Odor
Perception
in Children
in Relation
to
Nasal
Obstruction
S. Nasrin
Ghorbanian,
MD, MPH, Jack L. Paradise,
MD, and
Richard
L. Doty,
PhD
From the Ambulatory Care Center, Children’s Hospital of Pittsburgh, and Departments of Pediatrics and Community Medicine, University of Pittsburgh, Pittsburgh, and Clinical
Smell and Taste Research Center, University of Pennsylvania School of Medicine, Philadelphia
ABSTRACT. To determine whether nasal obstruction in children results in impaired nasal function, olfactory
sensitivity was assessed in 65 children with various
de-grees of nasal obstruction and in 13 children without
nasal obstruction. Overall, olfactory detection thresholds were directly related to clinical ratings of degree of nasal obstruction. Of the subjects with nasal obstruction attrib-utable to adenoid hypertrophy, 28 received adenoidec-tomy; when retested 2 to 28 months later, 20 of the 28 subjects showed generally commensurate reductions in nasal obstruction ratings and olfactory detection thresh-olds. In a comparison subgroup of 16 subjects retested after intervals of 5 to 13 months without having received intervening adenoidectomy, both the nasal obstruction ratings and the olfactory detection thresholds remained relatively unchanged. These results suggest that, in chil-dren, nasal obstruction of varied degree is associated with commensurate impairment of the ability to smell, and that reduction in the degree of nasal obstruction results in commensurate recovery of the ability to smell.
Olfac-tory function, although hitherto accorded little pediatric attention, deserves consideration both in weighing the
importance of nasal obstruction in children and in de-ciding about therapeutic intervention. Pediatrics 1983; 72:510-516; nasal obstruction, olfactoryfunction, odor per-ception, adenoid hypertrophy, smell sensitivity.
Parents commonly complain of nasal obstruction
in their children and practitioners commonly ob-serve it, but as a clinical entity, nasal obstruction has evoked little pediatric interest. In particular,
Received for publication Oct 10, 1982; accepted Dec 21, 1982. Read by title at the annual meetings of the American Pediatric Society and the Society for Pediatric Research, New York, April
24-25, 1978.
Reprint requests to (J.L.P.) Children’s Hospital of Pittsburgh,
125 DeSoto St, Pittsburgh, PA 15213.
PEDIATRICS (ISSN 0031 4005). Copyright © 1983 by the
American Academy of Pediatrics.
little attention has been accorded the presumed or possible physical and psychological effects of nasal obstruction-on breathing comfort, olfaction, ventilatory function, morbidity (especially from up-per respiratory tract infection), facial appearance,
and craniofacial growth.
We report here a study of the relationship in children between nasal obstruction and one aspect of nasal function, the ability to smell. The clinical
significance of such a relationship derives from the primary role of olfaction in a variety of important functions that include early sensory learning, the perception of flavors, and protection from various
environmental hazards. To the best of our knowl-edge, this is the first evaluation of the influence of nasal obstruction on smell sensitivity in children.
SUBJECTS AND METHODS
The study subjects were 45 boys and 33 girls ranging in age from 5 to 15 years; their age and sex
distributions are shown in Table 1. Of the 78 sub-jects, 59 were white and 19 black, and all had been
enrolled in a comprehensive study of indications for tonsillectomy and adenoidectomy currently in
progress at the Children’s Hospital of Pittsburgh.1 In selecting subjects for the present study, an
at-tempt was made to provide a group in which varied degrees of nasal obstruction, from mild to severe,
as well as the absence of nasal obstruction, would
be adequately represented. Of the 78 subjects, 65
demonstrated nasal obstruction of some degree; the
causes of their obstruction, as diagnosed on the basis of combined clinical, laboratory, skin test, and
TABLE 1. Age and Sex of Study Subjects
Age (yr) No. of Subjects
Male Female Total
5-6
7-8 9-10
11-12 13-15
13 13
11
4 4
12
6 6
5 4
25
19 17
9 8
Total 45 33 78
TABLE 2. Distribution of Causes of Nasal Obstruction
in Study Subjects
Clinical Diagnosis No. of Subjects
Adenoid hypertrophy Chronic rhinitis (CR) Acute upper respiratory tract
infection (AURI) CRandAURI
Undetermined
43* 11
5
3 3
Total 65
* Twenty-six of these subjects also had chronic rhinitis or acute upper respiratory tract infection or both.
degree, if any, of nasal obstruction and the degree, if any, of impairment of smell sensitivity. In the second part, determination of this relationship was repeated after intervals ranging from 2 to 28 months in two subgroups: (1) 28 subjects who, when tested initially, had had nasal obstruction attrib-utable to enlarged adenoids, and who subsequently received adenoidectomy; and (2) 16 subjects who had initially had nasal obstruction of varied degree and cause, and who subsequently received no op-erative intervention. The latter group was tested to help determine whether any changes noted in the postadenoidectomy tests might have been attrib-utable to the effects of repeated testing, aging, or other temporal factors unrelated to the relief of nasal obstruction.
Determination of Nasal Obstruction Ratings and Causes of Nasal Obstruction
The degree of nasal obstruction of each subject was rated immediately before or after olfactory testing (which is described in the next section) by one of five pediatric-nurse-practitioner members or by a physician member (J.L.P.) ofthe tonsillectomy and adenoidectomy study team. Two clinically ob-served features, mouth breathing and hyponasality, were used in this assessment.2 The degree of mouth breathing was assessed when subjects were dis-tracted or in repose, whereas the degree of hypo-nasality was assessed both during subjects’ spon-taneously produced speech and while they repeated specific test phrases-”ninety-nine bananas,”
“funny bunny,” or “my name means money”-with their nostrils alternately open and pinched closed.
In a child without hyponasality, substantial nasal
resonance can be heard as those phrases are sounded, and a marked reduction in resonance is noted once the nostrils have been closed; in a hy-ponasal child, resonance is poor even with the nos-trils open, and closing of the nostrils results in little or no reduction in resonance. Mouth breathing and hyponasality were each rated on a four-point scale (none = 1, mild = 2, moderate = 3, and severe = 4) and the respective ratings were averaged to obtain a nasal obstruction index. The interobserver relia-bility of these ratings has been subjected to contin-uing assessment in the tonsillectomy and ade-noidectomy study and has been maintained at a high level; for example, a sample of 200 successive
paired observations during the period of the present
study showed agreement in 171 ratings, disagree-ment by one point in 28 ratings, and disagreement by two points in one rating.
In addition to these ratings, a clinical impression
was recorded for each subject concerning the pres-ence or the absence of adenoid hypertrophy, chronic (allergic or vasomotor) rhinitis, or acute upper res-piratory tract infection. This impression was based on a detailed history, the appearance of the nasal mucous membrane, the amount and character of nasal secretions, the results of screening allergy skin tests and of a nasal smear for eosinophils, a
WBC and differential count, and a lateral
radi-ograph of the nasopharynx.
Measurement
of Olfactory
Sensitivity
0 0 cXl 0 0 0 0 ?-I.25 -2.00 -3.00 0 -4.00 0 a -J (I) Ui I-. z UJO I-> Ui-.-E >-:, 0> I-.--U 4 U--J 0 0 -j 0 0 0 0 0 0 0 0 0 0 0 0 0 8 0 0 0 0 0 0 0 0 0 -4.50 0 m 000 3 00 000 4
been apparent, the subject was required to choose one or the other bottle.
The staircase was begun at the -4.50 log
concen-tration step of a quarter-log step (volume/volume) dilution series extending from -4.50 to -1.25 log concentrations, and was moved upward in 0.50 log steps (two trials per step) until correct detection
occurred on both trials. At this point, a third trial
was given to decrease the likelihood of chance de-tection at that concentration. If a correct response was not made on the third trial, the staircase was again moved upward in 0.50 log steps until detec-tion was evidenced on three consecutive trials at a
given concentration. When correct responses
oc-curred on all three trials, the staircase was reversed
and subsequently moved up or down in 0.25 log
increments or decrements, depending upon the sub-ject’s performance. Thus, the staircase was moved
up 0.25 log units if an incorrect response occurred
on either of the two trials, and down 0.25 log units if a correct response occurred on both trials. A 30-second interval was interposed between trials. The geometric mean of the first four staircase reversal points following the third staircase reversal was used to determine the threshold value. For cases in which a subject’s threshold was located outside the -4.50 to -1.25 log concentration range, the
con-servative procedure of assigning to the subject
either the -4.50 or the -1.25 log step value, as
appropriate, was adopted.
RESULTS
Relationship of Olfactory Detection Threshold
Values
to Nasal Obstruction
Values
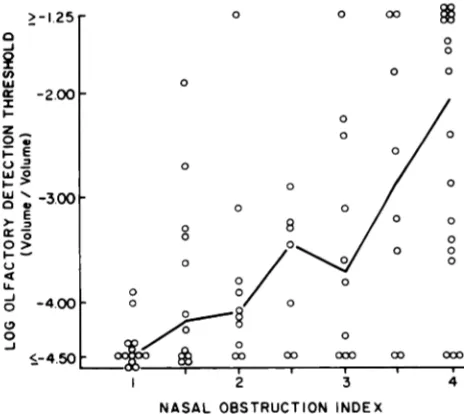
The log olfactory detection threshold values for
all 78 subjects in relation to the corresponding nasal obstruction index values are shown in Fig 1. It is apparent from this figure that these two variables were related directly, with the individuals who
ex-hibited the greatest degrees of nasal obstruction also evidencing, in general, the highest olfactory thresholds (ie, lowest olfactory sensitivity). Also
apparent is the fact that olfactory thresholds were
often heightened even in cases in which nasal ob-struction was of only mild or moderate degree. Not demonstrable from Fig 1 because of the method of assigning values is the fact that six of the children with severe nasal obstruction were unable to detect even undiluted odorant.
The relationship between nasal obstruction and olfactory threshold was significant statistically
when analyzed from several standpoints: the
Spear-man rank-order correlation’#{176} between these two variables was .54, P < .001; the olfactory threshold values differed significantly across the nasal ob-struction index values (Kruskal-Wallis analysis of
NASAL OBSTRUCTION INDEX
Fig 1. Relation between nasal obstruction index and olfactory detection threshold to phenylethyl alcohol in
78 children. Joined points signify median values.
Meth-ods used for determining nasal obstruction index and
olfactory detection threshold are described in text.
variance,’0 H = 24.76, P < .001); and the median olfactory threshold values for the subjects in every nasal obstruction index category above No. 1 (the no-nasal-obstruction category) were significantly greater than was the value for category No. 1
(Mann-Whitney U-tests’#{176}:P < .05 for 1
v
1.5; andP< .001 for 1
v
2, 1v
2.5, 1v3,
1v3.5,
and 1 v4).This relationship between nasal obstruction and olfactory sensitivity was present both in the 43 subjects whose obstruction was attributable wholly or in part to large adenoids (Spearman r = .46, P
< .001), and in the 22 subjects whose obstruction was attributable to other conditions (r = .42, P < .05). Analysis by age and sex showed that the rela-tionship between nasal obstruction and olfactory sensitivity was generally consistent across all subgroups. However, among the 26 subjects with nasal obstruction that was considered severe (nasal obstruction index 3.5 or 4), girls 9 years of age and
older showed significantly greater olfactory
sensi-tivity (lower thresholds) than did either comparably
aged boys or younger girls (Mann-Whitney
U-tests,’#{176}P = .03 and .02, respectively).
Nasal Obstruction
and Olfactory
Sensitivity
Before
and After Adenoidectomy
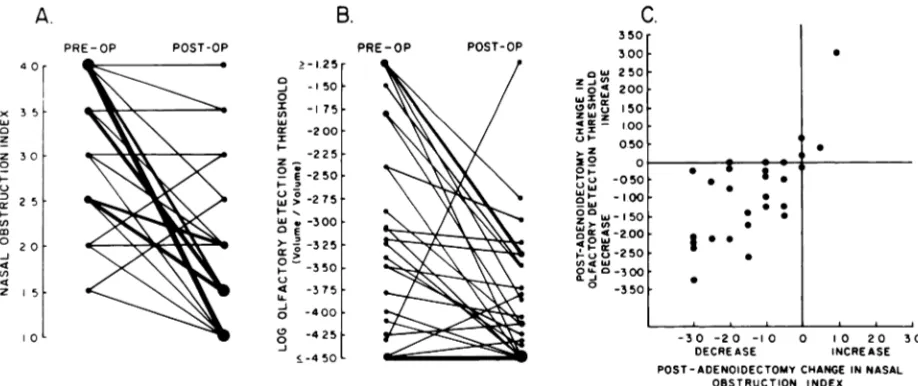
The 28 subjects who had nasal obstruction at-tributed to large adenoids, and who subsequently received adenoidectomy were evaluated both before
adenoidectomy (interval one day to 18 months) and after adenoidectomy (interval 2 to 28 months).
The preoperative-to-postoperative changes in the
30 25 20 w a z z 0 I-U a I-U) 0 -j 4 U) 4 z
A.
B.
350PRE-OP POST-OP PRE-OP POST-OP
300 -I.25 -ISO 0 I -175 U) UJ -200 I-. -225 z-2-25O -, -275 I.-.-.W.-300 a >2325 a:: - -350 C-) 4 -375 U-o,i 250 200 wx ),(nu ISO 100 o 2 C 0k.. -050 -100 00w-I5O )-u) o2OO 4-a: ,.uu-2 C#)4W 03OO 0 -350 -400 -425 -j -4 50 . IC . .. . $ #{149}. . . S. S
I
:
SFig 2. Nasal obstruction index and olfactory detection
threshold in 28 children before and after adenoidectomy:
A, Nasal obstruction index values before and after
ad-enoidectomy. Each line joins preoperative and postoper-ative values for individual subject. Heavy lines denote confluence of several subjects who had same preoperative
changes to each other, are shown graphically in Fig 2. In the case of the nasal obstruction index values,
23 values decreased following adenoidectomy, three values remained unchanged, and two values in-creased. Each of the three subjects showing no change had evidence of allergic rhinitis, whereas one of the two subjects showing an increase had developed an acute upper respiratory tract infec-tion. In the case of the olfactory threshold values, 21 values decreased following adenoidectomy, three remained unchanged, and four increased. Each of the subjects showing no change had had preopera-tive values at the lowest concentration presented, so that postoperative detection of a decrease in threshold would not have been possible by the method used. In the four subjects in whom increases occurred, the postoperative nasal obstruction index values were either the same as or higher than they had been preoperatively. Both the nasal obstruction index values and the olfactory detection threshold values decreased following adenoidectomy in 20 of the 28 subjects. Among the 28 subjects, the corre-lation between the preoperative-to-postoperative
change in nasal obstruction index values and the preoperative-to-postoperative change in olfactory threshold values was significant (Spearman r = .66,
P < .001), indicating proportionality between the
degree of change in nasal obstruction and that in olfactory sensitivity.
Consistency of Relationship Between Nasal Obstruction and Olfactory Sensitivity
Reevaluations after intervals of 5 to 13 months were also carried out in a clinically heterogeneous
-30 -20 -IC 0 0 20 30
DECREASE INCREASE
POST -ADENOIDECTOMY CHANGE IN NASAL
OBSTRUCTION INDEX
and same postoperative values. B, Log olfactory detection
threshold values before and after adenoidectomy. Each
line joins preoperative and postoperative values for mdi-vidual subject. C, Postadenoidectomy changes in nasal
obstruction index in relation to changes in olfactory
detection threshold.
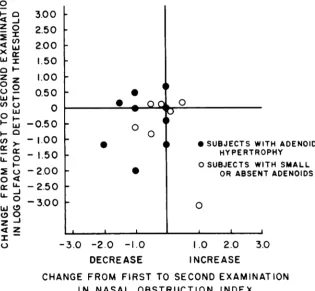
subgroup of 16 subjects in whom the initial degree of nasal obstruction had ranged from none to
se-vere; nine of the subjects had nasal obstruction due to adenoid hypertrophy. The 16 subjects were se-lected on the basis that they were available for a second evaluation and had not received intervening adenoidectomy. Correlations between the nasal ob-struction index values and the olfactory detection threshold values for the 16 subjects were significant on both the first and second evaluations (Spearman r = .57 and .60, respectively; P < .01).
The changes from the first to the second evalu-ation in both nasal obstruction index and olfactory detection threshold values are summarized graphi-cally in Fig 3. The nasal obstruction index de-creased from the first to the second evaluation in eight of the 16 subjects, it remained unchanged in six subjects, and it increased in two subjects; the changes that occurred were, on the whole, of small
magnitude. Similarly, relatively little overall
change occurred in the olfactory detection thresh-old values. Paradoxically, in the one subject whose nasal obstruction index increased from the first to the second examination, the olfactory detection threshold declined markedly. The correlation be-tween the changes in nasal obstruction and those in olfactory threshold was not significant (Spear-man r = .002).
Like the preadenoidectomy and
3.00
2.50
2.00
1.50
1.00
0.50
0
-0.50
- 1.00
- 1.50
-2.00
- 2.50
-3.00 z
0
‘-ci Z
(I,
Xc: WI
ci’-zz
0
0-cfl W
0
‘-ci
Li0
I.-.
0
I-0
.
.oc 0
.
0 0
S
. SUBJECTS WITH ADENOID
HY PE RTROPHV
OSUBJECTS WITH SMALL
OR ABSENT ADENOIDS
0
-3.0 -2.0 -1.0 1.0 2.0 3.0
DECREASE INCREASE
CHANGE FROM FIRST TO SECOND EXAMINATION
IN NASAL OBSTRUCTION INDEX
Fig 3. Changes from first to second examination in nasal obstruction index values and olfactory detection threshold values in 16 children not receiving intervening adenoidec-tomy.
also did the degree of olfactory sensitivity; when the nasal obstruction lessened somewhat, olfactory sensitivity either remained constant or increased. DISCUSSION
Our results suggest that, in children, nasal
ob-struction of varied degree will be associated with commensurate impairment of the ability to smell, and that reduction in the degree of nasal obstruc-tion will result in commensurate recovery of such
ability. This effect of obstruction was observed in our subjects whether the site of obstruction was
within the nasal cavity or posterior to it, in the nasopharynx. Whereas in adults olfactory function is known to be influenced by changes in nasal
patency resulting from conditions such as
vasomo-tor rhinitis,” to the best of our knowledge no pre-vious work has directly assessed the relationship
between olfactory function and nasal breathing in children. Our finding that in older girls with nasal
obstruction olfactory sensitivity remained
rela-tively high may have been attributable to chance,
but it seems in keeping with other observations
that olfactory sensitivity is greater in females than
in males.’2”3
The ability to detect and discern odors serves
children in a number of ways: orientation to the
environment, recognition of noxious or dangerous
airborne substances, detection of fire, perception of
the flavor of foods, and enhancement of the quality
of life through the appreciation of fragrances and
other pleasant aromas. Nonetheless, olfactory func-tion in infants and children has received relatively little investigation. Term newborns, and even
pre-mature infants as young as 28 weeks of gestational age, have been found capable of responding to
ol-factory stimuli.’4’6 More specifically, evidence has
been reported that newborns can distinguish their
own mothers from breast odors alone,’7’9 although the degree to which this ability is used in
infant-mother interactions is not known. Clearly, the loss of smell in children must result in a drastic
reduc-tion of their perceptual world-an effect, inciden-tally, that would be particularly depriving in those
with other sensory or perceptual handicaps. Adults
who have lost their sense of smell complain that
food lacks “taste,” and that both the flavor
sensa-tions and the enjoyment derived from eating are diminished.20 Whether such impairment of the
ap-preciation of food adversely influences nutrition, growth, or developmental processes in children is not known, but clearly deserves study. In this
con-nection it should be noted that children with
especially if they have not recently experienced normal or near-normal olfactory sensitivity. In fact, it was the expression of pleasant surprise by a few children upon first becoming aware, following ad-enoidectomy, of the aroma of certain foods (eg, “I can smell cookies now!”) that initially aroused our interest in studying olfactory sensitivity as an index of nasal function. For the practitioner treating the child with well-established nasal obstruction, the findings of the present study point to olfactory function as a factor to be considered both in weigh-ing the clinical importance of the obstruction and in deciding about the advisability of potentially therapeutic interventions.
The olfactory threshold procedure used in the present study seems the most sophisticated, clini-cally practical means available for measuring smell thresholds, and has several advantages over most other reported procedures. First, the use of forced-choice responses minimizes the biasing influences of response criteria upon the detection measure. Second, the initially ascending staircase minimizes potential influences of adaptation on the threshold measure, since it starts at lower concentrations and rapidly reaches the threshold region with a mini-mum of trials. Finally, because the threshold region is sampled repeatedly, in a manner analogous to the tracking procedure used in audiometry, a rela-tively stable estimate of the threshold is obtained. Nonetheless, the present data must be viewed as reflecting relative rather than absolute measures of olfactory sensitivity, as liquid dilution of odorants may result in fewer molecules in the headspace than predicted by ideal gas laws and fewer than produced by air-dilution systems.2’ Even when the exact stimulus concentration is known at the exter-nal nares, the true concentration at the receptor membrane can be only roughly estimated because of (1) interindividual differences in the configura-tion of the nasal chambers which in turn influence patterns of intranasal airflow; (2) interindividual differences in the volume and rate of inhalation during sniffing; (3) the tendency for molecules to be absorbed differentially by the walls of the nasal chambers before reaching the olfactory epithelium (such effects are analogous to a gas chromato-graph’s retention process)22; and (4) systematic changes in the airflow dynamics of the nose as a result of fluctuations in the engorgement of the turbinates.” Nonetheless, natural sniffing is gen-erally assumed to be superior to procedures that “blast” materials into the nose,” as the latter pro-duce atypical patterns of airflow and stimulus ar-tifacts that result in unreliable measurements.
ACKNOWLEDGMENTS
This study was supported in part by a grant (HD
07403) from the National Institute of Child Health and
Human Development, a grant (NS 16365) from the Na-tional Institute of Neurological and Communicative Dis-orders and Stroke, and a training grant (MCT 298) from the Maternal and Child Health Service, US Public Health Service.
The authors thank the following physicians who re-ferred patients for this study: Drs Charles P. Ashe, Mark Diamond, Barbara Frank, Gilbert A. Friday, Robert F. Good, Harry E. Harris, James G. Hawkins, John Love,
Robert R. Macdonald, Fred S. Marks, Maribel McKelvy,
H. Richard Paul, and James J. Reilly, Sr. Ruth Z. Bach-man, RN, BS, Beverly S. Bernard, RN, BS, Kathleen M.
DiGaudio, RN, Georgann Karantonis, RN, BS, and
Elaine Curcio, RN, BS, examined and rated study sub-jects for nasal obstruction. Gilbert A. Friday, MD, pro-vided consultations regarding the presence or absence of
allergic rhinitis. Bertram R. Girdany, MD, and Mutsuhisa Fuijioka, MD, provided radiographic consultations.
Charles D. Bluestone, MD, and Sylvan E. Stool, MD,
provided otolaryngologic consultations, and also per-formed the adenoidectomies received by study subjects. D. Kathleen Colborn, BS, assisted in collating the data, and Floyd H. Taylor, ScD, provided statistical advice and assisted with the statistical analyses.
REFERENCES
1. Paradise JL: Clinical trials of tonsillectomy and
adenoidec-tomy: Limitations of existing studies and a current effort to
evaluate efficacy. South Med J 1976;69:1049-1053
2. Paradise JL, Bluestone CD, Carrasco MM: Nasal
obstruc-tion due to adenoid hypertrophy: Two year course with and without adenoidectomy. Abstracts, Eighteenth Annual
Meet-irig of the Ambulatory Pediatric Association, New York City,
April 24-25, 1978, p 43
3. Cornsweet TN: The staircase method in psychophysics. Am
J Psychol 1962;75:485-491
4. Doty RL: The effect of duration of stimulus presentation upon the angular acceleration threshold. J Exp Psychol
1969;80:317-321
5. Levitt H: Transformed up-down methods in psychophysics.
J Acoust Soc Am 1971;49:467-477
6. Kunka M, Doty RL, Settle RG: An examination of intertrial interval and gender influences on sucrose detection
thresh-olds established by a modified staircase procedure. Percep-tion 1981;10:35-38
7. McBurney DH, Moskat U: Taste thresholds in college-aged smokers and non-smokers. Percept Psychophys
1975;18:71-73
8. Doty RL, Brugger WE, Jurs PC, et al: Intranasal trigeminal stimulation from odorous volatiles: Psychometric responses
from anosmic and normal humans. Physiol Behav
1978;20:175-185
9. Silver WL, Moulton DG: Chemosensitivity of rat nasal trigeminal receptors. Physiol Be/wv 1982;28:927-933
10. Siegel 5: Nonparametric Statistics for the Behavioral
Sci-ences. New York, McGraw-Hill, 1956
11. Doty RL: A review of olfactory dysfunctions in man. Am J Otolaryngol 1979;1:57-59
12. LeMagnen J: Les ph#{233}nom#{232}nesolfacto-sexuels chez l’homme. Arch Sci Physiol 1952;6:125-160
13. Koeleja HS, Koster EP: Some experiments on sex differ-ences in odor perception. Ann NY Acad Sci 1974;237:234-246
14. Rieser J, Wonas A, Wikner K: Radial localization of odors
by human newborns. Child Dev 1976;47:856-859
15. Sarnat HB: Olfactory reflexes in the newborn infant. J
Pediatr 1978;92:624-626
16. Engen T: Psychophysical analysis of the odor intensity of homologous alcohols. J Exp Psychol 1965;70:611-616
pref-erences in the human neonate. Ciba Found Symp
1975;33:103-117
18. Russell MJ: Human olfactory communication. Nature 1976;260:520-522
19. Schaal B, Montagner H, Hertling E, et al.: Les stimulations
olfactives dans les relations entre l’enfant et la mere. Rep rod
Nutr Dev 1980;20:843-858
20. Doty RL: Food preference ratings of congenitally-anosmic humans, in: Kare MR, Maller 0 (eds): Chemical Senses and
Nutrition II. New York, Academic Press, 1977, pp 315-325 21. Haring HG: Vapor pressures and Raoult’s law deviations in
relation to odor enhancement and suppression, in Turk A, Johnston JW Jr, Moulton DG (eds): Human Responses to Environmental Odors. New York, Academic Press, 1974, pp
199-226
22. Mozell MM, Jagodowicz M: Chromatographic separation of odorants by the nose: Retention times measured across in
vivo olfactory mucosa. Science 1973;181: 1247-1249
DOES VITAMIN SUPPLEMENTATION PREVENT NEURAL TUBE
DEFECTS?
A problem associated with the evaluation of a new treatment regime is that while the expected benefit is specific and often inferred on the basis of previous
uncontrolled experiments, the possibility of harm remains hypothetical until
someone suspects a specific abnormality associated with the treatment. This, of course, should not be taken to mean that no hazard in fact exists. In general the evaluation of the efficacy of a treatment regime should also attempt to identify possible hazards. However, . .. there is a difficulty because, [for example] a study capable of detecting a 50% reduction in the risk of neural tube defects associated with vitamin supplementation among women who have already had
at least one affected infant, will not be statistically powerful enough to detect
adverse effects with an absolute increased risk of, say, a serious malformation of about 2 per 1000-equivalent to increasing the background risk of many serious malformations two- or three-fold. (In assessing the likely benefit in relation to the possible hazard it should be borne in mind that about 90% of
neural tube defects can be detected antenatally, but any abnormality caused by
the vitamin supplementation may not.) Perhaps the sensible course of action is
to design unbiased studies capable of assessing whether vitamin supplementa-tion is effective, and so, the approximate magnitude of the effect, while at the
same time collecting sufficient identifying information on infants born to the
treated women so that .. . the children can be followed up for closer scrutiny of adverse effects. ...
From Wald N: Possible prevention of neural tube defects by vitamin supplementation, in Dobbing J (ed): The Prevention of Spina Bifida and Other Neural Tube Defects. London, Academic Press,