metal-organic papers
m1086
Shawkatalyet al. [Cr(C27H26P2)(CO)4] doi:10.1107/S1600536806013638 Acta Cryst.(2006). E62, m1086–m1087
Acta Crystallographica Section E Structure Reports
Online
ISSN 1600-5368
[1,3-Bis(diphenylphosphino)propane]-tetracarbonylchromium(0)
Omar bin Shawkataly,a* Shahinoor Dulal Islam,a Hoong-Kun Funband Claude Didierjeanc
aChemical Sciences Programme, Centre for
Distance Education, Universiti Sains Malaysia, 11800 USM, Penang, Malaysia,bX-Ray
Crystal-lography Unit, School of Physics, Universiti Sains Malaysia, 11800 USM, Penang, Malaysia, andcLaboratoire de Cristallographie et
Mode´li-sation des Mate´riaux Mine´raux et Biologiques (LCM3B), UMR No. 7036, Universite´ Henri Poincare´, Nancy I, Faculte´ des Sciences, BP 239, 54506 Vandoeuvre le`s Nancy Cedex, France
Correspondence e-mail: omarsa@usm.my
Key indicators
Single-crystal X-ray study
T= 293 K
Mean(C–C) = 0.004 A˚
Rfactor = 0.038
wRfactor = 0.106
Data-to-parameter ratio = 20.4
For details of how these key indicators were automatically derived from the article, see http://journals.iucr.org/e.
Received 23 March 2006 Accepted 15 April 2006
#2006 International Union of Crystallography
All rights reserved
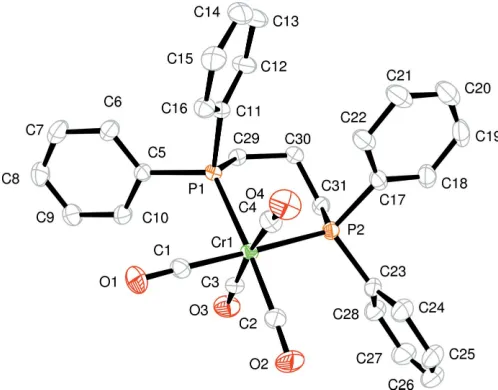
In the title molecule, [Cr(C27H26P2)(CO)4], the Cr atom is octahedrally coordinated by the four carbonyl ligands and a bidentate phosphine ligand, which is bonded as a chelate in a
cisposition. The average Cr—P and Cr—C bond lengths are 2.3770 (5) and 1.865 (2) A˚ , respectively.
Comment
It is generally believed that the metal (M) to carbon monoxide bond involves both OC—M-bonding and M—CO -back-bonding. In view of this phenomenon, the bonding char-acteristics of metal carbonyls with a phosphine ligand in phosphine-substituted metal carbonyls are of interest. A search of the Cambridge Structural Database (Version 5.27; Allen, 2002) revealed only 21 complexes of group VI metal carbonyls with a 3-carbon backbone bidentate phosphine. However, there were no examples of chromium carbonyl complexes. Previously, we reported several crystal structures of phosphine-substituted group VI metal carbonyls (Shaw-katalyet al., 1996, 1997). We present here the crystal structure of the title compound, (I).
The title molecule has an expected octahedral geometry (Fig. 1). The Cr—C bond lengths of theciscarbonyl ligands (with respect to the P atom) are slightly longer than those for
the trans carbonyl group (Table 1). This trend was also
observed in Cr[Ph2P(CH2)2PPh2](CO)4(Bennettet al., 1971) and Cr[Ph2P(CH2)4PPh2](CO)4 (Ueng & Shih, 1992). The bidentate phosphine bite angle [89.124 (17)] is intermediate
between that observed in Cr[Ph2P(CH2)2PPh2](CO)4 [83.41 (8)] and that in Cr[Ph
2P(CH2)4PPh2](CO)4 [93.29 (5)]. Comparison of the mean Cr—C and C—O
2.3770 (5) A˚ , are relatively short in spite of the presence of the bulky phosphine ligand.
Experimental
A mixture of Cr(CO)6(234 mg, 1.064 mmol) and Ph2P(CH2)3PPh2
(439 mg, 1.065 mmol) was refluxed in a purified mixture of petroleum ether (373–403 K, 30 ml) andn-butanol (20 ml) forca12 h under dry deoxygenated nitrogen. The solvent was rotary evaporated and the reaction mixture was dissolved in acetone (3 ml) and filtered. Yellow crystals (454 mg, 74% yield) were obtained by slow evaporation of the acetone solution at room temperature. Analysis calculated for C31H26CrO4P2: C 64.58, H 4.42%; found C 64.59, H 4.15%.
Crystal data
[Cr(C27H26P2)(CO)4]
Mr= 576.46 Monoclinic,P21=n a= 10.0562 (1) A˚
b= 20.8494 (3) A˚
c= 14.1616 (2) A˚
= 106.4183 (7) V= 2848.13 (6) A˚3
Z= 4
Dx= 1.344 Mg m 3 MoKradiation
= 0.55 mm 1
T= 293 (2) K Prism, yellow 0.40.380.28 mm
Data collection
Siemens SMART CCD diffractometer
!scans
Absorption correction: multi-scan (SADABS; Sheldrick, 1996)
Tmin= 0.772,Tmax= 0.899
22851 measured reflections 7010 independent reflections 5175 reflections withI> 2(I)
Rint= 0.055 max= 28.3
Refinement
Refinement onF2 R[F2> 2(F2)] = 0.038
wR(F2) = 0.106
S= 1.01 7010 reflections 343 parameters
H-atom parameters constrained
w= 1/[2
(Fo2) + (0.0569P)2
+ 0.004P]
whereP= (Fo2+ 2Fc2)/3
(/)max= 0.001
max= 0.27 e A˚ 3
[image:2.610.45.294.71.266.2]min= 0.44 e A˚ 3
Table 1
Selected geometric parameters (A˚ ,).
Cr1—C2 1.854 (2) Cr1—C1 1.8547 (19) Cr1—C3 1.863 (2)
Cr1—C4 1.889 (2) Cr1—P2 2.3719 (5) Cr1—P1 2.3821 (5)
C2—Cr1—C3 87.71 (10) C2—Cr1—C4 86.29 (10)
P2—Cr1—P1 89.124 (17)
All H atoms were placed at calculated positions and refined using a riding model, with C—H = 0.93 and 0.97 A˚ , respectively, and
Uiso(H) = 1.2Ueq(C).
Data collection: XSCANS (Siemens, 1994); cell refinement:
XSCANS; data reduction: XSCANS; program(s) used to solve structure:SHELXS86(Sheldrick, 1985); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics:
ORTEP-3 for Windows(Farrugia, 1997); software used to prepare material for publication:WinGX(Farrugia, 1999).
We thank the Malaysian Government and Universiti Sains Malaysia for supportviaIRPA grant Nos. 09-02-05-0008 and 190-9609-2801.
References
Allen, F. H. (2002).Acta Cryst.B58, 380–388.
Bennett, M. J., Cotton, F. A. & LaPrade, M. D. (1971).Acta Cryst.B27, 1899– 1904.
Farrugia, L. J. (1997).J. Appl. Cryst.30, 565. Farrugia, L. J. (1999).J. Appl. Cryst.32, 837–838.
Jost, A., Rees, B. & Yelon, W. B. (1975).Acta Cryst.B31, 2649–2658. Shawkataly, O. b., Saminathan, T., Muniswaran, K., Fun, H.-K. & Sivakumar,
K. (1996).Acta Cryst.C52, 1352–1355.
Shawkataly, O. b., Umathavan, A., Ramalingam, K., Fun, H.-K. & Ibrahim, A. R. (1997).Acta Cryst.C53, 1543–1545.
Sheldrick, G. M. (1985). Crystallographic Computing 3, edited by G. M. Sheldrick, C. Kruger & R. Goddard, pp. 175–189. Oxford University Press. Sheldrick, G. M. (1996).SADABS. University of Go¨ttingen, Germany. Sheldrick, G. M. (1997).SHELXL97. University of Go¨ttingen, Germany. Siemens (1994).XSCANS. Version 2.1. Siemens Analytical X-ray Instruments
Inc., Madison, Wisconsin, USA.
[image:2.610.314.566.205.264.2]Ueng, C.-H. & Shih, G.-Y. (1992).Acta Cryst.C48, 988–991. Whitaker, A. & Jeffery, J. W. (1967).Acta Cryst.23, 977–984. Figure 1
supporting information
sup-1 Acta Cryst. (2006). E62, m1086–m1087
supporting information
Acta Cryst. (2006). E62, m1086–m1087 [https://doi.org/10.1107/S1600536806013638]
[1,3-Bis(diphenylphosphino)propane]tetracarbonylchromium(0)
Omar bin Shawkataly, Shahinoor Dulal Islam, Hoong-Kun Fun and Claude Didierjean
[1,3-Bis(diphenylphosphino)propane]tetracarbonylchromium(0)
Crystal data
[Cr(C27H26P2)(CO)4]
Mr = 576.46 Monoclinic, P21/n Hall symbol: -P 2yn
a = 10.0562 (1) Å
b = 20.8494 (3) Å
c = 14.1616 (2) Å
β = 106.4183 (7)°
V = 2848.13 (6) Å3
Z = 4
F(000) = 1192
Dx = 1.344 Mg m−3
Mo Kα radiation, λ = 0.71073 Å Cell parameters from 319 reflections
θ = 2–27.5°
µ = 0.55 mm−1
T = 293 K Prism, yellow 0.4 × 0.38 × 0.28 mm
Data collection
Siemens SMART CCD diffractometer
Graphite monochromator
ω scans
Absorption correction: multi-scan (SADABS; Sheldrick, 1996)
Tmin = 0.772, Tmax = 0.899 22851 measured reflections
7010 independent reflections 5175 reflections with I > 2σ(I)
Rint = 0.055
θmax = 28.3°, θmin = 1.8°
h = −13→13
k = −27→20
l = −18→18
Refinement
Refinement on F2 Least-squares matrix: full
R[F2 > 2σ(F2)] = 0.038
wR(F2) = 0.106
S = 1.01 7010 reflections 343 parameters
0 restraints
H-atom parameters constrained
w = 1/[σ2(F
o2) + (0.0569P)2 + 0.004P] where P = (Fo2 + 2Fc2)/3
(Δ/σ)max = 0.001 Δρmax = 0.27 e Å−3 Δρmin = −0.44 e Å−3
Special details
Experimental. IR (cyclohexane) υ(CO) 1886 (s), 1916 (s), 2005 (s) cm-1.
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
x y z Uiso*/Ueq
Cr1 0.96448 (3) 0.111085 (13) 0.735830 (19) 0.03725 (9)
P1 0.86042 (4) 0.00814 (2) 0.73235 (3) 0.03493 (11)
P2 0.80586 (5) 0.13384 (2) 0.58048 (3) 0.04135 (12)
C1 1.09147 (19) 0.08794 (10) 0.85356 (14) 0.0504 (5)
C2 1.0557 (2) 0.18880 (11) 0.74023 (16) 0.0594 (5)
C3 1.07736 (18) 0.07890 (10) 0.66285 (14) 0.0471 (4)
C4 0.8556 (2) 0.15279 (10) 0.80628 (15) 0.0530 (5)
C5 0.97166 (17) −0.05727 (8) 0.79854 (13) 0.0421 (4)
C6 0.9292 (2) −0.10028 (12) 0.85827 (19) 0.0686 (6)
H6 0.8425 −0.0956 0.8687 0.082*
C7 1.0151 (3) −0.15045 (14) 0.9028 (2) 0.0912 (9)
H7 0.9852 −0.179 0.943 0.109*
C8 1.1419 (3) −0.15842 (12) 0.8888 (2) 0.0778 (7)
H8 1.1984 −0.1923 0.9187 0.093*
C9 1.1859 (2) −0.11645 (11) 0.8304 (2) 0.0707 (7)
H9 1.2725 −0.1217 0.8201 0.085*
C10 1.1015 (2) −0.06578 (10) 0.78640 (17) 0.0609 (6)
H10 1.1335 −0.0369 0.7478 0.073*
C11 0.71110 (17) 0.00413 (9) 0.78154 (13) 0.0412 (4)
C12 0.5824 (2) −0.02047 (12) 0.72960 (15) 0.0594 (6)
H12 0.5716 −0.0399 0.6688 0.071*
C13 0.4710 (2) −0.01622 (14) 0.76790 (18) 0.0762 (8)
H13 0.3852 −0.032 0.7318 0.091*
C14 0.4847 (2) 0.01087 (14) 0.85839 (18) 0.0735 (7)
H14 0.4086 0.0141 0.8831 0.088*
C15 0.6117 (2) 0.03305 (12) 0.91175 (16) 0.0639 (6)
H15 0.6225 0.0501 0.9741 0.077*
C16 0.7242 (2) 0.03034 (10) 0.87383 (14) 0.0502 (5)
H16 0.8095 0.0463 0.9106 0.06*
C17 0.62374 (19) 0.15565 (9) 0.56493 (16) 0.0508 (5)
C18 0.5421 (2) 0.17804 (11) 0.4749 (2) 0.0691 (6)
H18 0.5816 0.187 0.4244 0.083*
C19 0.3995 (3) 0.18698 (11) 0.4607 (3) 0.0890 (10)
H19 0.344 0.2011 0.4001 0.107*
C20 0.3413 (3) 0.17507 (13) 0.5358 (3) 0.0912 (10)
H20 0.247 0.1818 0.526 0.109*
C21 0.4202 (2) 0.15370 (13) 0.6236 (2) 0.0799 (8)
H21 0.3801 0.1457 0.6741 0.096*
C22 0.5611 (2) 0.14359 (10) 0.63890 (19) 0.0616 (6)
H22 0.6145 0.1285 0.6996 0.074*
C23 0.8652 (2) 0.19773 (10) 0.51331 (16) 0.0555 (5)
C24 0.8604 (3) 0.26097 (12) 0.5449 (2) 0.0761 (7)
H24 0.8225 0.2702 0.5961 0.091*
C25 0.9132 (3) 0.31046 (13) 0.4987 (3) 0.0974 (10)
supporting information
sup-3 Acta Cryst. (2006). E62, m1086–m1087
C26 0.9687 (3) 0.29688 (17) 0.4223 (3) 0.1008 (11)
H26 1.0026 0.3299 0.3915 0.121*
C27 0.9739 (3) 0.23567 (17) 0.3920 (2) 0.0944 (10)
H27 1.0125 0.2268 0.3411 0.113*
C28 0.9219 (2) 0.18603 (13) 0.43645 (19) 0.0719 (7)
H28 0.9252 0.1442 0.4143 0.086*
C31 0.77742 (19) 0.06610 (9) 0.49387 (12) 0.0453 (4)
H31A 0.7208 0.0804 0.43 0.054*
H31B 0.866 0.0523 0.4865 0.054*
C30 0.70663 (18) 0.00914 (9) 0.52721 (13) 0.0425 (4)
H30A 0.6698 −0.0189 0.4712 0.051*
H30B 0.629 0.0249 0.5485 0.051*
C29 0.80093 (18) −0.03010 (8) 0.61057 (12) 0.0405 (4)
H29A 0.8821 −0.0424 0.5908 0.049*
H29B 0.7523 −0.0692 0.6176 0.049*
O1 1.17206 (17) 0.07395 (9) 0.92573 (11) 0.0808 (5)
O2 1.1160 (2) 0.23610 (9) 0.74588 (16) 0.0982 (7)
O3 1.14855 (16) 0.06075 (9) 0.61755 (13) 0.0756 (5)
O4 0.79485 (18) 0.18096 (9) 0.85031 (14) 0.0837 (5)
Atomic displacement parameters (Å2)
U11 U22 U33 U12 U13 U23
Cr1 0.03476 (15) 0.03623 (16) 0.03828 (16) −0.00351 (11) 0.00630 (11) −0.00363 (11)
P1 0.0327 (2) 0.0362 (2) 0.0363 (2) −0.00120 (16) 0.01032 (17) −0.00165 (17)
P2 0.0410 (2) 0.0363 (2) 0.0425 (2) −0.00067 (18) 0.00493 (19) 0.00066 (18)
C1 0.0458 (10) 0.0551 (12) 0.0460 (10) −0.0068 (9) 0.0060 (8) −0.0034 (9)
C2 0.0561 (11) 0.0537 (12) 0.0592 (12) −0.0118 (10) 0.0012 (10) −0.0001 (10)
C3 0.0403 (9) 0.0492 (11) 0.0515 (11) −0.0062 (8) 0.0124 (8) 0.0034 (9)
C4 0.0542 (11) 0.0447 (11) 0.0563 (12) −0.0019 (9) 0.0097 (9) −0.0138 (9)
C5 0.0423 (9) 0.0380 (9) 0.0441 (9) −0.0005 (7) 0.0092 (7) 0.0012 (7)
C6 0.0601 (12) 0.0646 (15) 0.0863 (16) 0.0098 (11) 0.0291 (12) 0.0292 (12)
C7 0.0884 (19) 0.0743 (18) 0.115 (2) 0.0119 (14) 0.0351 (17) 0.0516 (16)
C8 0.0703 (15) 0.0564 (14) 0.0961 (19) 0.0146 (12) 0.0063 (14) 0.0230 (13)
C9 0.0524 (12) 0.0620 (14) 0.0958 (18) 0.0153 (10) 0.0180 (12) 0.0164 (13)
C10 0.0502 (11) 0.0547 (12) 0.0811 (15) 0.0098 (9) 0.0240 (10) 0.0202 (11)
C11 0.0373 (8) 0.0453 (10) 0.0426 (9) 0.0005 (7) 0.0140 (7) 0.0039 (7)
C12 0.0448 (10) 0.0891 (17) 0.0455 (11) −0.0164 (10) 0.0145 (9) −0.0043 (10)
C13 0.0433 (11) 0.127 (2) 0.0606 (14) −0.0172 (13) 0.0189 (10) 0.0052 (14)
C14 0.0533 (12) 0.110 (2) 0.0672 (15) 0.0085 (13) 0.0338 (12) 0.0165 (14)
C15 0.0742 (14) 0.0752 (15) 0.0533 (12) 0.0089 (12) 0.0357 (11) 0.0003 (11)
C16 0.0503 (10) 0.0567 (12) 0.0459 (10) −0.0012 (9) 0.0174 (9) −0.0015 (9)
C17 0.0427 (9) 0.0352 (9) 0.0659 (12) 0.0020 (8) 0.0012 (9) −0.0053 (9)
C18 0.0604 (12) 0.0453 (12) 0.0842 (16) 0.0021 (10) −0.0076 (12) 0.0053 (11)
C19 0.0656 (15) 0.0396 (12) 0.126 (2) 0.0040 (11) −0.0318 (16) 0.0050 (14)
C20 0.0454 (12) 0.0513 (14) 0.161 (3) 0.0022 (11) 0.0025 (17) −0.0185 (17)
C21 0.0449 (12) 0.0684 (16) 0.123 (2) −0.0003 (11) 0.0184 (14) −0.0202 (16)
C23 0.0506 (10) 0.0472 (11) 0.0583 (12) −0.0050 (9) −0.0019 (9) 0.0140 (9)
C24 0.0785 (16) 0.0509 (14) 0.0904 (18) −0.0074 (12) 0.0103 (14) 0.0089 (12)
C25 0.092 (2) 0.0496 (15) 0.129 (3) −0.0130 (14) −0.0043 (19) 0.0267 (17)
C26 0.0872 (19) 0.089 (2) 0.110 (3) −0.0260 (17) 0.0020 (18) 0.047 (2)
C27 0.0843 (18) 0.111 (3) 0.0856 (19) −0.0198 (17) 0.0206 (15) 0.0378 (18)
C28 0.0725 (14) 0.0749 (16) 0.0663 (14) −0.0099 (13) 0.0162 (12) 0.0191 (12)
C31 0.0494 (10) 0.0468 (10) 0.0360 (9) −0.0022 (8) 0.0064 (8) −0.0007 (7)
C30 0.0424 (9) 0.0452 (10) 0.0378 (9) −0.0034 (7) 0.0082 (7) −0.0086 (7)
C29 0.0455 (9) 0.0356 (9) 0.0420 (9) −0.0033 (7) 0.0151 (8) −0.0063 (7)
O1 0.0704 (10) 0.0977 (14) 0.0563 (9) −0.0015 (9) −0.0113 (8) 0.0078 (9)
O2 0.0938 (13) 0.0655 (11) 0.1168 (16) −0.0410 (10) −0.0003 (12) 0.0030 (10)
O3 0.0662 (9) 0.0894 (13) 0.0869 (12) 0.0007 (9) 0.0470 (9) −0.0062 (9)
O4 0.0917 (12) 0.0727 (12) 0.0935 (13) 0.0057 (10) 0.0374 (10) −0.0357 (10)
Geometric parameters (Å, º)
Cr1—C2 1.854 (2) C14—H14 0.93
Cr1—C1 1.8547 (19) C15—C16 1.384 (3)
Cr1—C3 1.863 (2) C15—H15 0.93
Cr1—C4 1.889 (2) C16—H16 0.93
Cr1—P2 2.3719 (5) C17—C18 1.387 (3)
Cr1—P1 2.3821 (5) C17—C22 1.389 (3)
P1—C11 1.8271 (18) C18—C19 1.403 (4)
P1—C29 1.8397 (17) C18—H18 0.93
P1—C5 1.8431 (17) C19—C20 1.374 (4)
P2—C23 1.832 (2) C19—H19 0.93
P2—C17 1.8388 (19) C20—C21 1.348 (4)
P2—C31 1.8393 (18) C20—H20 0.93
C1—O1 1.148 (2) C21—C22 1.388 (3)
C2—O2 1.148 (3) C21—H21 0.93
C3—O3 1.152 (2) C22—H22 0.93
C4—O4 1.150 (2) C23—C28 1.386 (3)
C5—C10 1.376 (3) C23—C24 1.397 (3)
C5—C6 1.381 (3) C24—C25 1.403 (4)
C6—C7 1.389 (3) C24—H24 0.93
C6—H6 0.93 C25—C26 1.380 (5)
C7—C8 1.355 (4) C25—H25 0.93
C7—H7 0.93 C26—C27 1.352 (5)
C8—C9 1.360 (3) C26—H26 0.93
C8—H8 0.93 C27—C28 1.388 (4)
C9—C10 1.388 (3) C27—H27 0.93
C9—H9 0.93 C28—H28 0.93
C10—H10 0.93 C31—C30 1.526 (2)
C11—C16 1.388 (3) C31—H31A 0.97
C11—C12 1.393 (3) C31—H31B 0.97
C12—C13 1.378 (3) C30—C29 1.526 (2)
C12—H12 0.93 C30—H30A 0.97
supporting information
sup-5 Acta Cryst. (2006). E62, m1086–m1087
C13—H13 0.93 C29—H29A 0.97
C14—C15 1.368 (3) C29—H29B 0.97
C2—Cr1—C1 89.35 (9) C14—C15—C16 120.7 (2)
C2—Cr1—C3 87.71 (10) C14—C15—H15 119.7
C1—Cr1—C3 91.85 (9) C16—C15—H15 119.7
C2—Cr1—C4 86.29 (10) C15—C16—C11 120.74 (18)
C1—Cr1—C4 89.97 (9) C15—C16—H16 119.6
C3—Cr1—C4 173.71 (9) C11—C16—H16 119.6
C2—Cr1—P2 93.04 (7) C18—C17—C22 118.5 (2)
C1—Cr1—P2 175.97 (7) C18—C17—P2 120.24 (19)
C3—Cr1—P2 85.01 (6) C22—C17—P2 120.89 (15)
C4—Cr1—P2 93.42 (6) C17—C18—C19 119.4 (3)
C2—Cr1—P1 176.57 (7) C17—C18—H18 120.3
C1—Cr1—P1 88.34 (6) C19—C18—H18 120.3
C3—Cr1—P1 89.82 (6) C20—C19—C18 120.5 (3)
C4—Cr1—P1 96.25 (6) C20—C19—H19 119.8
P2—Cr1—P1 89.124 (17) C18—C19—H19 119.8
C11—P1—C29 104.65 (8) C21—C20—C19 120.4 (2)
C11—P1—C5 102.61 (8) C21—C20—H20 119.8
C29—P1—C5 98.38 (8) C19—C20—H20 119.8
C11—P1—Cr1 115.91 (6) C20—C21—C22 120.2 (3)
C29—P1—Cr1 115.75 (6) C20—C21—H21 119.9
C5—P1—Cr1 117.14 (6) C22—C21—H21 119.9
C23—P2—C17 102.57 (9) C21—C22—C17 121.0 (2)
C23—P2—C31 102.96 (9) C21—C22—H22 119.5
C17—P2—C31 98.52 (8) C17—C22—H22 119.5
C23—P2—Cr1 113.24 (6) C28—C23—C24 118.4 (2)
C17—P2—Cr1 123.12 (7) C28—C23—P2 123.13 (18)
C31—P2—Cr1 113.70 (6) C24—C23—P2 118.3 (2)
O1—C1—Cr1 178.71 (19) C23—C24—C25 119.6 (3)
O2—C2—Cr1 177.5 (2) C23—C24—H24 120.2
O3—C3—Cr1 178.03 (18) C25—C24—H24 120.2
O4—C4—Cr1 176.08 (19) C26—C25—C24 120.3 (3)
C10—C5—C6 117.47 (18) C26—C25—H25 119.9
C10—C5—P1 119.54 (14) C24—C25—H25 119.9
C6—C5—P1 122.96 (15) C27—C26—C25 120.2 (3)
C5—C6—C7 120.5 (2) C27—C26—H26 119.9
C5—C6—H6 119.8 C25—C26—H26 119.9
C7—C6—H6 119.8 C26—C27—C28 120.5 (3)
C8—C7—C6 121.0 (2) C26—C27—H27 119.8
C8—C7—H7 119.5 C28—C27—H27 119.8
C6—C7—H7 119.5 C27—C28—C23 121.0 (3)
C7—C8—C9 119.4 (2) C27—C28—H28 119.5
C7—C8—H8 120.3 C23—C28—H28 119.5
C9—C8—H8 120.3 C30—C31—P2 112.92 (12)
C8—C9—C10 120.0 (2) C30—C31—H31A 109
C10—C9—H9 120 C30—C31—H31B 109
C5—C10—C9 121.6 (2) P2—C31—H31B 109
C5—C10—H10 119.2 H31A—C31—H31B 107.8
C9—C10—H10 119.2 C31—C30—C29 114.47 (14)
C16—C11—C12 117.96 (17) C31—C30—H30A 108.6
C16—C11—P1 118.14 (13) C29—C30—H30A 108.6
C12—C11—P1 123.81 (15) C31—C30—H30B 108.6
C13—C12—C11 120.4 (2) C29—C30—H30B 108.6
C13—C12—H12 119.8 H30A—C30—H30B 107.6
C11—C12—H12 119.8 C30—C29—P1 117.33 (12)
C14—C13—C12 121.1 (2) C30—C29—H29A 108
C14—C13—H13 119.5 P1—C29—H29A 108
C12—C13—H13 119.5 C30—C29—H29B 108
C15—C14—C13 119.1 (2) P1—C29—H29B 108
C15—C14—H14 120.4 H29A—C29—H29B 107.2
C13—C14—H14 120.4
C11—P1—C5—C10 −174.66 (16) C31—P2—C17—C22 108.51 (17)
C29—P1—C5—C10 78.19 (17) Cr1—P2—C17—C22 −17.15 (19)
Cr1—P1—C5—C10 −46.50 (18) C22—C17—C18—C19 −0.8 (3)
C11—P1—C5—C6 7.6 (2) P2—C17—C18—C19 172.25 (17)
C29—P1—C5—C6 −99.61 (19) C17—C18—C19—C20 1.4 (4)
Cr1—P1—C5—C6 135.71 (17) C18—C19—C20—C21 −1.0 (4)
C10—C5—C6—C7 −0.8 (4) C19—C20—C21—C22 0.1 (4)
P1—C5—C6—C7 177.1 (2) C20—C21—C22—C17 0.5 (4)
C5—C6—C7—C8 −0.1 (5) C18—C17—C22—C21 −0.1 (3)
C6—C7—C8—C9 0.3 (5) P2—C17—C22—C21 −173.14 (17)
C7—C8—C9—C10 0.4 (4) C17—P2—C23—C28 −123.19 (18)
C6—C5—C10—C9 1.5 (3) C31—P2—C23—C28 −21.2 (2)
P1—C5—C10—C9 −176.4 (2) Cr1—P2—C23—C28 101.97 (18)
C8—C9—C10—C5 −1.3 (4) C17—P2—C23—C24 60.63 (18)
C29—P1—C11—C16 −179.29 (15) C31—P2—C23—C24 162.58 (17)
C5—P1—C11—C16 78.41 (16) Cr1—P2—C23—C24 −74.21 (18)
Cr1—P1—C11—C16 −50.52 (16) C28—C23—C24—C25 −0.3 (3)
C29—P1—C11—C12 −3.0 (2) P2—C23—C24—C25 176.05 (18)
C5—P1—C11—C12 −105.32 (18) C23—C24—C25—C26 0.4 (4)
Cr1—P1—C11—C12 125.75 (17) C24—C25—C26—C27 −0.7 (4)
C16—C11—C12—C13 2.6 (3) C25—C26—C27—C28 0.9 (4)
P1—C11—C12—C13 −173.70 (19) C26—C27—C28—C23 −0.8 (4)
C11—C12—C13—C14 −1.4 (4) C24—C23—C28—C27 0.5 (3)
C12—C13—C14—C15 −1.0 (4) P2—C23—C28—C27 −175.68 (19)
C13—C14—C15—C16 2.2 (4) C23—P2—C31—C30 −171.53 (13)
C14—C15—C16—C11 −1.0 (4) C17—P2—C31—C30 −66.44 (14)
C12—C11—C16—C15 −1.4 (3) Cr1—P2—C31—C30 65.57 (13)
P1—C11—C16—C15 175.12 (17) P2—C31—C30—C29 −75.43 (17)
C23—P2—C17—C18 41.00 (19) C31—C30—C29—P1 68.68 (18)
C31—P2—C17—C18 −64.41 (18) C11—P1—C29—C30 76.92 (14)
supporting information
sup-7 Acta Cryst. (2006). E62, m1086–m1087