The Chemistry of the Platinum Group Metals212-219
Full text
Figure
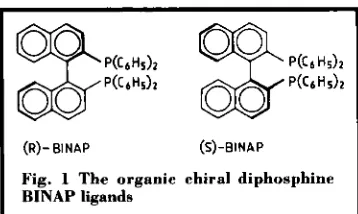
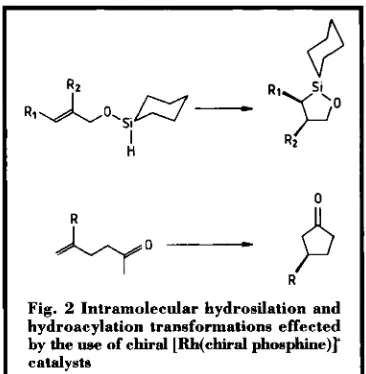
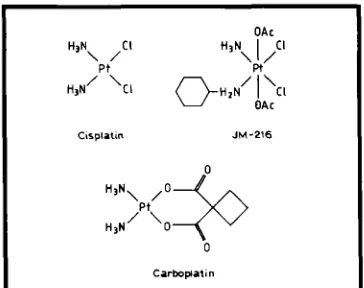
Related documents
SUMMARY ANSWER: Patients identified as research priorities that deserve further investigation: success rates and risks of ART, side-effects of treatments, resources to
Here we report liquid phase epitaxy growth of channel waveguides on laser-machined substrates to fabricate high optical quality lithium niobate channel waveguides with single
Characteristics of new brain lesions in patients with cancer that should raise suspicion of diagnoses other than brain metastasis include (1) primary malignancy without regional
SiZer analysis from mortality data in Denmark in 2013, transformed under a Gamma frailty hazard model.... SiZer analysis from mortality data in Iceland in 2013, transformed under
Since the Ginsparg-Wilson (GW) relation [ 16 ] for the Möbius domain-wall fermion is slightly violated especially for larger lattice spacings [ 17 ], we applied the domain-wall
Wosiek, Positive Representations of a Class of Complex Measures , in Proceedings, 35th International Symposium on Lattice Field Theory (Lattice 2017): Granada, Spain , to ap- pear
Among the health professionals, 15% thought that women generally accepted current uncertainty about the nature and treatment of breast cancer when they were informed; 25%
Using data from the China Family Panel Studies, this study examines the socioeconomic characteristics of Internet users, as well as the relationships between the dynamics of different