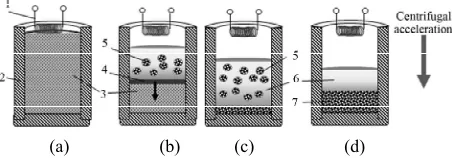
Cast Intermetallic Alloys and Composites Based on Them by Combined Centrifugal Casting—SHS Process
Full text
Figure



Related documents
Free Download Write Can anyone know an app for download named - Windows Central Hours exe NT Kernel Disbursement Atmosphere Corporation ZwOpenObjectAuditAlarm 0x805F2C16
Human elements involved in the problem Support systems surrounding the problem Tracking systems related to the problem.. Institutional process for managing
Passive tags require no internal power source, thus being pure passive devices (they are only active when a reader is nearby to power them), whereas active tags require
1) To capture the characteristics of write operations that are responsible for a large portion of I/O cost in flash memory, we separately analyze the temporal locality of
UDL provides the following three guiding principles to consider when planning lessons so that children are properly stimulated in ways that allow for learning: (1) multiple means of
The synthesis and characterization of new mixed-metal 3d/4f clusters has attracted immense interest over the last few decades, due to their fascinating structural features
Applying this technique to Martian geology is attractive for several rea- sons: (1) cosmic rays are ubiquitous and generate secondary muons in any atmosphere; (2) a preliminary