www.pharmascholars.com
44
Original Article
CODEN: IJPNL6
TOXICOLOGICAL STUDIES OF AN AYURVEDIC MEDICINE KANKAYANA
GUTIKA
Tanmony Sana
1, Md. Mamun Sikder
1, Paritosh Chakma
1, Marjana Khalil
2, Nayma Abedin
1, Md.
Al Foyjul Islam
1, Mohammad Jashim Uddin
3and M. S. K. Choudhuri
1*
1
Department of Pharmacy, Jahangirnagar University, Savar, Dhaka -1342, Bangladesh
2Department of Pharmacy, East West University, Dhaka-1212, Bangladesh
3
Department of Pharmacy, Jessore University of Science and Technology, Jessore-7408,
Bangladesh
*Corresponding author e-mail: mskc1954@gmail.com
Received on: 28-02-2017; Revised on: 21-04-2017; Accepted on: 10-06-2017
ABSTRACT
Kankayana Gutika (KKY) have been used for the treatment of gulma (localized abdominal swelling or tumor) in Indian subcontinent. However, safety and efficacy of this treatment have not been evaluated. Therefore, the present study was carried out to evaluate the efficacy and safety of KKY in experimental animals. During this study, various experiments on body growth rate, organ-body weight ratio and tissue hydration indices were performed to evaluate
its efficacy and toxicity.To find out the toxicological characteristic of KKY, it was administered chronically to the
male Sprague-Dawley rats at a dose of 400 mg/kg for 46 days and the following toxicological changes were noted. All throughout the experimental period the KKY treated animals were always maintaining negligible changes in body weight. There is a statistically very highly significant (p=0.001, 25.4% decrease) decrease in the relative
percent weight of the male rat liver. There is also a statistically significant (p=0.026, 35.80% decrease) decreasein
the relative percent weight of the male rat thymus.
Keywords: Kankayana Gutika, ayurvedic formulation, percent organ weight, organ water content.
INTRODUCTION
Ayurveda, the ancient Indian traditional system of medicine, aimed with prevention and cure of diseases as well as promotion of quality of life [1, 2]. Thousands
of therapeutics have been described in various classical texts of Ayurveda for the prevention and management of different maladies using numerous plants either single or in combinations. The great scholars of Ayurveda always tried to validate Ayurveda according to that era. Therefore they also
used to include new treatment principles,
formulations and plants from other system of medicine. This was the generosity of the great
Acharyas of Ayurveda for the service of humanity. Kankayan Gutika or vati is ayurvedic medicine in
tablet form which is used in treatment of Gulma roga. In Ayurveda gulma roga is a condition of localised abdominal tumor, lump or swelling. This medicine is made from blend of many natural herbs like
Hedychium spicatum, Inula recemosa, Baliospermum montamum, Plumbago zeylanica, Cajanus cajan,
Zingiber officinale, Acorus calamus, Ipomea turpethum, Ferula asafoetida, Potassium carbonate,
Garcinia pedunculata, Trachyspermum ammi,
Cumminum cyminum, Piper nigrum, Coriandrum sativum, Nigella sativa, Apium graveolens.
Kankayana Gutika is included (page 265) in the Bangladesh National Formulary of Ayurvedic Medicine 1992 (Approved by the Government of Bangladesh vide Ministry of Health and Family
International Journal of Pharmacy
www.pharmascholars.com
45
Welfare Memo No. Health-1/Unani-2/89/ (Part-1) 116 dated 3-6-1991). Bangladesh National Formulary of Ayurvedic Medicine is compiled by the National Unani and Ayurvedic Formulary Committee and published by the Bangladesh Board of Unani and Ayurvedic Systems of Medicine, 75/B Indira Road, Dhaka-1215 under the authority vested in the Board vide section 13(j) of the Bangladesh Unani and Ayurvedic practitioners Ordinance, 1983 [3, 4].
Ayurvedic medicine uses a variety of products and practices. The safety profile of these drugs has not been fully investigated. It is also not clear, whether these preparations might interact with other drugs or diagnostic tests. Therefore the present study was carried out to evaluate the efficacy and safety of KKY in experimental animals.
MATERIALS AND METHODS
Drugs, Chemicals and Reagents: For the toxicological study, Kankayana Gutika (KKY) was collected from Sri Kundeswari Aushadhalaya
Limited, Chittagong. Ketamine injection was
purchased from ACI Limited, Bangladesh. All other reagents, assay kits and chemicals used in this work were purchased from Human GmbH, Wiesbaden, Germany.
Experimental Animals: Six to eight-week old male Sprague-Dawley rats bred and maintained at the animal house of the Department of Pharmacy,
Jahangirnagar University, were used in the
toxicological experiment. These animals were apparently healthy and weighed 70-80 g. The animals were housed in a well-ventilated clean experimental animal house under constant environmental and adequate nutritional conditions throughout the period of the experiment. They were fed with rat chow prepared according to the formula developed at Bangladesh Council of Scientific and Industrial
Research (BCSIR). Water was provided ad libitum
and the animals maintained at 12 h day and 12 h night cycle. All experiments on rats were carried out in absolute compliance with the ethical guide for care and use of laboratory animals approved by Ethical Review Committee, Faculty of Life Sciences, Department of Pharmacy, Jahangirnagar University.
Experimental Design
Acute toxicity study: The acute oral toxicity test was performed following the guidelines of Organization for Economic Co-operation and Development (OECD) for testing of chemicals with minor
modifications (OECD Guideline 425) [5]. Sixteen
male mice (30-40 g body weight) were divided into
four groups of four animals each. Different doses (1000 mg/kg, 2000 mg/kg, 3000 mg/kg and 4000
mg/kg) of experimental drug (KKY) were
administered by stomach tube. The dose was divided into two fractions and given within 12 hours. Then all the experimental animals were observed for mortality and clinical signs of toxicity (general behavior, respiratory pattern, cardiovascular signs, motor activities, reflexes and changes in skin and fur texture) at 1, 2, 3 and 4 hours and thereafter once a day for the next three days following KKY administration.
Chronic toxicity studies: Prior to the experiment, rats were randomly divided into 2 groups of 8 animals each. One group was treated with KKY and another was used as a control. The control animals were administered with distilled water only as per the same volume as the drug treated group for 46 days. For all the pharmacological studies the drugs were administered per oral route at a dose of 400 mg/kg body weight [6]. After acclimatization, Ayurvedic
medicinal preparation was administered to the rats by intra-gastric syringe between the 10 am to 12 am daily throughout the study period. All experiments on rats were carried out in absolute compliance with the ethical guide for care and use of laboratory animals. The experiment animals were marked carefully on the tail which helped to identify a particular animal. By using identification mark, responses were noted separately for a particular period prior to and after the administration [7].
Overall Body Weight Analysis: Careful monitoring of body weights of rats of both groups were
performed throughout the 46 days drug
administration period. Body weights were recorded at regular intervals (2-3 days) until the treatment period was completed. All rats were kept under close observation throughout the experimental period. Statistical analysis of the initial and final growth rates were performed. The growth rate, expressed as percent increment in the body weight. The growth rate of the treatment group was compared with that of the Control group.
Organ Toxicity Study: At the end of the 46 days treatment period, the animals were fasted for 18 hours and also twenty-four hours after the last administration. Ketamine (500 mg/kg i.p.) was administered for the purpose of anesthesia [8].Rats of
www.pharmascholars.com
46
were then detached and preserved in 13% formalin and sent for the evaluation of histological anomalies. The tissues thus subjected to histo-pathological evaluation are: Heart, kidney, lungs, liver, spleen, thymus, stomach, caecum, pancreas, adrenal glands, urinary bladder, reproductive organs, which include
testis, seminal vesicles, prostate gland and
epididymis in case of males and ovaries, fallopian tube and uterus in case of females. Organs like heart, lungs, liver and spleen, portions of these tissues were excised and preserved for histological examination. The remaining portions were dried for determination of water content.
Relative weight of organ= ×100
AOW= Absolute organ weight BW= Body weight
Water content in tissue= ×100
OW1 = organ wet weight
OD = organ dry weight OF = organ foil weight
Statistical Analysis: The data were analyzed using independent sample t-test with the help of SPSS (Statistical Package for Social Science) Statistics 11.5 package (SPSS Inc., Chicago Ill). All values are expressed as mean ± SEM (Standard Error of the Mean) and p<0.05, p<0.01, p<0.001 was taken as the level of significant.
RESULTS
Acute toxicity study: The drug (KKY) administered up to a high dose of 4000 mg/kg produced no mortality. Thus the LD50 value was found to be
greater than 4000 mg/kg body weight. According to the OECD test guideline 425 when there is information in support of low or non-toxicity and immortality nature of the test material, then the limit test at the highest starting dose level (4000 mg/kg body weight) was conducted. There were no mortality and toxicity signs observed at 4000 mg/kg body weight. Therefore, it can be concluded that KKY when administered at single dose is non-toxic and can be used safely in oral formulations.
Chronic growth study
Effect of KKY on Overall Body Weight: The total treatment period was of 46 days. From 16th day
onwards of the total treatment period of 46 days, the body weights of the KKY treated rats were found to be lower than the control group rats. It would be interesting to note here that in the initial 13 days the KKY treated rats were on the contrary having higher body weights, when compared with the control
animals. None of these initial increase followed by decrease in body weights of the KKY treated rats were statistically significantly different from their corresponding body weights of the control animal.
Effect of KKY on Organ Toxicity Study: There is an (p=0.208, 4.10% decrease) decrease in the relative percent weight of the male rat heart, the decrease though not significant yet it was prominent. There is an increase in the relative percent weight of the male rat kidney (p=0.258, 4.57% increase), rat lungs (p=0.164, 11.22% increase), rat spleen (p=0.076, 16.52% increase) and rat testis (p=0.348, 64.64% increase), the increase though not significant yet it was prominent. There is a statistically very highly significant (p=0.001, 25.4 % decrease) decrease in the relative percent weight of the male rat liver. There is also a statistically significant (p=0.026,
35.8% decrease) decrease in the relative percent
weight of the male rat thymus.
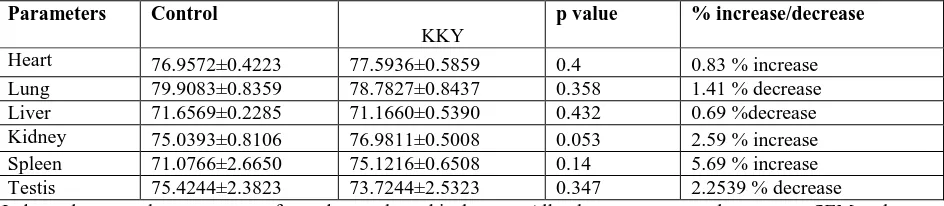
Effect of KKY on Tissue Hydration Index: There is an increase in the organ water content of the male rat heart (p=0.400, 0.83 % increase), rat kidney (p=0.053, 2.59% increase) and rat spleen (p=0.140, 5.69% increase), the increase though not significant yet it was prominent. There is a decrease in the organ water content of the male rat lungs (p=0.358, 1.41 % decrease), rat liver (p=0.432, 0.69% decrease) and rat testis (p=0.347, 2.25% decrease), the decrease though not significant yet it was prominent.
DISCUSSION
The evaluation of organ weights is fundamental to many biological studies. This is particularly true in the field of toxicological drug testing. To eliminate the well-known deviations found in absolute organ weights, the ratio of organ-to-body weight (in percent) is often used, whereas other reference parameters are sometimes preferred, brain or heart weight. But a survey of the relevant literature reveals equally wide deviations in studies of relative organ weights [9-11].
www.pharmascholars.com
47
toxic effects. Severe, rapid body weight loss can also prompt the removal of an animal from a study. Importantly, loss of body weight can impact the interpretation of organ weight data at the conclusion of a study [12]. In our study, from 16th day onwards of
the total treatment period of 46 days, the body weights of the KKY treated rats were found to be lower than the control group rats.
Effect of KKY on Organ Toxicity Study: Dose-related increases in liver weight are commonly observed in repeat-dose toxicity studies performed in rodents. The causes of liver weight changes are diverse. One documented aged-related change in both humans and laboratory rodents is a decline in liver volume [13]. Here we found significantly decrease of
liver weight to the KKY treated rats.
It can be difficult to make a clear distinction in routine toxicity studies from the thymic weight loss and here we found thymus weight decrease to the KKY treated rats but the result is not significant.
Atrophy produced by agents with immune
modulating properties from those compounds that produce similar morphological changes as a result of a generalized, high dose stress response. Indeed thymic involution, decreased responsiveness to Concanavalin A and lowered NK cell activity can be produced by a generalized stress response in a manner similar to that produced by the administration
of glucocorticosteroids [14]. The dose-response
relationship however is of some help in deciding whether thymic atrophy is a direct result of immunosuppression or a non-specific result of stress.
Powerful immunosuppressive drugs such as
cyclophosphamide produce thymic effects in a dose-related manner, with thymic weight loss and atrophy occurring at essentially non-toxic doses. By contrast, atrophy resulting from stress is usually limited to
high doses where there is other clear evidence of stress-related phenomena such as general clinical depression, weight loss or other overt evidence of intoxication [15].
Effect of KKY on Tissue Hydration Index: The water content of tissues is essential to know in the
development of physiologically based
pharmacokinetic modeling [16] and in the
interpretation of drug tissue distribution data [17, 18].
Changes in tissue water content can be also used to evaluate alterations in tissue physiology which are associated with an increase in tissue weight, such as the development of tissue edema [19-21]. In our study
we found that KKY did not cause any significant increase or decrease in percent water content of any organs.
CONCLUSION
From the above experiment it can be concluded that KKY should not be administered chronically at a higher dose as it decreases percent relative weight of rat liver and thymus expressively. Further studies should be done by reducing the administered dose.
ACKNOWLEDGMENT
www.pharmascholars.com
48
Figure 1: The effect KKY (400 mg/kg) on the body weights (g) of Spraque-Dawley rats with the time of treatment. Independent sample t-test was performed to analyze this weight variation in different days. All values are expressed as mean ± SEM and p<0.05, p<0.01, p<0.001 were taken as the level of significant
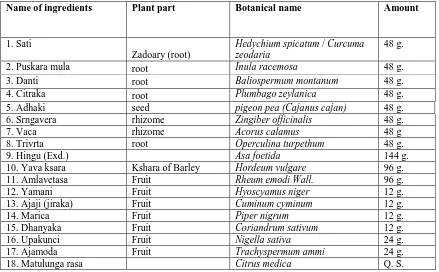
Table1: Name of the ingredients/herbs used in the preparation of Kankayana Gutika (KKY)
Name of ingredients Plant part Botanical name Amount
1. Sati
Zadoary (root)
Hedychium spicatum / Curcuma zeodaria
48 g.
2. Puskara mula root Inula racemosa 48 g.
3. Danti root Baliospermum montanum 48 g.
4. Citraka root Plumbago zeylanica 48 g.
5. Adhaki seed pigeon pea (Cajanus cajan) 48 g.
6. Srngavera rhizome Zingiber officinalis 48 g.
7. Vaca rhizome Acorus calamus 48 g
8. Trivrta root Operculina turpethum 48 g.
9. Hingu (Exd.) Asa foetida 144 g.
10. Yava ksara Kshara of Barley Hordeum vulgare 96 g.
11. Amlavetasa Fruit Rheum emodi Wall. 96 g.
12. Yamani Fruit Hyoscyamus niger 12 g.
13. Ajaji (jiraka) Fruit Cuminum cyminum 12 g.
14. Marica Fruit Piper nigrum 12 g.
15. Dhanyaka Fruit Coriandrum sativum 12 g.
16. Upakunci Fruit Nigella sativa 24 g.
17. Ajamoda Fruit Trachyspermum ammi 24 g.
www.pharmascholars.com
49
Table 2: The effect of Kankayana Gutika (KKY) (400 mg/kg) on the relative organ weights of male rats
Parameters Control KKY p value % increase/decrease
Heart 0.2660±0.0077 0.2551±0.0026 0.208 4 .10 % decrease
Lung 0.5097±0.0331 0.5669±0.0225 0.164 11.22 % increase
Liver 3.6821±0.0820 2.7468±0.1028 0.001 25.4 % decrease
Kidney 0.3105±0.0040 0.3246±0.0112 0.258 4.57 % increase
Spleen 0.2566±0.0139 0.2990±0.0172 0.076 16.52 % increase
Thymus 0.1710±0.0098 0.1372±0.0157 0.026 35.80 % decrease
Testis 0.8063±0.0322 0.8176±0.0210 0.348 64.64 % increase
Independent sample t-test was performed to analyze this data set. All values are expressed as mean ± SEM and p<0.05, p<0.01, p<0.001 were taken as the level of significant
Table 3: The effect of Kankayana Gutika (KKY) (400 mg/kg) on various tissue hydration indices of male rats.
Parameters Control
KKY
p value % increase/decrease
Heart 76.9572±0.4223 77.5936±0.5859 0.4 0.83 % increase
Lung 79.9083±0.8359 78.7827±0.8437 0.358 1.41 % decrease
Liver 71.6569±0.2285 71.1660±0.5390 0.432 0.69 %decrease
Kidney 75.0393±0.8106 76.9811±0.5008 0.053 2.59 % increase
Spleen 71.0766±2.6650 75.1216±0.6508 0.14 5.69 % increase
Testis 75.4244±2.3823 73.7244±2.5323 0.347 2.2539 % decrease
Independent sample t-test was performed to analyze this data set. All values are expressed as mean ± SEM and p<0.05, p<0.01, p<0.001 were taken as the level of significant
REFERENCE
1. Agnivesha, Charaka samhita. Commentary: Vidyotini by Kasinatha Sastri and Gorakha Natha Chaturvedi,
Sutrasthan Volume-I, chapter 30, verse no 26. Chaukhambha Bharati Academy Varanasi, India: 2011, pp. 259.
2. Sushruta, Sushruta samhita, Commentary: Ayurveda Tatva Sandeepika by Ambikadutta Shastri, Sutrasthan
Volume I, chapter 01, verse no 22. Chaukhambha Sanskrit Sansthan Varanasi, India: 2010.
3. Anonymous. Bangladesh National Formulary of Ayurvedic Medicine 1992 (Approved by the Government of
Bangladesh vide Ministry of Health and Family Welfare Memo No. Health-1/Unani-2/89/(Part-1) 116 dated 3-6-1991). National Unani and Ayurvedic Formulary Committee Bangladesh Board of Unani and Ayurvedic Systems of Medicine, 38, Bangabandhu Avenue, Dhaka-1000. (Second ed.), Dhaka, Bangladesh: 2011a.
4. Anonymous. Bangladesh National Formulary of Ayurvedic Medicine 1992 (Approved by the Government of
Bangladesh vide Ministry of Health and Family Welfare Memo No. Health-1/Unani-2/89/(Part-1) 116 dated 3-6-1991). National Unani and Ayurvedic Formulary Committee Bangladesh Board of Unani and Ayurvedic Systems of Medicine, 38, Bangabandhu Avenue, Dhaka, Bangladesh: 2011b.
5. OECD Guideline (425) for the testing of chemicals. Guidance document on acute oral toxicity, Environmental
Health and Safety Monograph Series on Testing and Assessment: 2008, pp. 1-27.
6. Gad S. C. Intl J Tox, 1988; 7(2): 127-38.
7. Stevens K. R. and Gallo M. A. Practical consideration in the conduct of chronic toxicity studies, Principles and
Methods of Toxicology, 2nd edn. Chap. VIII: 1989.
8. Ringler H. and Dabich L. Hematology and clinical biochemistry. In: The Laboratory Rat Biology and Disease
[Baker HL ed]. American College of Laboratory Animal Medicine Series Academic Press: 1979.
9. Webster SH, Liljegren EJ and Zimmer DJ. Amer. J. Anat, 1947; 81: 477-513.
10. Benitz KF, Morask RM and Cummings JR. Lab. Invest, 1961; 10: 936-46.
11. Ber A. Endokrinologie, 1972; 59: 187-96.
12. Adams, ET and Crabbs TA. Basic Approaches in Anatomic Toxicologic Pathology. Chapter 6 of Haschek and
Rousseaux's Handbook of Toxicologic Pathology. Ed W.M. Haschek, C.G. Rousseaux and M.A. Wallig. 3rd revised ed., Elsevier Science: 2013, pp. 158-9.
www.pharmascholars.com
50
14. Monjan AA and Collector MI. Science, 1977; 196: 307-8.
15. Smialowicz RJ, Luebke RW, Riddle MM et al. Journal of Toxicology and Environmental Health, 1985; 15:
561-74.
16. Blakey GE. Tissue kinetics for a series of barbiturates: PhD Thesis. Department of Pharmacy, University of
Manchester, Manchester, U. K: 1995.
17. Kato Y, Hirate J, Sakaguchi K, Ueno M, Horikoshi I. J Pharmacobiodyn, 1987; 10(9) :470-7
18. Khaiafallah N, Jusko WJ. J Pharm Sci, 1984; 73: 362-6.
19. Kanaiwa H, Kuchiwaki H, Inao S, Sugita K. Sue Netlrol, 1995; 44: 172-9.
20. Wang XD, Deng XM, Haraldsen P, Andersson R, Ihse I. Scan J Gastroenterol, 1995; 30:1129-36.
21. Heatherington AC. A physiological based pharmacokinetic model for drug distribution. PhD Thesis.