organic papers
Acta Cryst.(2005). E61, o2261–o2262 doi:10.1107/S1600536805019355 Zhouet al. C
19H14ClNO3S
o2261
Acta Crystallographica Section E Structure Reports
Online
ISSN 1600-5368
3-[3-(4-Chlorophenyl)-4-oxothiazolidin-2-yl]-6-methyl-4H-benzopyran-4-one
Zhong-Zhen Zhou, Wei Huang, Pei-Liang Zhao, Qiong Chen and Guang-Fu Yang*
Key Laboratory of Pesticide and Chemical Biology of Ministry of Education, College of Chemistry, Central China Normal University, Wuhan 430079, People’s Republic of China
Correspondence e-mail: gfyang@mail.ccnu.edu.cn
Key indicators
Single-crystal X-ray study
T= 293 K
Mean(C–C) = 0.003 A˚
Rfactor = 0.044
wRfactor = 0.134
Data-to-parameter ratio = 16.5
For details of how these key indicators were automatically derived from the article, see http://journals.iucr.org/e.
#2005 International Union of Crystallography Printed in Great Britain – all rights reserved
The molecular structure of the title compound,
C19H14ClNO3S, contains a 4-oxothiazolidine ring in a near
planar geometry.
Comment
3-Substituted chromones are important because of their widespread occurrence in nature and their interesting biolo-gical activities. Meanwhile, thiazolidinones have been reported to possess a wide range of biological activities, including antifungal, antibacterial, antihistaminic, anti-microbial and anti-inflammatory activities. We are interested in heterocyclic compounds with a combination of thia-zolidinonyl and chromonyl groups, in the hope of discovering novel lead structures for the development of antifungal and antimicrobial agents. The title compound, (I), is readily synthesized by condensing 6-methyl-4-oxo-4H -chromene-3-carbaldehyde with p-chloroaniline and mercaptoacetic acid (Fittonet al., 1984).
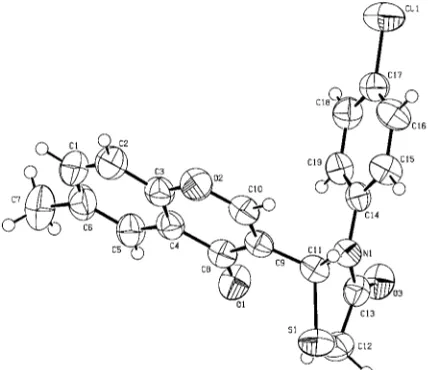
The crystal structure (Fig. 1) shows that the benzene ring of the 4-chlorophenyl substituent is not coplanar with the thia-zolidine ring, and the thiathia-zolidine ring is also not coplanar
[image:1.610.226.440.537.722.2]Received 22 March 2005 Accepted 17 June 2005 Online 24 June 2005
Figure 1
with the pyran ring [the dihedral angles between the thiazo-lidine ring and pyran ring is 85.69 (4)] so they cannot form a
conjugated system in the solid state (Table 1).
Experimental
A mixture of 6-methyl-4-oxo-4H-chromene-3-carbaldehyde (5 mmol), 4-chlorobenzenamine (5 mmol) andp-toluenesulfonic acid (10 mg, 0.058 mmol) was refluxed and stirred in benzene (40 ml) for 1 h, and then mercaptoacetic acid (25 mmol) was added. The reactant was refluxed for another 8 h. The solvent was removedin vacuo. The resulting residue was washed with ethanol to afford the crude product, which was then recrystallized from ethanol to give colorless crystals. 1H NMR (400 MHz, CDCl
3): 2.45 (s, 3H), 3.77 (d, J=
15.6 Hz, 1H), 4.11 (dd,J= 15.6 Hz,J= 1.4 Hz, 1H), 6.08 (s, 1H), 7.25– 7.32 (m, 5H), 7.47 (dd,J= 8.6 Hz,J= 1.8 Hz, 1H), 7.74 (s, 1H), 7.99 (s, 1H); MS (EI, m/z): 373 (M+ + 1), 372 (M+); elemental analysis calculated for C19H14ClNO3S: C 61.37, H 3.79, N 3.77%; found: C
61.46, H 3.78, N 3.71%.
Crystal data
C19H14ClNO3S
Mr= 371.82
Monoclinic,P21=n
a= 11.1830 (9) A˚
b= 13.9851 (10) A˚
c= 12.3419 (9) A˚
= 116.445 (1)
V= 1728.2 (2) A˚3
Z= 4
Dx= 1.429 Mg m
3 MoKradiation Cell parameters from 3801
reflections
= 2.4–27.7 = 0.36 mm1
T= 293 (2) K Block, colorless 0.400.380.34 mm
Data collection
Bruker SMART CCD area-detector diffractometer
’and!scans
Absorption correction: multi-scan (SADABS; Sheldrick, 1997)
Tmin= 0.870,Tmax= 0.888 9946 measured reflections
3747 independent reflections 2974 reflections withI> 2(I)
Rint= 0.018
max= 27.0
h=13!14
k=17!13
l=14!15
Refinement
Refinement onF2
R[F2> 2(F2)] = 0.044
wR(F2) = 0.134
S= 1.05 3747 reflections 227 parameters
H-atom parameters constrained
w= 1/[2(F
o2) + (0.0722P)2 + 0.3678P]
whereP= (Fo2+ 2Fc2)/3 (/)max= 0.001
max= 0.31 e A˚
3 min=0.20 e A˚
[image:2.610.313.566.106.221.2]3
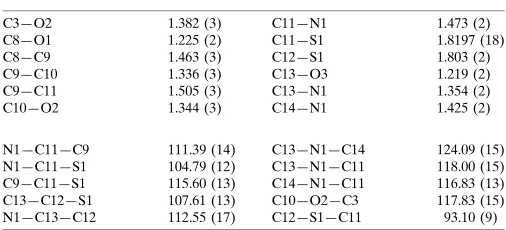
Table 1
Selected geometric parameters (A˚ ,).
C3—O2 1.382 (3)
C8—O1 1.225 (2)
C8—C9 1.463 (3)
C9—C10 1.336 (3)
C9—C11 1.505 (3)
C10—O2 1.344 (3)
C11—N1 1.473 (2)
C11—S1 1.8197 (18)
C12—S1 1.803 (2)
C13—O3 1.219 (2)
C13—N1 1.354 (2)
C14—N1 1.425 (2)
N1—C11—C9 111.39 (14)
N1—C11—S1 104.79 (12)
C9—C11—S1 115.60 (13)
C13—C12—S1 107.61 (13)
N1—C13—C12 112.55 (17)
C13—N1—C14 124.09 (15)
C13—N1—C11 118.00 (15)
C14—N1—C11 116.83 (13)
C10—O2—C3 117.83 (15)
C12—S1—C11 93.10 (9)
All H atoms were initially located in a difference Fourier map. The methyl H atoms were then constrained to an ideal geometry, with C— H distances of 0.96 A˚ andUiso(H) = 1.5Ueq(C), but each group was
allowed to rotate freely about its C—C bond. All other H atoms were placed in geometrically idealized positions and constrained to ride on their parent atoms, with C—H distances in the range 0.93–0.98 A˚ and withUiso(H) = 1.2Ueq(C).
Data collection:SMART(Bruker, 2003); cell refinement:SAINT
(Bruker, 2003); data reduction:SAINT; program(s) used to solve structure:SHELXS97(Sheldrick, 1997a); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997a); molecular graphics:
SHELXTL(Sheldrick, 1997b); software used to prepare material for publication:SHELXTL.
The present work was supported by the National Key Project for Basic Research (Nos. 2003CB114400 and 2002CCA00500), the National Natural Science Foundation of China (Nos. 20172017 and 20203009), and the Program for New Century Excellent Talents in University of China.
References
Bruker (2003).SAINT(Version 6.45) andSMART(Version 5.628). Bruker AXS Inc., Madison, Wisconsin, USA.
Fitton, A. O., Humphrey, G. L., Kosmirak, M., Suschitzy, H. & Suschitzy, J. L. (1984).J. Chem. Res. (S),8, 248–249.
Sheldrick, G. M. (1997a). SHELXS97 and SHELXL97. University of Go¨ttingen, Germany.
supporting information
sup-1
Acta Cryst. (2005). E61, o2261–o2262
supporting information
Acta Cryst. (2005). E61, o2261–o2262 [https://doi.org/10.1107/S1600536805019355]
3-[3-(4-Chlorophenyl)-4-oxothiazolidin-2-yl]-6-methyl-4
H
-benzopyran-4-one
Zhong-Zhen Zhou, Wei Huang, Pei-Liang Zhao, Qiong Chen and Guang-Fu Yang
3-[3-(4-Chlorophenyl)-4-oxothiazolidin-2-yl]-6-methyl-4H-benzopyran-4-one
Crystal data
C19H14ClNO3S
Mr = 371.82 Monoclinic, P21/n Hall symbol: -P 2yn
a = 11.1830 (9) Å
b = 13.9851 (10) Å
c = 12.3419 (9) Å
β = 116.445 (1)°
V = 1728.2 (2) Å3
Z = 4
F(000) = 768
Dx = 1.429 Mg m−3
Mo Kα radiation, λ = 0.71073 Å Cell parameters from 3801 reflections
θ = 2.4–27.7°
µ = 0.36 mm−1
T = 293 K Block, colorless 0.40 × 0.38 × 0.34 mm
Data collection
Bruker SMART CCD area-detector diffractometer
Radiation source: fine-focus sealed tube Graphite monochromator
φ and ω scans
Absorption correction: multi-scan (SADABS; Sheldrick, 1997)
Tmin = 0.870, Tmax = 0.888
9946 measured reflections 3747 independent reflections 2974 reflections with I > 2σ(I)
Rint = 0.018
θmax = 27.0°, θmin = 2.1°
h = −13→14
k = −17→13
l = −14→15
Refinement
Refinement on F2 Least-squares matrix: full
R[F2 > 2σ(F2)] = 0.044
wR(F2) = 0.134
S = 1.05 3747 reflections 227 parameters 0 restraints
Primary atom site location: structure-invariant direct methods
Secondary atom site location: difference Fourier map
Hydrogen site location: inferred from neighbouring sites
H-atom parameters constrained
w = 1/[σ2(F
o2) + (0.0722P)2 + 0.3678P] where P = (Fo2 + 2Fc2)/3
(Δ/σ)max = 0.001 Δρmax = 0.31 e Å−3 Δρmin = −0.20 e Å−3
Special details
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger.
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
x y z Uiso*/Ueq
C1 0.4528 (3) −0.13740 (18) 0.4983 (3) 0.0791 (7)
H1 0.4846 −0.1741 0.4539 0.095*
C2 0.3325 (3) −0.09187 (18) 0.4386 (2) 0.0777 (7)
H2 0.2826 −0.0983 0.3554 0.093*
C3 0.2867 (2) −0.03594 (14) 0.50518 (18) 0.0572 (5)
C4 0.3593 (2) −0.02567 (13) 0.62870 (18) 0.0518 (4)
C5 0.4808 (2) −0.07449 (15) 0.6867 (2) 0.0605 (5)
H5 0.5301 −0.0688 0.7701 0.073*
C6 0.5295 (3) −0.13087 (16) 0.6235 (3) 0.0712 (6)
C7 0.6605 (3) −0.1828 (2) 0.6854 (3) 0.0970 (9)
H7A 0.7156 −0.1672 0.6465 0.146*
H7B 0.6446 −0.2505 0.6804 0.146*
H7C 0.7049 −0.1639 0.7688 0.146*
C8 0.31050 (18) 0.03658 (13) 0.69621 (16) 0.0482 (4)
C9 0.18190 (17) 0.08215 (13) 0.62177 (16) 0.0458 (4)
C10 0.1207 (2) 0.06672 (14) 0.50249 (17) 0.0533 (5)
H10 0.0402 0.0982 0.4582 0.064*
C11 0.11957 (17) 0.14996 (13) 0.67667 (16) 0.0470 (4)
H11 0.0369 0.1750 0.6117 0.056*
C12 0.2199 (2) 0.15738 (16) 0.91390 (18) 0.0613 (5)
H12A 0.1922 0.1853 0.9711 0.074*
H12B 0.2903 0.1114 0.9566 0.074*
C13 0.26937 (19) 0.23403 (14) 0.85850 (17) 0.0510 (4)
C14 0.24323 (17) 0.29129 (12) 0.66110 (16) 0.0448 (4)
C15 0.14329 (19) 0.33736 (15) 0.56497 (18) 0.0566 (5)
H15 0.0555 0.3325 0.5536 0.068*
C16 0.1712 (2) 0.39050 (15) 0.48545 (19) 0.0617 (5)
H16 0.1030 0.4206 0.4198 0.074*
C17 0.3012 (2) 0.39843 (14) 0.50436 (18) 0.0540 (5)
C18 0.4035 (2) 0.35463 (15) 0.6009 (2) 0.0585 (5)
H18 0.4912 0.3612 0.6128 0.070*
C19 0.37468 (18) 0.30067 (14) 0.68015 (18) 0.0521 (4)
H19 0.4431 0.2708 0.7459 0.062*
Cl1 0.33525 (7) 0.46507 (5) 0.40205 (6) 0.0827 (2)
N1 0.20848 (14) 0.23126 (10) 0.73598 (13) 0.0460 (4)
O1 0.37359 (14) 0.05043 (12) 0.80520 (12) 0.0646 (4)
O2 0.16562 (16) 0.00957 (11) 0.44096 (12) 0.0643 (4)
O3 0.35456 (14) 0.29216 (11) 0.91807 (12) 0.0630 (4)
supporting information
sup-3
Acta Cryst. (2005). E61, o2261–o2262
Atomic displacement parameters (Å2)
U11 U22 U33 U12 U13 U23
C1 0.107 (2) 0.0602 (14) 0.0852 (17) 0.0015 (13) 0.0566 (17) −0.0136 (12)
C2 0.111 (2) 0.0625 (14) 0.0649 (14) −0.0052 (14) 0.0436 (15) −0.0106 (11)
C3 0.0684 (13) 0.0472 (10) 0.0547 (12) −0.0082 (9) 0.0263 (10) −0.0041 (9)
C4 0.0568 (11) 0.0422 (9) 0.0563 (11) −0.0088 (8) 0.0250 (9) −0.0038 (8)
C5 0.0605 (12) 0.0524 (11) 0.0658 (13) −0.0023 (9) 0.0256 (10) −0.0070 (10) C6 0.0778 (15) 0.0497 (11) 0.0931 (17) −0.0011 (11) 0.0444 (14) −0.0072 (12)
C7 0.0904 (19) 0.0806 (18) 0.121 (2) 0.0184 (15) 0.0482 (18) −0.0154 (17)
C8 0.0468 (9) 0.0482 (10) 0.0430 (10) −0.0087 (7) 0.0140 (8) −0.0033 (7)
C9 0.0446 (9) 0.0428 (9) 0.0437 (9) −0.0099 (7) 0.0139 (8) −0.0002 (7)
C10 0.0549 (11) 0.0491 (10) 0.0464 (10) −0.0076 (8) 0.0139 (9) 0.0005 (8)
C11 0.0412 (9) 0.0473 (10) 0.0460 (9) −0.0051 (7) 0.0136 (8) 0.0033 (8)
C12 0.0632 (12) 0.0746 (14) 0.0433 (10) 0.0026 (10) 0.0212 (9) 0.0020 (9)
C13 0.0467 (10) 0.0544 (11) 0.0461 (10) 0.0070 (8) 0.0153 (8) −0.0035 (8)
C14 0.0442 (9) 0.0375 (8) 0.0472 (9) −0.0035 (7) 0.0156 (8) −0.0056 (7)
C15 0.0393 (9) 0.0632 (12) 0.0579 (11) −0.0027 (8) 0.0132 (8) 0.0082 (9)
C16 0.0538 (11) 0.0619 (12) 0.0551 (12) −0.0038 (9) 0.0114 (9) 0.0106 (9)
C17 0.0618 (12) 0.0496 (10) 0.0501 (11) −0.0118 (9) 0.0245 (9) −0.0073 (8)
C18 0.0482 (10) 0.0572 (12) 0.0736 (14) −0.0056 (9) 0.0301 (10) −0.0062 (10)
C19 0.0424 (9) 0.0488 (10) 0.0561 (11) 0.0011 (8) 0.0138 (8) −0.0015 (8)
Cl1 0.0929 (5) 0.0925 (5) 0.0693 (4) −0.0208 (3) 0.0422 (4) 0.0058 (3)
N1 0.0444 (8) 0.0453 (8) 0.0407 (8) −0.0039 (6) 0.0120 (6) −0.0017 (6)
O1 0.0536 (8) 0.0795 (10) 0.0478 (8) 0.0087 (7) 0.0109 (7) −0.0079 (7)
O2 0.0756 (10) 0.0631 (9) 0.0442 (8) −0.0029 (7) 0.0176 (7) −0.0059 (6)
O3 0.0557 (8) 0.0698 (9) 0.0472 (7) −0.0035 (7) 0.0083 (6) −0.0137 (7)
S1 0.0587 (3) 0.0748 (4) 0.0582 (3) −0.0101 (2) 0.0274 (3) 0.0077 (2)
Geometric parameters (Å, º)
C1—C2 1.369 (4) C11—N1 1.473 (2)
C1—C6 1.397 (4) C11—S1 1.8197 (18)
C1—H1 0.93 C11—H11 0.98
C2—C3 1.387 (3) C12—C13 1.503 (3)
C2—H2 0.93 C12—S1 1.803 (2)
C3—C4 1.379 (3) C12—H12A 0.97
C3—O2 1.382 (3) C12—H12B 0.97
C4—C5 1.400 (3) C13—O3 1.219 (2)
C4—C8 1.468 (3) C13—N1 1.354 (2)
C5—C6 1.380 (3) C14—C15 1.375 (3)
C5—H5 0.93 C14—C19 1.388 (3)
C6—C7 1.503 (4) C14—N1 1.425 (2)
C7—H7A 0.96 C15—C16 1.373 (3)
C7—H7B 0.96 C15—H15 0.93
C7—H7C 0.96 C16—C17 1.370 (3)
C8—O1 1.225 (2) C16—H16 0.93
C9—C10 1.336 (3) C17—Cl1 1.742 (2)
C9—C11 1.505 (3) C18—C19 1.382 (3)
C10—O2 1.344 (3) C18—H18 0.93
C10—H10 0.93 C19—H19 0.93
C2—C1—C6 122.5 (2) N1—C11—H11 108.3
C2—C1—H1 118.7 C9—C11—H11 108.3
C6—C1—H1 118.7 S1—C11—H11 108.3
C1—C2—C3 118.5 (2) C13—C12—S1 107.61 (13)
C1—C2—H2 120.8 C13—C12—H12A 110.2
C3—C2—H2 120.8 S1—C12—H12A 110.2
C4—C3—O2 122.00 (19) C13—C12—H12B 110.2
C4—C3—C2 121.5 (2) S1—C12—H12B 110.2
O2—C3—C2 116.5 (2) H12A—C12—H12B 108.5
C3—C4—C5 118.3 (2) O3—C13—N1 124.19 (19)
C3—C4—C8 120.32 (19) O3—C13—C12 123.27 (17)
C5—C4—C8 121.37 (18) N1—C13—C12 112.55 (17)
C6—C5—C4 121.8 (2) C15—C14—C19 119.64 (18)
C6—C5—H5 119.1 C15—C14—N1 118.95 (16)
C4—C5—H5 119.1 C19—C14—N1 121.35 (16)
C5—C6—C1 117.4 (2) C16—C15—C14 120.92 (18)
C5—C6—C7 122.0 (3) C16—C15—H15 119.5
C1—C6—C7 120.7 (2) C14—C15—H15 119.5
C6—C7—H7A 109.5 C17—C16—C15 118.93 (19)
C6—C7—H7B 109.5 C17—C16—H16 120.5
H7A—C7—H7B 109.5 C15—C16—H16 120.5
C6—C7—H7C 109.5 C16—C17—C18 121.42 (19)
H7A—C7—H7C 109.5 C16—C17—Cl1 118.51 (16)
H7B—C7—H7C 109.5 C18—C17—Cl1 120.06 (16)
O1—C8—C9 123.03 (18) C17—C18—C19 119.40 (18)
O1—C8—C4 122.65 (18) C17—C18—H18 120.3
C9—C8—C4 114.31 (16) C19—C18—H18 120.3
C10—C9—C8 119.96 (18) C18—C19—C14 119.66 (18)
C10—C9—C11 119.03 (17) C18—C19—H19 120.2
C8—C9—C11 120.96 (15) C14—C19—H19 120.2
C9—C10—O2 125.56 (19) C13—N1—C14 124.09 (15)
C9—C10—H10 117.2 C13—N1—C11 118.00 (15)
O2—C10—H10 117.2 C14—N1—C11 116.83 (13)
N1—C11—C9 111.39 (14) C10—O2—C3 117.83 (15)
N1—C11—S1 104.79 (12) C12—S1—C11 93.10 (9)
C9—C11—S1 115.60 (13)
C6—C1—C2—C3 1.1 (4) C19—C14—C15—C16 1.8 (3)
C1—C2—C3—C4 −0.1 (3) N1—C14—C15—C16 −175.39 (18)
C1—C2—C3—O2 179.3 (2) C14—C15—C16—C17 −1.1 (3)
O2—C3—C4—C5 179.85 (18) C15—C16—C17—C18 −0.1 (3)
C2—C3—C4—C5 −0.9 (3) C15—C16—C17—Cl1 179.38 (16)
supporting information
sup-5
Acta Cryst. (2005). E61, o2261–o2262
C2—C3—C4—C8 177.94 (19) Cl1—C17—C18—C19 −178.91 (15)
C3—C4—C5—C6 0.8 (3) C17—C18—C19—C14 0.1 (3)
C8—C4—C5—C6 −177.99 (19) C15—C14—C19—C18 −1.3 (3)
C4—C5—C6—C1 0.2 (3) N1—C14—C19—C18 175.81 (17)
C4—C5—C6—C7 179.5 (2) O3—C13—N1—C14 5.0 (3)
C2—C1—C6—C5 −1.2 (4) C12—C13—N1—C14 −175.34 (16)
C2—C1—C6—C7 179.5 (3) O3—C13—N1—C11 172.67 (17)
C3—C4—C8—O1 −177.49 (19) C12—C13—N1—C11 −7.6 (2)
C5—C4—C8—O1 1.3 (3) C15—C14—N1—C13 −132.5 (2)
C3—C4—C8—C9 1.2 (3) C19—C14—N1—C13 50.3 (3)
C5—C4—C8—C9 179.97 (17) C15—C14—N1—C11 59.6 (2)
O1—C8—C9—C10 177.47 (18) C19—C14—N1—C11 −117.53 (19)
C4—C8—C9—C10 −1.2 (2) C9—C11—N1—C13 −107.42 (18)
O1—C8—C9—C11 0.1 (3) S1—C11—N1—C13 18.27 (19)
C4—C8—C9—C11 −178.56 (15) C9—C11—N1—C14 61.20 (19)
C8—C9—C10—O2 1.4 (3) S1—C11—N1—C14 −173.11 (12)
C11—C9—C10—O2 178.82 (17) C9—C10—O2—C3 −1.4 (3)
C10—C9—C11—N1 −119.16 (18) C4—C3—O2—C10 1.4 (3)
C8—C9—C11—N1 58.2 (2) C2—C3—O2—C10 −177.94 (19)
C10—C9—C11—S1 121.40 (16) C13—C12—S1—C11 15.02 (15)
C8—C9—C11—S1 −61.23 (19) N1—C11—S1—C12 −18.20 (13)
S1—C12—C13—O3 172.49 (16) C9—C11—S1—C12 104.80 (14)