 0.01 (1) −0.014 (8) 0.036 (8)
O6 0.10 (1) 0.10 (1) 0.073 (9) 0.001 (10) 0.008 (7) 0.023 (9)
N7 0.09 (1) 0.08 (1) 0.11 (1) 0.01 (1) 0.027 (10) 0.03 (1)
N8 0.06 (1) 0.10 (2) 0.08 (1) −0.013 (10) 0.006 (8) 0.00 (1)
C9 0.15 (2) 0.10 (2) 0.05 (1) −0.04 (2) 0.06 (1) −0.03 (1)
C10 0.15 (2) 0.11 (2) 0.05 (1) −0.07 (2) 0.04 (1) 0.01 (1)
C11 0.12 (2) 0.10 (2) 0.06 (1) −0.01 (2) 0.023 (10) 0.02 (1)
C12 0.08 (1) 0.07 (1) 0.06 (1) 0.00 (1) 0.021 (10) 0.010 (10)
C13 0.09 (2) 0.11 (2) 0.07 (1) −0.02 (2) 0.01 (1) 0.00 (1)
C14 0.12 (2) 0.16 (3) 0.06 (1) −0.04 (2) −0.01 (1) 0.03 (1)
C15 0.08 (2) 0.09 (2) 0.05 (1) 0.03 (1) 0.02 (1) −0.01 (1)
C16 0.11 (2) 0.06 (2) 0.06 (1) −0.01 (1) 0.00 (1) 0.00 (1)
C17 0.08 (1) 0.13 (2) 0.07 (1) −0.04 (2) −0.01 (1) −0.01 (1)
C18 0.11 (1) 0.09 (1) 0.061 (10) 0.02 (1) 0.009 (10) −0.017 (10)
C19 0.09 (1) 0.11 (2) 0.13 (2) 0.00 (1) −0.01 (1) 0.04 (1)
C20 0.10 (2) 0.13 (2) 0.08 (1) 0.01 (2) 0.02 (1) 0.01 (2)
C21 0.17 (2) 0.07 (1) 0.07 (1) 0.03 (1) 0.06 (1) 0.03 (1)
C22 0.09 (1) 0.09 (2) 0.062 (10) −0.01 (1) −0.006 (9) 0.00 (1)
C23 0.10 (2) 0.13 (2) 0.08 (1) −0.01 (2) −0.02 (1) 0.01 (1)
C24 0.09 (2) 0.14 (3) 0.10 (2) −0.03 (2) 0.06 (1) −0.06 (2)
C25 0.09 (2) 0.15 (2) 0.12 (2) −0.06 (2) −0.01 (2) 0.05 (2)
C26 0.14 (3) 0.18 (4) 0.19 (3) −0.06 (2) 0.00 (2) −0.01 (3)
C27 0.06 (2) 0.17 (3) 0.14 (2) 0.03 (2) 0.04 (1) −0.02 (2)
C28 0.07 (1) 0.10 (1) 0.06 (1) 0.01 (1) 0.008 (9) −0.024 (10)
C29 0.10 (2) 0.07 (1) 0.052 (10) −0.05 (1) 0.03 (1) −0.03 (1)
C30 0.12 (2) 0.15 (2) 0.14 (2) 0.04 (2) 0.04 (2) 0.02 (2)
C31 0.10 (2) 0.08 (2) 0.14 (2) −0.04 (1) −0.06 (1) 0.00 (2)
C32 0.11 (2) 0.07 (2) 0.13 (2) −0.02 (1) −0.01 (1) 0.02 (1)
C33 0.17 (2) 0.10 (2) 0.09 (1) −0.01 (2) −0.02 (2) 0.04 (2)
C34 0.10 (2) 0.06 (1) 0.06 (1) −0.02 (1) −0.02 (1) −0.01 (1)
C35 0.09 (2) 0.12 (2) 0.09 (1) −0.04 (2) −0.01 (1) 0.02 (1)
C36 0.10 (2) 0.08 (2) 0.09 (1) −0.03 (1) 0.00 (1) 0.02 (1)
C37 0.14 (2) 0.05 (1) 0.14 (2) 0.01 (1) −0.05 (2) 0.02 (1)
C38 0.09 (2) 0.12 (2) 0.032 (9) −0.02 (2) 0.032 (10) −0.01 (1)
C39 0.08 (1) 0.06 (1) 0.06 (1) −0.01 (1) 0.008 (10) 0.01 (1)
C40 0.12 (2) 0.14 (2) 0.07 (1) 0.02 (1) 0.02 (1) −0.06 (1)
C41 0.08 (1) 0.13 (2) 0.10 (2) 0.01 (1) 0.02 (1) 0.02 (1)
C42 0.06 (1) 0.08 (2) 0.12 (2) −0.01 (1) 0.02 (1) −0.03 (1)
C43 0.17 (2) 0.09 (1) 0.06 (1) −0.03 (1) 0.01 (1) 0.004 (10)
C44 0.09 (1) 0.14 (2) 0.06 (1) −0.03 (1) 0.032 (9) −0.05 (1)
C46 0.12 (2) 0.10 (2) 0.09 (2) −0.01 (2) 0.06 (2) −0.04 (1)
C47 0.10 (2) 0.14 (2) 0.09 (1) 0.01 (2) 0.03 (1) −0.05 (1)
C50 0.11 (2) 0.18 (2) 0.07 (1) −0.01 (2) −0.02 (1) −0.03 (2)
C51 0.11 (2) 0.12 (2) 0.08 (1) 0.00 (2) 0.00 (1) −0.05 (1)
C52 0.13 (2) 0.11 (2) 0.07 (1) 0.02 (1) 0.01 (1) 0.00 (1)
Geometric parameters (Å, º)
BR1—C9 1.88 (2) C28—C29 1.33 (3)
BR2—C31 2.00 (2) C28—H28 0.950
O3—C15 1.11 (2) C30—H30A 0.949
O4—N7 1.43 (2) C30—H30B 0.949
O4—C15 1.39 (2) C30—H30C 0.951
O5—C37 1.25 (3) C31—C32 1.38 (3)
O6—N8 1.40 (2) C31—C36 1.30 (3)
O6—C37 1.32 (3) C32—C33 1.45 (4)
N7—C16 1.29 (3) C32—H32 0.951
N8—C38 1.28 (3) C33—C34 1.32 (3)
C9—C10 1.34 (3) C33—H33 0.951
C9—C14 1.36 (3) C34—C35 1.40 (3)
C10—C11 1.34 (4) C34—C37 1.52 (4)
C10—H10 0.950 C35—C36 1.37 (3)
C11—C12 1.43 (3) C35—H35 0.950
C11—H11 0.950 C36—H36 0.950
C12—C13 1.39 (3) C38—C39 1.57 (3)
C12—C15 1.52 (3) C38—C51 1.58 (4)
C13—C14 1.35 (4) C39—C40 1.27 (3)
C13—H13 0.950 C39—C44 1.63 (3)
C14—H14 0.951 C39—H39 0.948
C16—C17 1.39 (3) C40—C41 1.54 (3)
C16—C29 1.43 (3) C40—H40A 0.950
C17—C18 1.42 (2) C40—H40B 0.951
C17—C22 1.60 (3) C41—C42 1.38 (3)
C17—H17 0.951 C41—H41A 0.951
C18—C19 1.54 (3) C41—H41B 0.950
C18—H18A 0.952 C42—C43 1.36 (3)
C18—H18B 0.950 C42—C52 1.52 (3)
C19—C20 1.39 (4) C43—C44 1.58 (3)
C19—H19A 0.950 C43—H43 0.950
C19—H19B 0.949 C44—C45 1.47 (3)
C20—C21 1.30 (3) C44—H44 0.950
C20—C30 1.46 (4) C45—C46 1.50 (4)
C21—C22 1.60 (3) C45—H45A 0.950
C21—H21 0.949 C45—H45B 0.950
C22—C23 1.47 (3) C46—C47 1.34 (4)
C22—H22 0.949 C46—C51 1.46 (4)
C23—C24 1.55 (4) C47—C48 1.30 (4)
supporting information
sup-6
Acta Cryst. (2005). E61, o1620–o1621
C23—H23B 0.951 C48—C49 1.35 (5)
C24—C25 1.50 (4) C48—H48 0.950
C24—C29 1.36 (4) C49—C50 1.45 (4)
C25—C26 1.24 (5) C49—H49 0.950
C25—H25 0.948 C50—C51 1.53 (4)
C26—C27 1.41 (5) C50—H50 0.948
C26—H26 0.950 C52—H52A 0.950
C27—C28 1.42 (3) C52—H52B 0.951
C27—H27 0.949 C52—H52C 0.949
N7—O4—C15 113 (1) H30A—C30—H30C 109.5
N8—O6—C37 114 (1) H30B—C30—H30C 109.5
O4—N7—C16 117 (1) BR2—C31—C32 110 (1)
O6—N8—C38 104 (1) BR2—C31—C36 118 (1)
BR1—C9—C10 121 (1) C32—C31—C36 129 (2)
BR1—C9—C14 119 (1) C31—C32—C33 110 (2)
C10—C9—C14 118 (2) C31—C32—H32 124.4
C9—C10—C11 122 (2) C33—C32—H32 124.9
C9—C10—H10 119.2 C32—C33—C34 121 (2)
C11—C10—H10 118.8 C32—C33—H33 119.2
C10—C11—C12 120 (1) C34—C33—H33 119.7
C10—C11—H11 120.1 C33—C34—C35 122 (2)
C12—C11—H11 119.8 C33—C34—C37 122 (2)
C11—C12—C13 117 (1) C35—C34—C37 115 (1)
C11—C12—C15 113 (1) C34—C35—C36 117 (1)
C13—C12—C15 126 (1) C34—C35—H35 121.1
C12—C13—C14 118 (1) C36—C35—H35 121.1
C12—C13—H13 120.5 C31—C36—C35 117 (1)
C14—C13—H13 120.5 C31—C36—H36 121.4
C9—C14—C13 123 (2) C35—C36—H36 121.4
C9—C14—H14 118.5 O5—C37—O6 122 (2)
C13—C14—H14 118.5 O5—C37—C34 118 (2)
O3—C15—O4 125 (2) O6—C37—C34 117 (1)
O3—C15—C12 127 (2) N8—C38—C39 132 (1)
O4—C15—C12 104 (1) N8—C38—C51 114 (1)
N7—C16—C17 119 (2) C39—C38—C51 112 (2)
N7—C16—C29 115 (2) C38—C39—C40 113 (1)
C17—C16—C29 124 (2) C38—C39—C44 108 (1)
C16—C17—C18 120 (2) C38—C39—H39 104.7
C16—C17—C22 113 (1) C40—C39—C44 120 (1)
C16—C17—H17 102.8 C40—C39—H39 104.5
C18—C17—C22 111 (1) C44—C39—H39 104.5
C18—C17—H17 102.9 C39—C40—C41 107 (1)
C22—C17—H17 103.0 C39—C40—H40A 109.9
C17—C18—C19 116 (1) C39—C40—H40B 109.9
C17—C18—H18A 107.8 C41—C40—H40A 109.9
C17—C18—H18B 107.7 C41—C40—H40B 109.8
C19—C18—H18B 107.8 C40—C41—C42 119 (1)
H18A—C18—H18B 109.3 C40—C41—H41A 106.9
C18—C19—C20 117 (1) C40—C41—H41B 107.0
C18—C19—H19A 107.5 C42—C41—H41A 107.1
C18—C19—H19B 107.5 C42—C41—H41B 107.2
C20—C19—H19A 107.5 H41A—C41—H41B 109.4
C20—C19—H19B 107.5 C41—C42—C43 121 (1)
H19A—C19—H19B 109.6 C41—C42—C52 122 (1)
C19—C20—C21 121 (2) C43—C42—C52 116 (1)
C19—C20—C30 119 (2) C42—C43—C44 122 (1)
C21—C20—C30 118 (2) C42—C43—H43 118.8
C20—C21—C22 124 (2) C44—C43—H43 118.8
C20—C21—H21 117.7 C39—C44—C43 103 (1)
C22—C21—H21 117.8 C39—C44—C45 108 (1)
C17—C22—C21 110 (1) C39—C44—H44 111.3
C17—C22—C23 109 (1) C43—C44—C45 110 (1)
C17—C22—H22 108.1 C43—C44—H44 111.4
C21—C22—C23 112 (1) C45—C44—H44 111.3
C21—C22—H22 108.1 C44—C45—C46 115 (1)
C23—C22—H22 108.1 C44—C45—H45A 108.0
C22—C23—C24 107 (1) C44—C45—H45B 108.0
C22—C23—H23A 109.9 C46—C45—H45A 107.9
C22—C23—H23B 109.9 C46—C45—H45B 107.9
C24—C23—H23A 109.9 H45A—C45—H45B 109.4
C24—C23—H23B 109.8 C45—C46—C47 125 (2)
H23A—C23—H23B 109.4 C45—C46—C51 111 (2)
C23—C24—C25 111 (2) C47—C46—C51 118 (2)
C23—C24—C29 128 (2) C46—C47—C48 120 (2)
C25—C24—C29 117 (2) C46—C47—H47 119.7
C24—C25—C26 117 (3) C48—C47—H47 119.4
C24—C25—H25 121.3 C47—C48—C49 126 (2)
C26—C25—H25 121.6 C47—C48—H48 117.0
C25—C26—C27 124 (3) C49—C48—H48 116.8
C25—C26—H26 117.7 C48—C49—C50 120 (2)
C27—C26—H26 118.2 C48—C49—H49 119.8
C26—C27—C28 119 (2) C50—C49—H49 119.3
C26—C27—H27 120.0 C49—C50—C51 112 (2)
C28—C27—H27 120.4 C49—C50—H50 124.1
C27—C28—C29 116 (2) C51—C50—H50 123.9
C27—C28—H28 121.8 C38—C51—C46 126 (2)
C29—C28—H28 122.0 C38—C51—C50 113 (2)
C16—C29—C24 111 (2) C46—C51—C50 119 (2)
C16—C29—C28 123 (2) C42—C52—H52A 109.4
C24—C29—C28 123 (2) C42—C52—H52B 109.4
C20—C30—H30A 109.4 C42—C52—H52C 109.5
C20—C30—H30B 109.4 H52A—C52—H52B 109.4
C20—C30—H30C 109.4 H52A—C52—H52C 109.6
supporting information
sup-8
Acta Cryst. (2005). E61, o1620–o1621
BR1—C9—C10—C11 178 (1) C21—C22—C23—C24 170 (1)
BR1—C9—C14—C13 −175 (2) C22—C17—C16—C29 33 (2)
BR2—C31—C32—C33 −175 (1) C22—C21—C20—C30 173 (2)
BR2—C31—C36—C35 177 (1) C22—C23—C24—C25 163 (1)
O3—C15—O4—N7 6 (2) C22—C23—C24—C29 −34 (3)
O3—C15—C12—C11 −31 (3) C23—C24—C25—C26 176 (2)
O3—C15—C12—C13 165 (2) C23—C24—C29—C28 −176 (1)
O4—N7—C16—C17 2 (2) C24—C25—C26—C27 0 (5)
O4—N7—C16—C29 −177 (1) C24—C29—C28—C27 8 (2)
O4—C15—C12—C11 161 (1) C25—C24—C29—C28 −16 (3)
O4—C15—C12—C13 −1 (2) C25—C26—C27—C28 −7 (5)
O5—C37—O6—N8 7 (3) C26—C25—C24—C29 12 (3)
O5—C37—C34—C33 0 (3) C26—C27—C28—C29 4 (3)
O5—C37—C34—C35 −174 (2) C31—C32—C33—C34 4 (3)
O6—N8—C38—C39 2 (2) C31—C36—C35—C34 −11 (3)
O6—N8—C38—C51 174 (1) C32—C31—C36—C35 13 (3)
O6—C37—C34—C33 −167 (2) C32—C33—C34—C35 −4 (3)
O6—C37—C34—C35 17 (3) C32—C33—C34—C37 −179 (2)
N7—O4—C15—C12 174 (1) C33—C32—C31—C36 −9 (3)
N7—C16—C17—C18 77 (2) C33—C34—C35—C36 7 (3)
N7—C16—C17—C22 −146 (1) C36—C35—C34—C37 −176 (2)
N7—C16—C29—C24 166 (1) C37—O6—N8—C38 −158 (1)
N7—C16—C29—C28 −2 (2) C38—C39—C40—C41 −170 (1)
N8—O6—C37—C34 174 (1) C38—C39—C44—C43 −179 (1)
N8—C38—C39—C40 72 (2) C38—C39—C44—C45 −62 (2)
N8—C38—C39—C44 −152 (2) C38—C51—C46—C45 13 (3)
N8—C38—C51—C46 172 (2) C38—C51—C46—C47 170 (2)
N8—C38—C51—C50 2 (2) C38—C51—C50—C49 −176 (2)
C9—C10—C11—C12 3 (3) C39—C38—C51—C46 −14 (3)
C9—C14—C13—C12 −8 (3) C39—C38—C51—C50 175 (1)
C10—C9—C14—C13 6 (3) C39—C40—C41—C42 −43 (2)
C10—C11—C12—C13 −4 (3) C39—C44—C43—C42 14 (2)
C10—C11—C12—C15 −169 (2) C39—C44—C45—C46 66 (2)
C11—C10—C9—C14 −4 (3) C40—C39—C38—C51 −99 (2)
C11—C12—C13—C14 6 (3) C40—C39—C44—C43 −47 (2)
C14—C13—C12—C15 169 (2) C40—C39—C44—C45 70 (2)
C15—O4—N7—C16 157 (1) C40—C41—C42—C43 17 (3)
C16—C17—C18—C19 −173 (2) C40—C41—C42—C52 −160 (1)
C16—C17—C22—C21 −175 (1) C41—C40—C39—C44 59 (2)
C16—C17—C22—C23 −51 (2) C41—C42—C43—C44 −5 (3)
C16—C29—C24—C23 14 (3) C42—C43—C44—C45 −101 (2)
C16—C29—C24—C25 174 (1) C43—C44—C45—C46 179 (1)
C16—C29—C28—C27 175 (1) C44—C39—C38—C51 35 (1)
C17—C16—C29—C24 −14 (2) C44—C43—C42—C52 172 (1)
C17—C16—C29—C28 177 (1) C44—C45—C46—C47 164 (2)
C17—C18—C19—C20 −41 (2) C44—C45—C46—C51 −39 (2)
C17—C22—C23—C24 47 (2) C45—C46—C51—C50 −177 (2)
C18—C17—C16—C29 −102 (2) C46—C47—C48—C49 −8 (4)
C18—C17—C22—C21 −35 (2) C46—C51—C50—C49 13 (3)
C18—C17—C22—C23 88 (1) C47—C46—C51—C50 −19 (3)
C18—C19—C20—C21 17 (3) C47—C48—C49—C50 1 (4)
C18—C19—C20—C30 −161 (2) C48—C47—C46—C51 16 (3)
C19—C18—C17—C22 49 (2) C48—C49—C50—C51 −4 (3)
C19—C20—C21—C22 −6 (3) C48—C49—C50—C51 −4 (3)