Hypertonic Saline in Bronchiolitis
and Type I Error: A Trial
Sequential Analysis
Wade Harrison, MD, MPH, a Francois Angoulvant, MD, PhD, b, c Samantha House, DO, MPH, d, e
Vincent Gajdos, MD, PhD, f, g Shawn L. Ralston, MD, MSd, e
BACKGROUND AND OBJECTIVES: Meta-analyses of nebulized hypertonic saline (HS) for acute viral
bronchiolitis have yielded disparate conclusions. Trial sequential analysis (TSA) is a novel method designed to account for potential sources of error in conventional meta-analysis. We sought to use TSA to determine if the existing literature base is sufficient to draw firm conclusions about the effectiveness of HS in bronchiolitis.
METHODS: We used the cohort of studies identified in previously published conventional
meta-analyses. Included studies were those in which authors compared treatment with HS versus normal saline (or supportive care) in children with bronchiolitis to reduce hospital length of stay (LOS) or hospitalizations. TSA results are used to provide a required information size and monitoring boundaries for statistical significance.
RESULTS: For the LOS outcome, 17 studies including 1866 patients analyzed in which authors
used conventional meta-analysis reveal a statistically significant benefit (mean difference =
−0.41 days; 95% confidence interval = −0.07 to −0.75); however, TSA suggests that those
conclusions are premature because of failure to reach the adequate information size of 2665 individuals. For the risk of hospitalization outcome, 8 studies including 1728 patients analyzed in which authors used conventional meta-analysis reveal a reduction in the relative risk of hospitalization (relative risk = 0.86; 95% confidence interval = 0.76 to 0.98); however, TSA suggests these conclusions are premature because of failure to reach the adequate information size of 4770. Both LOS and hospitalization results from conventional meta-analysis would be considered potentially false-positives by TSA.
CONCLUSIONS: TSA reveals that concluding benefit from HS for children with bronchiolitis
potentially represents type I error.
abstract
aDepartment of Pediatrics, Primary Children’s Hospital and University of Utah, Salt Lake City, Utah; bService
d’accueil des urgences pédiatriques, Necker-Enfants Malades University Hospital, Assistance Publique-Hôpitaux de Paris, Paris, France; cEpidémiologie Clinique et Évaluation Économique Appliquées aux Populations
Vulnérables, Institut National de la Santé et de la Recherche Médicale, Paris, France; dChildren’s Hospital
at Dartmouth-Hitchcock, Lebanon, New Hampshire; eDepartment of Pediatrics, Geisel School of Medicine,
Dartmouth College, Hanover, New Hampshire; fService de Pédiatrie, Antoine Béclère University Hospital,
Assistance Publique-Hôpitaux de Paris, Clamart, France; and gCentre for Research in Epidemiology and
Population Health, Villejuif, France
Dr Harrison performed a portion of the analysis and drafted the manuscript; Dr Angoulvant conceptualized the project and critically reviewed all analyses and the manuscript; Drs House and Gajdos critically reviewed and revised the analyses and the manuscript; Dr Ralston designed the project, performed a portion of the analysis, and drafted the manuscript; and all authors approved the final manuscript as submitted.
DOI: https:// doi. org/ 10. 1542/ peds. 2018- 1144 Accepted for publication May 16, 2018
WHAT’S KNOWN ON THIS SUBJECT: Researchers of numerous clinical trials have examined whether nebulized hypertonic saline in acute viral bronchiolitis reduces hospitalization or shortens hospital length of stay, with diminishing benefit observed over time. However, researchers using conventional meta-analyses still report statistically significant benefit.
WHAT THIS STUDY ADDS: Trial sequential analysis reveals that conclusions from conventional meta-analysis about hypertonic saline potentially represent type I error and that authors of further studies may fail to identify a clinically meaningful effect.
Hypertonic saline (HS) has been intensely scrutinized as a therapy for acute viral bronchiolitis, and its use varies widely between centers in the United States.1 There is persistent debate around whether the therapy shortens hospital length of stay (LOS) or decreases risk of hospitalization, the most relevant studied outcomes. The existing evidence base
includes conflicting results from individual trials, many of which are underpowered: a scenario in which meta-analysis is often considered the ideal method for achieving clarity.2–7 Several meta-analyses of HS in bronchiolitis have been published in the past few years, with the authors of the 3 most recent offering alternative interpretations of the evidence despite producing similar point estimates of effect size.4–6 Zhang et al4 conclude that HS is an effective and safe intervention on the basis of overall summary measures of effect showing statistically significant reductions in LOS and hospitalization. In contrast, Heikkilä
et al6 conclude that HS offers only limited clinical benefit, despite observing similar overall point estimates as those reported by Zhang et al.4 Finally, in a living systematic review initiated by Badgett et al, 5 it is concluded that HS is likely ineffective in reducing LOS but that there is potential evidence of benefit for preventing hospitalization. Their differing conclusions arise from several fundamental disagreements, including differing inclusion and exclusion criteria for the study cohort, differing beliefs about the significance of study heterogeneity, and lack of thresholds for clinically meaningful outcomes that are universally agreed on.4–6, 8 Although typically placed at the pinnacle of the evidence pyramid, a growing literature investigating the risk of generating faulty conclusions from conventional meta-analysis is emerging.9–11 In conventional meta-analysis, it is assumed that each study
is an independent, random sample of representative populations, and the evolution of the evidence base over time is not taken into account. Yet, we know from the work of Ioannidis and others that most published early trials are underpowered and suggestive of therapeutic benefit, with results over time tending to regress to a mean that often encompasses the null hypothesis.12, 13 In addition to being subject to publication bias, conventional meta-analytic techniques may be inadequate to address small trial bias and study heterogeneity arising from diversity in study populations, protocols, or outcomes. It is estimated that ˃25% of initial meta-analyses are inaccurately used to conclude a statistically significant treatment effect (type I error).14, 15 Conversely, among meta-analyses failing to reject the null hypothesis, <20% are adequately powered to do so, yet conventional meta-analytic techniques provide no information as to whether the lack of a statistically significant treatment effect represents a true finding or an insufficient number of individuals studied (type II error).9, 15
Trial sequential analysis (TSA) is a novel method for improving the quality of information provided through meta-analysis, specifically by being used to incorporate the element of time into the analysis to establish the sample size required to confirm the point estimate produced by conventional meta-analysis. TSA has recently been applied to a variety of pediatric topics and, in some cases, reveals conclusions contrary to conventional meta-analysis.16–20 Our aim in this study was to apply TSA to HS as therapy for acute viral bronchiolitis. Our specific question was as follows: is the existing literature base sufficient to draw firm conclusions on the impact of HS on hospital LOS and risk of hospitalization?
METHODS
TSA
used to quantify heterogeneity in conventional meta-analysis but is not downwardly biased when study weights are unequal.21 With Fig 1, we provide several hypothetical trajectories for TSA z curves with an explanation of their significance. Methods for TSA are described in greater detail elsewhere.15
Study Selection
Included studies were randomized clinical trials of HS (3%, 5%, or 7%) versus 0.9% saline or standard care alone, with or without
coadministration of bronchodilators, for acute viral bronchiolitis reporting on risk of hospitalization or hospital LOS as an outcome. We identified candidate studies for our analysis first on the basis of their inclusion in a previously published conventional analysis. To identify meta-analyses, we searched PubMed using search terms “hypertonic saline”
and “bronchiolitis” limited to meta-analyses and systematic reviews through March 13, 2018.
Statistical Analysis
Studies were incorporated into TSA in chronological order on the basis of the month and year of first publication (online publication used where available). All base assumptions of effect size (mean difference [MD] for LOS; relative risk [RR] for risk of hospitalization) were derived from conventional meta-analysis by using DerSimonian-Laird random effects models. For the hospitalization outcome, we assumed a control group admission rate of 30%, equal to the US median.22 Given the trend toward the null in published summary statistics over time, sensitivity analyses were also performed evaluating proposed minimum clinically important effect sizes of −0.25 hospital days and 10% absolute risk reduction in hospitalization. We also specified a 2-sided type I (α) error rate of 5% and a power of 80% (type II
[β] error rate of 20%). Data were independently extracted by 2 authors (W.H., S.L.R., and/or S.H.). Analysis was performed by using TSA Viewer version 0.9.5.10 β (Copenhagen Trial Unit, Copenhagen, Denmark).
RESULTS
Search Results
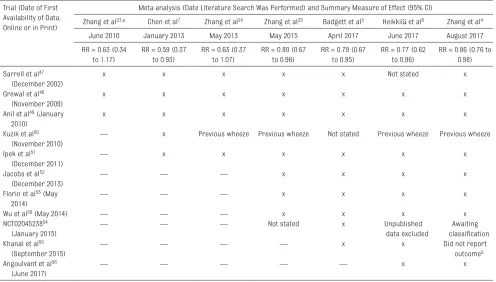
With our initial search, we identified 30 references. After title, abstract, and full-text review, we identified 7 meta-analyses meeting our inclusion and exclusion criteria. We specifically excluded meta-analyses in which authors did not report on both of our study outcomes, LOS and hospitalization.2, 3 We then compared the analytic cohorts for each meta-analysis, cross-referencing all studies included in the 7 meta-analyses by year of publication (or online availability) and the search dates provided in each meta-analysis (Tables 1 and 2). We noted discrepancies between all of the
study cohorts making it difficult to identify a “consensus” cohort of studies. Thus, we chose to perform TSA using the cohort of studies assembled by Zhang et al4 because their inclusion and exclusion criteria have been relatively consistently applied across multiple updates over time. The data extracted and characteristics of the studies included in the TSA are detailed in Supplemental Tables 3 and 4.
LOS
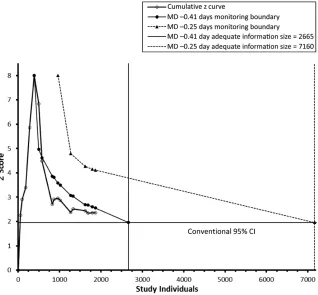
Conventional meta-analysis involving 17 studies (including 1 unpublished study)26–29, 31–36, 38, 40–43, 45, 46 and 1866 patients reveals that HS may reduce hospital LOS (MD = −0.41 days; 95% CI = −0.07 to −0.75) with substantial heterogeneity (I2 = 79%; D2 = 84%). However, under TSA, the z curve fails to cross the monitoring boundary indicating that conclusions based on this point estimate remain at risk for being false-positives and that an additional 799 individuals would FIGURE 1
need to be studied to achieve the required information size to reach firm conclusions (Fig 2). If the current trend in studies were to continue, and we assumed a
minimum clinically important effect size of −0.25 days (6 hours), an information size of 7160 individuals would be required to reach definitive conclusions.
Risk of Hospitalization
Conventional meta-analysis of 8 studies39, 47–49, 51–53, 56 and 1728 patients reveals that HS may reduce
TABLE 1 Agreement in Study Inclusion Across Conventional Meta-analyses on HS and Hospital LOS in Bronchiolitis Trial (Date of First
Availability of Data, Online or in Print)
Meta-analysis (Date Literature Search Was Performed) and Summary Measure of Effect (95% CI)
Zhang et al23,a Chen et al7 Zhang et al24 Zhang et al25,b Badgett et al5 Heikkilä et al6 Zhang et al4
June 2010 January 2013 May 2013 May 2015 April 2017 June 2017 August 2017
MD = −1.16 d (−1.55 to −0.77)
MD = −0.96 d (−1.38 to −0.54)
MD = −1.15 d (−1.49 to −0.82)
MD = −0.45 d (−0.82 to −0.08)
MD = −0.41 d (−0.71 to −0.12)
MD = −0.47 dc
(−0.77 to −0.18)
MD = −0.41 d (−0.75 to −0.07) Mandelberg et al26
(February 2003)
x x x x x x x
Tal et al27 (March
2006)
x x x x x x x
Kuzik et al28
(September 2007)
x x x x x x x
Luo et al29 (August
2009)
x x x x x x x
Al-Ansari et al30 (June
2010) —
x Considered
outpatient setting
Considered outpatient
setting
x x Considered
outpatient setting
Luo et al31 (July 2010) — x x x x x x
Miraglia Del Giudice
et al32 (April 2012) —
Not stated x x x x x
NCT01238848833
(August 2012) —
Unpublished data excluded
Unpublished data excluded
x x Unpublished
data excluded
x Sharma et al34
(December 2012) —
Not stated Not stated x x x x
Mahesh Kumar et al35
(February 2013) — —
Not indexed in searched databases
Not indexed in searched databases
x x x
Pandit et al36 (June
2013) — — —
x x x x
Nenna et al37
(November 2013) — — —
Evaluates other drug
Evaluates other drug
x Evaluates other
drug Ojha et al38 (January
2014) — — —
x x x x
Wu et al39 (May 2014) — — — x x x Includes admitted
patients from ED study Teunissen et al40
(June 2014) — — —
x x x x
Tinsa et al41
(November 2014) — — —
x x x x
Everard et al42
(December 2014) — — —
x x x x
Flores et al43
(September 2015) — — — —
x x x
Silver et al44,d
(November 2015) — — —
x x Previous
wheezing
Previous wheezing Köse et al45 (March
2016) — — — —
x x x
Ratajczyk-Pekrul
et al46 (August 2016) — — — —
Not indexed in searched databases
Not indexed in searched databases
x
a Citation was previously indexed as the 2008 version; however, only the updated 2011 version is currently available through Cochrane Review. b Internal update is by the same author group as previous Cochrane Reviews but is published in Pediatrics.
the risk of hospitalization (RR = 0.86; 95% CI = 0.76 to 0.98) with minimal heterogeneity (I2 = 7%; D2 = 25%). However, under TSA, the z curve fails to cross the monitoring boundary indicating that conclusions based on this point estimate remain at high risk of being false-positives and that an additional 3042 patients would need to be studied to achieve the required information size to reach firm conclusions (Fig 3). If the current trend in studies were to continue, and we assumed a minimum clinically important effect size of 10% absolute RR in hospitalization rates (RR = 0.9), an information size of 9469 individuals would be required to reach definitive conclusions.
DISCUSSION
TSA reveals that the required information size to draw firm conclusions from meta-analysis regarding the use of HS in acute viral bronchiolitis has not been
achieved to date for either LOS or hospitalization. For these outcomes,
∼70% and 36% of the required sample size to do so have been achieved, respectively. Furthermore, preliminary testing results for statistical significance under TSA are negative despite significance under conventional meta-analysis. Thus, the summary statistics generated from existing meta-analyses are more appropriately treated as hypotheticals rather than definitive conclusions. In this sense, the results obtained under conventional meta-analysis, although not mathematically incorrect, are potentially misleading.
With our results, we suggest that continued skepticism about the effectiveness of HS in children with bronchiolitis is reasonable, and we add further support to the concern that the statistical methods used in most meta-analyses can lead to inflated type I error rates.9, 12 This concern is supported by the pattern revealed in this cohort of studies,
in which early meta-analyses revealed large treatment effects that diminished as later and larger studies were incorporated. Systematic error can drive false-positive results in meta-analysis because of publication bias, the inclusion of biased or diverse trials, and post hoc selection bias when conducting the analysis.16 TSA is particularly suited for addressing some of these sources of error by being used to identify a required information size representing “sufficient” as opposed to the “best available”
evidence, directly account for study heterogeneity, and to explicitly consider evolution of the evidence base over time in the primary analysis.15
For clinicians seeking to reconcile daily practice decisions with an uncertain evidence base, routine use of HS may be viewed as a trade-off between making an error of omission versus one of commission. The more conservative significance threshold of
TABLE 2 Agreement in Study Inclusion Across Conventional Meta-analyses on HS and Hospital Admission Rates in Bronchiolitis Trial (Date of First
Availability of Data, Online or in Print)
Meta-analysis (Date Literature Search Was Performed) and Summary Measure of Effect (95% CI)
Zhang et al23,a Chen et al7 Zhang et al24 Zhang et al25 Badgett et al5 Heikkilä et al6 Zhang et al4
June 2010 January 2013 May 2013 May 2015 April 2017 June 2017 August 2017
RR = 0.63 (0.34 to 1.17)
RR = 0.59 (0.37 to 0.93)
RR = 0.63 (0.37 to 1.07)
RR = 0.80 (0.67 to 0.96)
RR = 0.79 (0.67 to 0.95)
RR = 0.77 (0.62 to 0.96)
RR = 0.86 (0.76 to 0.98) Sarrell et al47
(December 2002)
x x x x x Not stated x
Grewal et al48
(November 2009)
x x x x x x x
Anil et al49 (January
2010)
x x x x x x x
Kuzik et al50
(November 2010) —
x Previous wheeze Previous wheeze Not stated Previous wheeze Previous wheeze Ipek et al51
(December 2011) —
x x x x x x
Jacobs et al52
(December 2013) — — —
x x x x
Florin et al53 (May
2014) — — —
x x x x
Wu et al39 (May 2014) — — — x x x x
NCT0204523854
(January 2015) — — —
Not stated x Unpublished
data excluded
Awaiting classification Khanal et al55
(September 2015) — — — —
x x Did not report
outcomeb
Angoulvant et al56
(June 2017) — — — — —
x x
TSA favors the former over the latter and could delay the use of potentially beneficial therapies. However, given that effect sizes have trended toward the null over time, skepticism is likely warranted in this case. For clinical investigators, an inherent strength of TSA is that it quantifies the necessary size of future studies, a question that conventional meta-analysis cannot be used to answer. Both Zhang et al4 and Heikkilä et al6 state that further clinical trials of nebulized HS are needed. However, given that exceptionally large sample sizes would be necessary to confirm both the currently published effect sizes and those proposed to be of minimum clinical importance, the question of whether more studies truly are needed remains open for debate.
Our study is subject to a number of limitations. First, the findings from any meta-analysis are subject to the strengths, weaknesses, and particular circumstances of the individual trials composing it, an issue our tables outlining the existing study cohorts illustrate clearly by revealing wide variation in individual trial inclusion and exclusion between meta-analyses. Such variability, however, lends further support for employing the more conservative methods for determining statistical significance under TSA. Additionally, we did not perform an independent search for potential studies, instead relying on those identified by previous meta-analyses because part of our aim was to directly compare results from conventional meta-analysis with those obtained by TSA. It is possible that the identification and inclusion of a different group of studies would have affected our results, particularly in regard to whether a required information size had been achieved. Elsewhere, we have previously criticized the choice to interpret meta-analysis at face value when heterogeneity signals are high.2 There was substantial statistical heterogeneity in the LOS FIGURE 2
Cumulative z curve and monitoring boundaries for LOS outcome. Conventional and TSA monitoring boundaries are 2 sided with an α = 5%. For clarity in presentation, we only present positive z scores in figures. Studies are listed in the order they entered the analysis and are as follows: Mandelberg et al, 26 Tal et al, 27 Kuzik et al, 28 Luo et al, 29 Sharma et al, 34 Maheshkumar et al, 35 Pandit et al, 36 Ojha et al, 38
and Teunissen et al.40
FIGURE 3
Cumulative z curve and monitoring boundaries for hospitalization outcome. Conventional and TSA monitoring boundaries are 2 sided with an α = 5%. For clarity in presentation, we only present positive z scores in figures. Studies are listed in the order they entered the analysis and are as follows: Sarrell et al, 47 Grewal et al, 48 Anil et al, 49 Ipek et al, 51 Jacobs et al, 52 Florin et al, 53 Wu et al, 39
cohort that we did not attempt to resolve through sensitivity or subgroup analysis. However, a relative strength of TSA is the explicit incorporation of study heterogeneity adjustments into the analysis. Finally, TSA results depend on making correct assumptions about effect size and event rate. It is important to note that we accepted the empirical estimates of effect size generated by conventional meta-analytic methods as our base assumption; thus, any critique of these assumptions may also be seen as a critique of conventional meta-analysis. Assuming larger effect sizes
than the current point estimates would generate smaller required information sizes and likely reveal HS to be effective; however, such assumptions are counterfactual to the existing evidence.
CONCLUSIONS
The information size necessary to draw definitive conclusions about the impact of HS on hospital LOS and admission rates in acute viral bronchiolitis has not been achieved to date. Estimates from conventional meta-analysis suggesting clinical benefit may represent false-positives;
we also suggest that if the current trend in study results continues, authors of further studies are unlikely to confirm a clinically meaningful effect.
ABBREVIATIONS CI: confidence interval D2: diversity index HS: hypertonic saline I2: inconsistency index LOS: length of stay MD: mean difference RR: relative risk
TSA: trial sequential analysis
REFERENCES
1. Davis J, Thompson AD, Mansbach JM, et al. Multicenter observational study of the use of nebulized hypertonic saline to treat children hospitalized for bronchiolitis from 2008 to 2014. Hosp Pediatr. 2017;7(8):483–491
2. Maguire C, Cantrill H, Hind D, Bradburn M, Everard ML. Hypertonic saline (HS) for acute bronchiolitis: systematic review and meta-analysis. BMC Pulm Med. 2015;15:148
3. Brooks CG, Harrison WN, Ralston SL. Association between hypertonic saline and hospital length of stay in acute viral bronchiolitis: a reanalysis of 2 meta-analyses. JAMA Pediatr. 2016;170(6):577–584
4. Zhang L, Mendoza-Sassi RA, Wainwright C, Klassen TP. Nebulised hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev. 2017;12:CD006458
5. Badgett RG, Vindhyal M, Stirnaman JT, Gibson CM, Halaby R. A living
systematic review of nebulized hypertonic saline for acute
bronchiolitis in infants. JAMA Pediatr. 2015;169(8):788–789
6. Heikkilä P, Renko M, Korppi M. Hypertonic saline inhalations in bronchiolitis-A cumulative meta-analysis. Pediatr Pulmonol. 2018;53(2):233–242
7. Chen YJ, Lee WL, Wang CM, Chou HH. Nebulized hypertonic saline treatment reduces both rate and duration of hospitalization for acute bronchiolitis in infants: an updated meta-analysis.
Pediatr Neonatol. 2014;55(6):431–438 8. Haidich AB. Meta-analysis in medical research. Hippokratia. 2010;14(suppl 1):29–37
9. Brok J, Thorlund K, Gluud C, Wetterslev J. Trial sequential analysis reveals insufficient information size and potentially false positive results in many meta-analyses. J Clin Epidemiol. 2008;61(8):763–769
10. Imberger G, Thorlund K, Gluud C, Wetterslev J. False-positive findings in Cochrane meta-analyses with and without application of trial sequential analysis: an empirical review. BMJ Open. 2016;6(8):e011890
11. Garcia-Alamino JM, Bankhead C, Heneghan C, Pidduck N, Perera R. Impact of heterogeneity and effect size on the estimation of the optimal information size: analysis of recently published meta-analyses. BMJ Open. 2017;7(11):e015888
12. Ioannidis JPA. Why most published research findings are false. PLoS Med. 2005;2(8):e124
13. Alahdab F, Farah W, Almasri J, et al. Treatment effect in earlier trials of patients with chronic medical conditions: a meta-epidemiologic study. Mayo Clin Proc. 2018;93(3):278–283
14. Thorlund K, Devereaux PJ, Wetterslev J, et al. Can trial sequential monitoring
Address correspondence to Shawn L. Ralston, MD, MS, Children’s Hospital at Dartmouth-Hitchcock, 1 Medical Dr, Lebanon, NH 03745. E-mail: shawn.l.ralston@ dartmouth.edu
PEDIATRICS (ISSN Numbers: Print, 0031-4005; Online, 1098-4275). Copyright © 2018 by the American Academy of Pediatrics
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose. FUNDING: No external funding.
boundaries reduce spurious inferences from meta-analyses? Int J Epidemiol. 2009;38(1):276–286 15. Thorlund K, Engstrom J, Wetterslev
J, et al. Copenhagen Trials Unit. Trial sequential analysis. 2017. Available at: www. ctu. dk/ tools- and- links/ trial- sequential- analysis. aspx. Accessed March 30, 2018
16. Brok J, Thorlund K, Wetterslev J, Gluud C. Apparently conclusive meta-analyses may be inconclusive– trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta-analyses. Int J Epidemiol. 2009;38(1):287–298
17. Kelley GA, Kelley KS, Pate RR. Exercise and BMI in overweight and obese children and adolescents: a systematic review and trial sequential meta-analysis. Biomed Res Int. 2015;2015:704539
18. Storebø OJ, Krogh HB, Ramstad E, et al. Methylphenidate for attention-deficit/ hyperactivity disorder in children and adolescents: Cochrane systematic review with meta-analyses and trial sequential analyses of randomised clinical trials. BMJ. 2015;351:h5203 19. Zhang ZQ, Zhong Y, Huang XM,
Du LZ. Airway administration of corticosteroids for prevention of bronchopulmonary dysplasia in premature infants: a meta-analysis with trial sequential analysis.
BMC Pulm Med. 2017;17(1):207 20. Holmskov M, Storebø OJ, Moreira-Maia
CR, et al. Gastrointestinal adverse events during methylphenidate treatment of children and adolescents with attention deficit hyperactivity disorder: a systematic review with meta-analysis and trial sequential analysis of randomised clinical trials.
PLoS One. 2017;12(6):e0178187 21. Wetterslev J, Thorlund K, Brok J, Gluud
C. Estimating required information size by quantifying diversity in random-effects model meta-analyses. BMC Med Res Methodol. 2009;9:86
22. Bourgeois FT, Monuteaux MC, Stack AM, Neuman MI. Variation in emergency department admission rates in US children’s hospitals. Pediatrics. 2014;134(3):539–545
23. Zhang L, Mendoza-Sassi RA, Wainwright C, Klassen TP. Nebulized hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev. 2008;(4):CD006458
24. Zhang L, Mendoza-Sassi RA, Wainwright C, Klassen TP. Nebulised hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev. 2013;(7):CD006458
25. Zhang L, Mendoza-Sassi RA, Klassen TP, Wainwright C. Nebulized hypertonic saline for acute bronchiolitis: a systematic review. Pediatrics. 2015;136(4):687–701
26. Mandelberg A, Tal G, Witzling M, et al. Nebulized 3% hypertonic saline solution treatment in hospitalized infants with viral bronchiolitis. Chest. 2003;123(2):481–487
27. Tal G, Cesar K, Oron A, Houri S, Ballin A, Mandelberg A. Hypertonic saline/ epinephrine treatment in hospitalized infants with viral bronchiolitis reduces hospitalization stay: 2 years experience. Isr Med Assoc J. 2006;8(3):169–173
28. Kuzik BA, Al-Qadhi SA, Kent S, et al. Nebulized hypertonic saline in the treatment of viral bronchiolitis in infants. J Pediatr. 2007;151(3): 266–270, 270.e1
29. Luo Z, Liu E, Luo J, et al. Nebulized hypertonic saline/salbutamol solution treatment in hospitalized children with mild to moderate bronchiolitis. Pediatr Int. 2010;52(2):199–202
30. Al-Ansari K, Sakran M, Davidson BL, El Sayyed R, Mahjoub H, Ibrahim K. Nebulized 5% or 3% hypertonic or 0.9% saline for treating acute bronchiolitis in infants. J Pedatr. 2010;157(4):630–634
31. Luo Z, Fu Z, Liu E, et al. Nebulized hypertonic saline treatment in hospitalized children with moderate to severe viral bronchiolitis. Clin Microbiol Infect. 2011;17(12):1829–1833 32. Miraglia Del Giudice M, Saitta F,
Leonardi S, et al. Effectiveness of nebulized hypertonic saline and epinephrine in hospitalized infants with bronchiolitis. Int J Immunopathol Pharmacol. 2012;25(2):485–491 33. NCT01238848. Efficacy of nebulized
hypertonic saline in the treatment
of acute bronchiolitis (hypertonic). Available at: https:// clinicaltrials. gov/ ct2/ show/ NCT01238848. Accessed March 2, 2018
34. Sharma BS, Gupta MK, Rafik SP. Hypertonic (3%) saline vs 0.93% saline nebulization for acute viral bronchiolitis: a randomized controlled trial. Indian Pediatr. 2013;50(8):743–747
35. Maheshkumar KB, Karunakara BP, Nagalli MM, Mallikarjuna HB. Aerosolised hypertonic saline in hospitalized young children with acute bronchiolitis: a randomized controlled clinical trial. J Pediatr Sci. 2013;5(1):e174
36. Pandit S, Dhawan N, Thakur D. Utility of hypertonic saline in the management of acute bronchiolitis in infants: a randomised controlled study. Int J Clin Pediatr. 2013;2(1):24–29
37. Nenna R, Papoff P, Moretti C, et al. Seen percent hypertonic saline--0.1% hyaluronic acid in infants with mild-to-moderate bronchiolitis. Pediatric Pulmonology. 2014;49(9):919–925 38. Ojha AR, Mathema S, Sah S, Aryal UR.
A comparative study on use of 3% saline versus 0.9% saline nebulization in children with bronchiolitis. J Nepal Health Res Counc. 2014;12(26):39–43 39. Wu S, Baker C, Lang ME, et al. Nebulized
hypertonic saline for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2014;168(7):657–663
40. Teunissen J, Hochs AH, Vaessen-Verberne A, et al. The effect of 3% and 6% hypertonic saline in viral bronchiolitis: a randomised controlled trial. Eur Respir J. 2014;44(4):913–921 41. Tinsa F, Abdelkafi S, Bel Haj I, et al.
A randomized, controlled trial of nebulized 5% hypertonic saline and mixed 5% hypertonic saline with epinephrine in bronchiolitis. Tunis Med. 2014;92(11):674–677
42. Everard ML, Hind D, Ugonna K, et al; SABRE Study Team. SABRE: a multicentre randomised control trial of nebulised hypertonic saline in infants
hospitalised with acute bronchiolitis.
Thorax. 2014;69(12):1105–1112 43. Flores P, Mendes AL, Neto AS. A
the treatment of acute bronchiolitis in hospitalized infants. Pediatr Pulmonol. 2016;51(4):418–425
44. Silver AH, Esteban-Cruciani N, Azzarone G, et al. 3% Hypertonic Saline Versus Normal Saline in Inpatient Bronchiolitis: A Randomized Controlled Trial. Pediatrics. 2015;136(6):1036–1043 45. Köse S, Şehriyaroğlu A, Esen F, et al.
Comparing the efficacy of 7%, 3% and 0.9% saline in moderate to severe bronchiolitis in infants. Balkan Med J. 2016;33(2):193–197
46. Ratajczyk-Pekrul K, Gonerko P, Peregud-Pogorzelski J. The clinical use of hypertonic saline/salbutamol in treatment of bronchiolitis. Pediatr Pol. 2016;91(4):301–307
47. Sarrell EM, Tal G, Witzling M, et al. Nebulized 3% hypertonic saline solution treatment in ambulatory children with viral bronchiolitis decreases symptoms. Chest. 2002;122(6):2015–2020 48. Grewal S, Ali S, McConnell DW,
Vandermeer B, Klassen TP. A randomized trial of nebulized 3% hypertonic saline with epinephrine in
the treatment of acute bronchiolitis in the emergency department.
Arch Pediatr Adolesc Med. 2009;163(11):1007–1012
49. Anil AB, Anil M, Saglam AB, Cetin N, Bal A, Aksu N. High volume normal saline alone is as effective as nebulized salbutamol-normal saline, epinephrine-normal saline, and 3% saline in mild bronchiolitis. Pediatr Pulmonol. 2010;45(1):41–47 50. Kuzik BA, Flavin MP, Zielinski D, et al.
Effect of inhaled hypertonic saline on hospital admission rate in children with viral bronchiolitis: a randomized trial. CJEM. 2010;12(6):477–484 51. Ipek IO, Yalcin EU, Sezer RG, Bozaykut
A. The efficacy of nebulized salbutamol, hypertonic saline and salbutamol/ hypertonic saline combination in moderate bronchiolitis. Pulm Pharmacol Ther. 2011;24(6):633–637 52. Jacobs JD, Foster M, Wan J, Pershad
J. 7% Hypertonic saline in acute bronchiolitis: a randomized controlled trial. Pediatrics. 2014;133(1). Available at: www. pediatrics. org/ cgi/ content/ full/ 133/ 1/ e8
53. Florin TA, Shaw KN, Kittick M, Yakscoe S, Zorc JJ. Nebulized hypertonic saline for bronchiolitis in the emergency department: a randomized clinical trial. JAMA Pediatr. 2014;168(7):664–670
54. NCT02045238. Inhaled hypertonic saline use in the emergency department to treat acute viral bronchiolitis. Available at: https:// clinicaltrials. gov/ ct2/ show/
NCT02045238. Accessed March 2, 2018 55. Khanal A, Sharma A, Basnet S,
Sharma PR, Gami FC. Nebulised hypertonic saline (3%) among children with mild to moderately severe bronchiolitis--a double blind randomized controlled trial. BMC Pediatr. 2015;15(115):
DOI: 10.1542/peds.2018-1144 originally published online August 16, 2018;
2018;142;
Pediatrics
L. Ralston
Wade Harrison, Francois Angoulvant, Samantha House, Vincent Gajdos and Shawn
Hypertonic Saline in Bronchiolitis and Type I Error: A Trial Sequential Analysis
Services
Updated Information &
http://pediatrics.aappublications.org/content/142/3/e20181144
including high resolution figures, can be found at:
References
http://pediatrics.aappublications.org/content/142/3/e20181144#BIBL
This article cites 53 articles, 10 of which you can access for free at:
Subspecialty Collections
http://www.aappublications.org/cgi/collection/bronchiolitis_sub
Bronchiolitis
http://www.aappublications.org/cgi/collection/pulmonology_sub
Pulmonology
following collection(s):
This article, along with others on similar topics, appears in the
Permissions & Licensing
http://www.aappublications.org/site/misc/Permissions.xhtml
in its entirety can be found online at:
Information about reproducing this article in parts (figures, tables) or
Reprints
http://www.aappublications.org/site/misc/reprints.xhtml
DOI: 10.1542/peds.2018-1144 originally published online August 16, 2018;
2018;142;
Pediatrics
L. Ralston
Wade Harrison, Francois Angoulvant, Samantha House, Vincent Gajdos and Shawn
Hypertonic Saline in Bronchiolitis and Type I Error: A Trial Sequential Analysis
http://pediatrics.aappublications.org/content/142/3/e20181144
located on the World Wide Web at:
The online version of this article, along with updated information and services, is
http://pediatrics.aappublications.org/content/suppl/2018/08/14/peds.2018-1144.DCSupplemental
Data Supplement at:
by the American Academy of Pediatrics. All rights reserved. Print ISSN: 1073-0397.